Atomic Masses and Composition of Nucleus
Nucleus
The entire positive charge and nearly the entire mass of atom is concentrated in a very small space called the nucleus of an atom. The nucleus consists of protons and neutrons. They are called nucleons.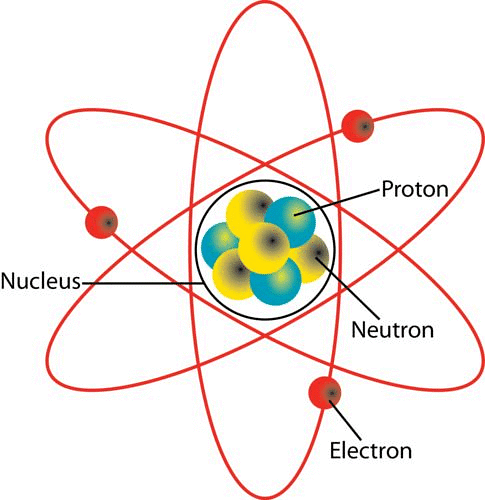 Atomic Nucleus
Atomic Nucleus
Terms Related to Nucleus:
(I) Atomic Number: The number of protons in the nucleus of an atom of the element is called atomic number (Z) of the element.
(II) Mass Number: The total number of protons and neutrons present inside the nucleus of an atom of the element is called mass number (A) of the element.
(III) Nuclear Size: The radius of the nucleus R ∝ A1/3
⇒ R = Ro A1/3
where, Ro = 1.1 * 10-15 m is an empirical constant.
(IV) Nuclear Density: Nuclear density is independent of mass number and therefore same for all nuclei.
ρ = mass of nucleus / volume of nucleus ⇒ ρ = 3m / 4π R3o
where, m = average mass of a nucleon.
(V) Atomic Mass Unit: It is defined as 1 / 12th the mass of carbon nucleus.
It is abbreviated as amu and often denoted by u. Thus
1 amu = 1.992678 * 10-26 / 12 kg
= 1.6 * 10-27 kg = 931 Me V
Isotopes:
The atoms of an element having same atomic number but different mass numbers are called isotopes.
e.g., 1H1, 1H2, 1H3 are isotopes of hydrogen.
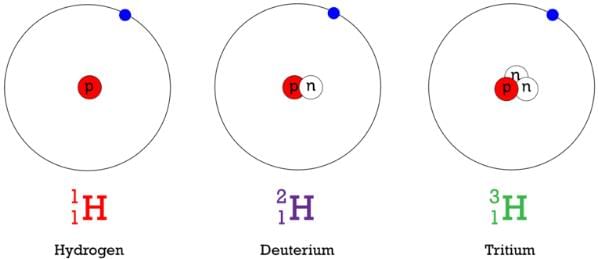 Fig: Isotopes of hydrogen
Fig: Isotopes of hydrogen
Isobars:
The atoms of different elements having same mass numbers but different atomic numbers, are called isobars.
e.g., 1H3, 2He3 and 10Na22, 10Ne22 are isobars.
Isotones:
The atoms of different elements having different atomic numbers and different mass numbers but having same number of neutrons, are called isotones.
e.g., 1H3, 2He4 and 6C14, 8O16 are isobars.
Isomers:
Atoms having the same mass number and the same atomic number but different radioactive properties are called isomers.
Nuclear Force:
The force acting inside the nucleus or acting between nucleons is called nuclear force. Nuclear forces are the strongest forces in nature.
- It is a very short range attractive force.
- It is non-central. non-conservative force.
- It is neither gravitational nor electrostatic force.
- It is independent of charge.
- It is 100 times that of electrostatic force and 1038 times that of gravitational force.
According to the Yukawa, the nuclear force acts between the nucleon due to continuous exchange of meson particles.
Mass Defect:
The difference between the sum of masses of all nucleons (M) mass of the nucleus (m) is called mass defect.
Mass Defect (Δm) = M - m = [Zmp + (A - Z)mn - mn]
Nuclear Binding Energy:
The minimum energy required to separate the nucleons up to an infinite distance from the nucleus, is called nuclear binding energy.
