Group 17 Elements: Halogens Family
What are Halogens?
Halogens are the elements of Group 17 of the periodic table. They are reactive non-metals and include fluorine (F), chlorine (Cl), bromine (Br), iodine (I) and astatine (At). The term halogen comes from Greek: halo = salt and genes = producing, meaning "salt-forming".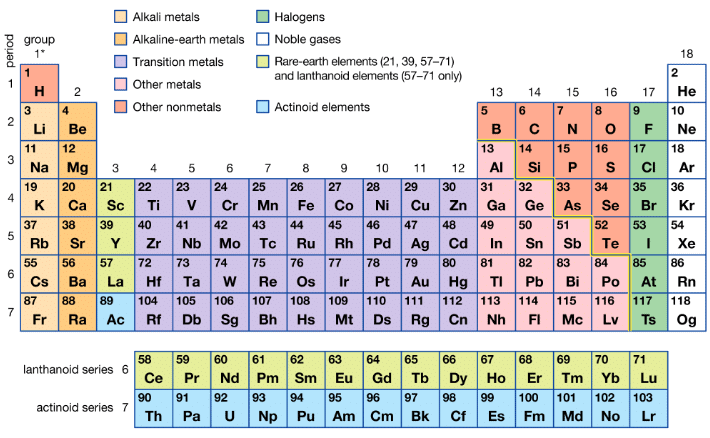 Group 17 - Halogens
Group 17 - Halogens
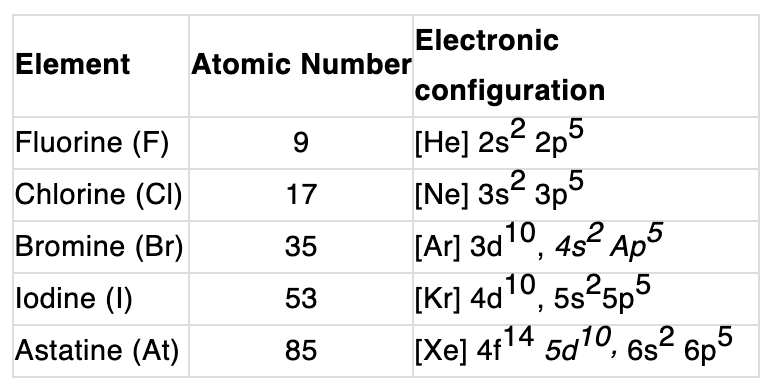
All halogens have seven electrons in their outermost shell; the general valence-shell electronic configuration is ns² np⁵. They are one electron short of the noble gas configuration and therefore are commonly found in the -1 oxidation state. These elements show strong similarities with each other, a regular gradation of properties down the group and high reactivity. Astatine is the only naturally occurring radioactive member of the group.
General Physical Properties of Group 17 Elements
- Electronic configuration: The valence shell configuration is ns² np⁵, which explains their high tendency to gain one electron and form -1 anions.
- Physical state: Intermolecular van der Waals forces increase down the group. As a result, F₂ and Cl₂ are gases at room temperature, Br₂ is a volatile liquid and I₂ is a solid.
- Atomicity: All the halogens exist as diatomic molecules (X₂) in the elemental state.
- Abundance: Being highly reactive, halogens are not found free in nature. Their relative natural abundance (in minerals and compounds) generally decreases down the group and is often quoted in the order:
F2 > Cl2 > Br2 > I2 - Colour: Halogen molecules absorb visible light and therefore are coloured. Typical colours are: F₂ - very pale yellow, Cl₂ - yellowish-green, Br₂ - reddish-brown, I₂ - deep violet (purple).
- Non-metallic / metallic character: All halogens are non-metals; the tendency to show metallic character (and to form positive oxidation states) increases slightly down the group.
- Bond length and bond energy: Bond length increases down the group (F-F < Cl-Cl < Br-Br < I-I). Generally bond dissociation energy decreases down the group. The F-F bond is weaker than expected because of strong lone-pair repulsion between the small fluorine atoms.
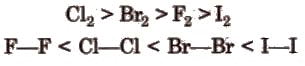
- Density: Density of the elemental halogens increases down the group:
F₂ < Cl₂ < Br₂ < I₂. - Ionisation enthalpy: Halogens have relatively high ionisation enthalpies; these decrease down the group.
- Electron affinity: Halogens have large (exothermic) electron affinities; the experimentally observed order is Cl > F > Br > I. Fluorine's electron affinity is lower than expected because addition of an electron to very small F atom causes strong electron-electron repulsion in the compact 2p shell.
- Reduction potentials and oxidising power: Standard reduction potentials (E°red) of X₂/X⁻ are positive; oxidising power decreases down the group: F₂ > Cl₂ > Br₂ > I₂. Fluorine is the strongest oxidising agent and reacts even with water and many metals; it is prepared industrially by electrolysis of fluoride mixtures under special conditions.
- Solubility in water: Molecular halogens are sparingly soluble in water. Solubility of X₂ in water typically decreases down the group. Iodine's solubility in water is greatly enhanced by addition of iodide ions (forming triiodide, I₃⁻): KI + I₂ ⇌ KI₃ ⇌ K⁺ + I₃⁻. Iodine forms a characteristic blue complex with starch (I₃⁻ + starch → blue complex).
Chemical Properties of Group 17 Elements
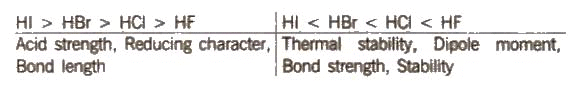
- Hydrides (HX): Halogen hydrides are binary compounds of hydrogen and halogen. Hydrogen fluoride (HF) is a low-boiling liquid due to strong intermolecular hydrogen bonding. Hydrogen chloride (HCl), hydrogen bromide (HBr) and hydrogen iodide (HI) are gases at room temperature. The typical trend of boiling points is HF > HI > HBr > HCl, where HF is anomalously high because of hydrogen bonding.
- Oxides: Fluorine forms oxides such as OF₂ and O₂F₂; OF₂ is the more stable oxide at standard conditions. Chlorine forms several oxides and oxyacids (Cl₂O, Cl₂O₃, ClO₂, Cl₂O₇ etc.); ClO₂ is used industrially and ClO₂ is a bleaching agent. Bromine and iodine form oxides that are generally less stable and many exist only at low temperatures; iodine pentoxide (I₂O₅) is a stable oxide used as an oxidising agent in analysis (for example, for estimation of CO).
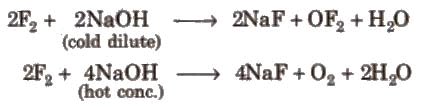
- Reaction with alkali: Halogens react with alkali (e.g., NaOH) to give different oxyhalide species depending on conditions. Dilute cold alkali gives hypohalite and halide; concentrated and hot alkali gives halate and halide.
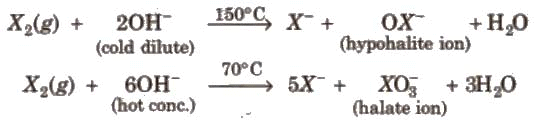
- Oxo-acids of halogens: Many halogens form oxyacids (hypohalous, halous, halic acids, etc.). Fluorine is so electronegative and lacks accessible d-orbitals that higher oxoacids analogous to other halogens are not well formed; only a few unstable species such as HOF (hypofluorous acid) are known. For heavier halogens, several oxoacids exist in aqueous media or as salts. Due to the inert-pair effect, odd lower oxidation states (for example +3) of heavier halogens are less stable. For oxoacids of the same central halogen, acidity and thermal stability increase with the number of bonded oxygen atoms. The general acidic strength order for a given oxidation series follows the increasing electronegativity/electron-withdrawing effect of oxygen atoms.