Nuclear binding energy per nucleon = Nuclear binding energy / Total number of nucleons
Binding energy, Eb = [Zmp + (A - Z) mn - mN]c2
Binding Energy Curve (BE/A vs Mass Number A)
This is the graph of binding energy per nucleon (BE/A) plotted against mass number (A).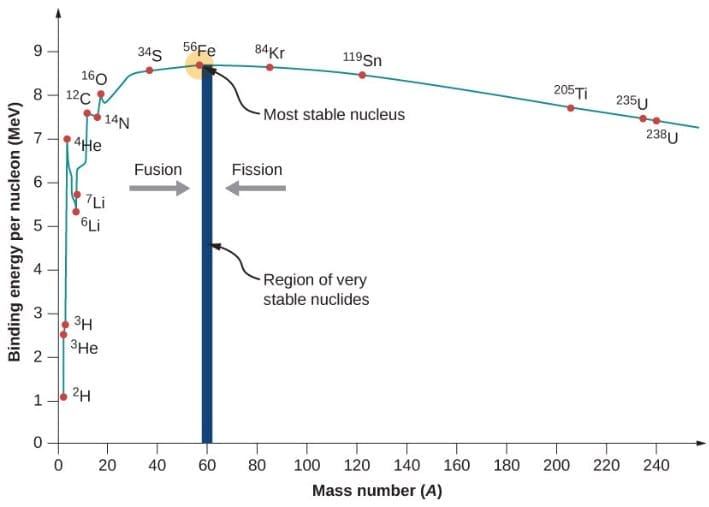
Key points from the graph:
- For very light nuclei (A < 20), BE/A is low and rises sharply - this means fusion of light nuclei releases energy
- BE/A rises and reaches a maximum of about 8.8 MeV at A ≈ 56 (Iron - Fe-56)
- Fe-56 is the most stable nucleus in nature
- For heavy nuclei (A > 56), BE/A slowly decreases - this means fission of heavy nuclei releases energy
- The curve explains why both fission and fusion release energy
Conclusion from the curve:
- Fission: Heavy nucleus (U-235) → medium nuclei → BE/A increases → energy released
- Fusion: Light nuclei (H) → heavier nucleus → BE/A increases → energy released
- Both processes move towards the peak (Fe-56) and release energy
Nuclear Stability - N/Z Ratio Rule
The stability of a nucleus depends on the ratio of neutrons (N) to protons (Z):
- For light nuclei (A < 40): Stable nuclei have N/Z ≈ 1
- For heavy nuclei (A > 40): Stable nuclei need N/Z > 1 (more neutrons needed to overcome proton repulsion)
- If N/Z is too high → nucleus undergoes β⁻ decay (neutron converts to proton)
- If N/Z is too low → nucleus undergoes β⁺ decay or electron capture (proton converts to neutron)
- If A > 83 → nucleus undergoes α decay to reduce both N and Z
Magic Numbers: Nuclei with proton or neutron number equal to 2, 8, 20, 28, 50, 82, or 126 are exceptionally stable. These are called magic numbers.
Packing Fraction (P)
p = (Exact nuclear mass) - (Mass number) / Mass number = M - A / M
- The larger the value of packing friction. greater is the stability of the nucleus.
- The nuclei containing even number of protons and even number of neutrons are most stable.
- The nuclei containing odd number of protons and odd number of neutrons are most unstable.
Solved Examples
Q1. The radius of a nucleus of mass number A is R. The radius of a nucleus of mass number 8A will be:
Solution:
R ∝ A^(1/3)
R₁/R₂ = (A₁/A₂)^(1/3)
R/R₂ = (A/8A)^(1/3) = (1/8)^(1/3) = 1/2 R₂ = 2R
Q2. If the nuclear radius of Al-27 is 3.6 fm, find the approximate nuclear radius of Cu-64.
Solution:
R ∝ A^(1/3)
R(Cu)/R(Al) = (64/27)^(1/3) = (64/27)^(1/3) = 4/3 R(Cu) = 3.6 × 4/3 = 4.8 fm
Q3. Which of the following has the highest binding energy per nucleon?
(A) ²H
(B) ⁵⁶Fe
(C) ²³⁵U
(D) ⁴He
Answer: (B)
Solution:
Fe-56 has the highest BE/nucleon of approximately 8.8 MeV. It sits at the peak of the binding energy curve and is the most stable nucleus.
Q4. The mass defect of a nucleus is 0.04 u. Find the binding energy in MeV.
Solution:
BE = Δm × 931.5 BE = 0.04 × 931.5 = 37.26 MeV
Q5. Which of the following statements is correct about nuclear forces?
(A) Nuclear force is a long range force
(B) Nuclear force is charge dependent
(C) Nuclear force becomes repulsive at very short distances (< 0.8 fm)
(D) Nuclear force follows inverse square law
Answer: (C)
Solution:
Nuclear force is attractive between 0.8 fm to ~2-3 fm but becomes repulsive below 0.8 fm. This repulsion prevents the nucleus from collapsing. It is charge independent and short range, and does NOT follow inverse square law.
FAQs on Atomic Masses and Composition of Nucleus
| 1. What is the atomic mass of a nucleus? |  |
| 2. How is the composition of a nucleus determined? |  |
| 3. How do you calculate the atomic mass of an element using the composition of its nucleus? |  |
| 4. Why is the composition of a nucleus important in understanding the properties of an element? |  |
| 5. Can the composition of a nucleus change through nuclear reactions? |  |