Interhalogen Compounds
When two different halogens combine, they form interhalogen compounds. These are covalent and generally diamagnetic molecules. They are usually more reactive than the constituent halogen molecules (except fluorine which is the most reactive element). Typical general formulas are XY, XY₃, XY₅ and XY₇, where X is the larger, less electronegative halogen and Y is the more electronegative one.
- Shapes: XY₃ (T-shaped), XY₅ (square pyramidal) and IF₇ (pentagonal bipyramidal).
- BrF₃ is T-shaped because it has three bonding pairs and two lone pairs around bromine.
- Interhalogens like ICl are often more reactive than the corresponding dihalogen (I₂) because the X-Y bond can be weaker or more polar.
- ClF₃ and BrF₃ are used industrially, for example in uranium chemistry. A reaction used in uranium processing is:
U(s) + 3ClF₃(l) → UF₆(g) + 3ClF(g)
Pseudohalogens and Pseudohalides
Species that behave chemically like halogens are called pseudohalogens. Their corresponding anions are called pseudohalide ions. Examples include:
| Pseudohalogen | Pseudohalide ion |
|---|---|
| (CN)₂ - cyanogen | CN⁻ - cyanide |
| (OCN)₂ - oxycyanogen | OCN⁻ - cyanate |
| (SCN)₂ - thiocyanogen | SCN⁻ - thiocyanate |
Compounds of Group 17 Elements
1. Chlorine (Cl₂)
(i) Occurrence
The most important source of chlorine is common salt (NaCl). Chlorine is also present in seawater (as chloride) and in mineral deposits (rock salt).
(ii) Preparation of chlorine
- By oxidation of concentrated HCl with MnO₂ (laboratory):
4NaCl + MnO₂ + 4H₂SO₄ → 4NaHSO₄ + MnCl₂ + 2H₂O + Cl₂ - Weldon's process (laboratory/old industrial):
MnO₂ + 4HCl → MnCl₂ + 2H₂O + Cl₂ - Deacon's process (industrial):Hydrogen chloride is oxidised by oxygen in the presence of a catalyst (e.g., CuCl₂) at elevated temperature:
4HCl + O₂ → 2Cl₂ + 2H₂O - Electrolytic process (industrial):Electrolysis of brine (sodium chloride solution) in cells (e.g., Nelson cell, diaphragm cell or membrane cell) yields chlorine at the anode and hydrogen and sodium hydroxide at the cathode.
Half-reactions:
At anode: 2Cl⁻ → Cl₂ + 2e⁻
At cathode (in aqueous medium): 2H₂O + 2e⁻ → H₂ + 2OH⁻
Overall: 2NaCl + 2H₂O → 2NaOH + H₂ + Cl₂
(iii) Physical properties
Chlorine is a yellowish-green gas at room temperature, has a pungent suffocating odour, is poisonous and is moderately soluble in water. Aqueous chlorine is called chlorine water.
(iv) Chemical reactions of chlorine
- Action of water (bleaching & disinfection):
Chlorine reacts with water to give a mixture of hydrochloric acid and hypochlorous acid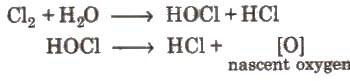
The bleaching action of chlorine is due to the oxidative action of hypochlorous acid and related species; it can be permanent if oxidation of chromophores occurs.
- Action of hydrogen:
Chlorine reacts explosively with hydrogen in the presence of light or at elevated temperature to form hydrogen chloride:
- Displacement reactions:
More reactive halogens displace less reactive halide ions from their salts. For example:
- Action of cold NaOH (bleaching water formation):
Cl₂ + 2NaOH → NaCl + NaOCl + H₂O (Javelle or bleaching water contains sodium hypochlorite)
- Action on hydrogen sulphide (H₂S):
Cl₂ oxidises H₂S to sulphur or sulphuric acid depending on conditions; it can be used to remove H₂S from gases.
- Action of dry SO₂:
Chlorine oxidises SO₂ to SO₃ under certain conditions; reactions depend on catalysts and conditions.
- Action of carbon monoxide (CO):
Chlorine can oxidise CO to CO₂ under appropriate conditions, often in the presence of catalysts.
- Oxidising properties:
Chlorine is a good oxidising agent and can oxidise many reducing substances.
- Reaction with ammonia:
Chlorine reacts with ammonia to give bleaching compounds and nitrogen oxides depending on conditions. For example, controlled reaction gives nitrogen and hydrogen chloride; other conditions may give chloramines.
- Chromyl chloride test: When a mixture of chloride and solid K2Cr2O7 is heated with concentrated H2SO4 in a dry test tube, deep red vapours of chromyl chloride are evolved.
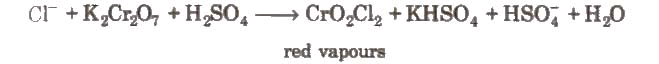 When these vapours are passed through NaOH solution, the solution becomes yellow due to the formation of sodium chromate.
When these vapours are passed through NaOH solution, the solution becomes yellow due to the formation of sodium chromate.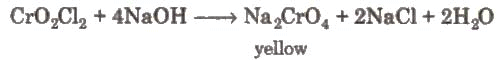 The yellow solution is neutralised with acetic acid and on the addition of lead acetate gives a yellow precipitate of lead chromate.
The yellow solution is neutralised with acetic acid and on the addition of lead acetate gives a yellow precipitate of lead chromate.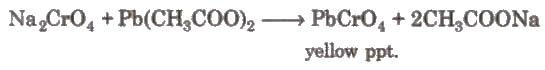
(v) Uses of chlorine
- Used as a bleaching agent and disinfectant (water chlorination).
- Used in manufacture of organic compounds such as chloroform (CHCl₃), carbon tetrachloride (CCl₄), DDT and other chlorinated organics (industrial uses are controlled for environmental reasons).
- Used in production of bleaching powder and many inorganic chlorides.
2. Hydrochloric acid (HCl)
(i) Preparation
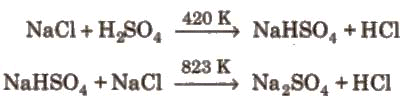
Hydrogen chloride gas is produced in the laboratory by reaction of a chloride salt with concentrated sulphuric acid or by direct combination of hydrogen and chlorine. In industry, HCl is produced as a by-product in many chemical processes and can be dissolved in water to give hydrochloric acid.
(ii) Properties
Hydrogen chloride is a colourless gas with a pungent odour; it is extremely soluble in water and ionises fully in aqueous solution:
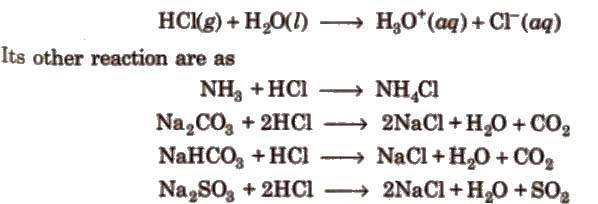
HCl(aq) ⇌ H⁺ + Cl⁻ (in water). Concentrated HCl is a strong acid. Noble metals such as gold and platinum dissolve only in mixtures such as aqua regia (three parts concentrated HCl : one part concentrated HNO₃), which generate oxidising chlorine-containing species capable of oxidising and complexing these metals.
(iii) Uses
- Used in the manufacture of chlorides and in many industrial processes (textile and dyeing industries, pickling of steel).
- Used in laboratory and industrial contexts as a strong acid and for pH control, and in processes for extraction of glue from animal tissues and bones.
3. Iodine (I₂)
- Sources: Major sources include marine algae (kelp, e.g., laminaria), whose ashes (kelp) contain iodide salts; natural mineral deposits such as caliche (crude Chile saltpetre) containing iodates can also be used as sources.
- Extraction and purification: Iodine is commonly obtained by treating iodide-containing materials with oxidising agents and then purifying by sublimation.
- Chemical behaviour: Iodine has low solubility in water but dissolves in organic solvents and forms more soluble triiodide in presence of iodide. The tincture of iodine is a solution of I₂ and KI in alcohol and is used as an antiseptic.
4. Oxoacids of Halogens
Oxoacids are acids in which halogen atoms are bonded to oxygen atoms. Fluorine, due to its extreme electronegativity and lack of accessible d-orbitals, does not form higher oxoacids analogous to other halogens except for a few unstable species such as HOF. Chlorine, bromine and iodine form several oxoacids (hypohalous, halous, halic acids etc.), many of which are stable only in solution or as salts. For a given halogen, acidic strength and thermal stability generally increase with the number of oxygen atoms attached to the halogen centre.
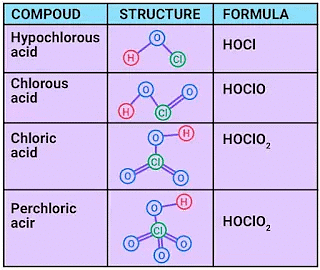 Oxoacids of Chlorine
Oxoacids of Chlorine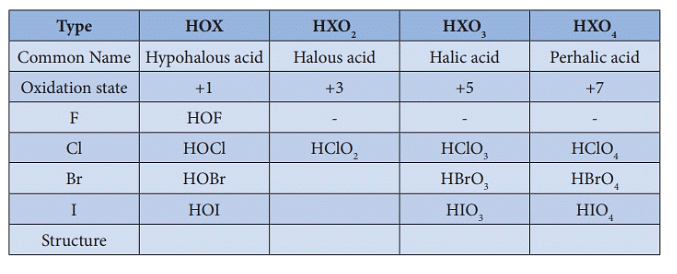 Oxoacids of Halogens
Oxoacids of HalogensImportant Points
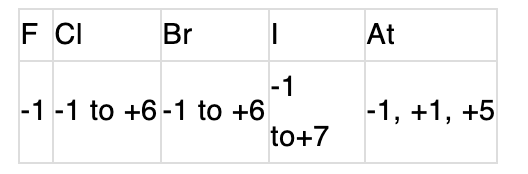
- Reactivity trend: Reactivity (ability to oxidise other species) of elemental halogens decreases down the group: F₂ > Cl₂ > Br₂ > I₂. Fluorine is the most reactive and oxidising.
- Common oxidation state: The most common oxidation state for halogens is -1. Positive oxidation states (+1, +3, +5, +7) occur for heavier halogens, particularly when bonded to oxygen or fluorine.
- Preparation of fluorine: Due to its high reactivity, fluorine is prepared by electrolysis of mixtures such as potassium hydrogen fluoride (KHF₂) in anhydrous HF; special corrosion-resistant materials such as Monel metal are used for electrodes and apparatus.
- Applications: Halogens and their compounds are widely used - disinfectants, bleaching agents, reagents in organic synthesis, pharmaceuticals, photographic chemicals, and in analytical chemistry (e.g., iodometry).
- Safety: Elemental halogens and many halogen compounds are toxic and corrosive; they must be handled with appropriate safety precautions and under proper ventilation.
Summary
The Group 17 elements (halogens) are a family of highly reactive non-metals with general valence electronic configuration ns² np⁵. They display systematic physical and chemical trends down the group: increasing atomic and ionic sizes, decreasing electronegativity and oxidising power, and increasing density and molar mass. Their chemistry includes formation of diatomic molecules, halides, hydrogen halides, oxoacids, interhalogen compounds and pseudohalide species. Industrially important processes include the electrolysis of brine to produce chlorine and caustic soda, manufacture and use of hypochlorite bleaches, and specialised fluorine chemistry carried out under controlled conditions.
FAQs on Group 17 Elements: Halogens Family
| 1. What are halogens? |  |
| 2. What are the general physical properties of Group 17 elements? |  |
| 3. What are the chemical properties of Group 17 elements? |  |
| 4. What are interhalogen compounds? |  |
| 5. What are chlorine and its compounds? |  |







