Dual Nature Of Radiation And Matter
This chapter explores how light and matter exhibit both wave and particle characteristics. Understanding these dual natures is crucial for explaining phenomena like photoelectric effect and electron diffraction. These concepts form the foundation of quantum mechanics and are frequently tested in competitive physics examinations through numerical problems and conceptual questions.
1. Electron Emission
Free electrons exist in metals but cannot normally escape because the metal surface exerts an attractive force on them. To remove an electron from a metal surface, we must supply minimum energy called work function (φ₀).
1.1 Work Function
- Definition: Minimum energy required to remove an electron from the metal surface
- Symbol: φ₀ (phi zero)
- Unit: electron volt (eV) where 1 eV = 1.602 × 10⁻¹⁹ J
- Nature: Work function varies with different metals and depends on surface properties
- Physical meaning: Represents the attractive pull of positive ions on free electrons
1.2 Methods of Electron Emission
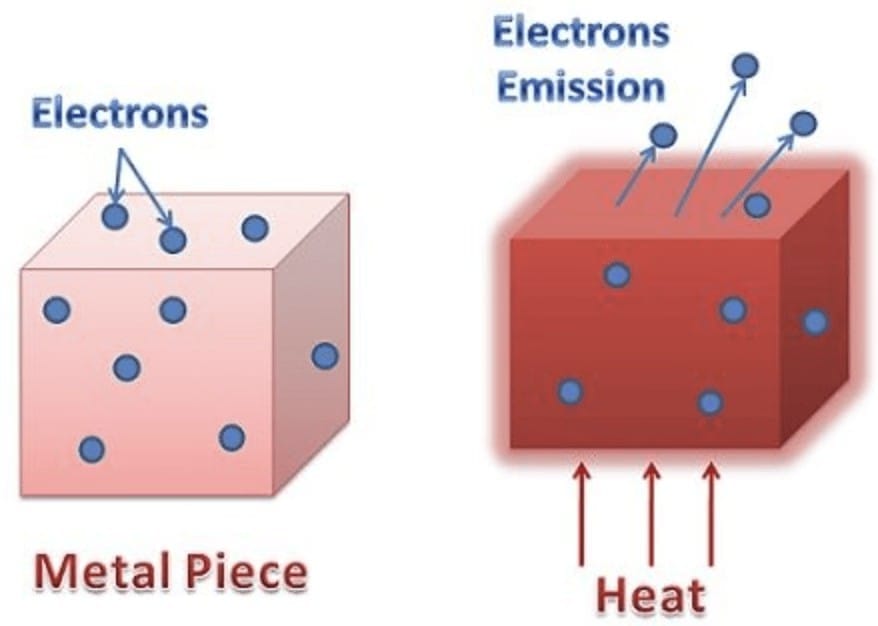
1.2.1 Thermionic Emission
- Electrons are emitted by heating the metal to high temperatures
- Thermal energy provides kinetic energy to overcome work function
- Used in cathode ray tubes and vacuum tubes
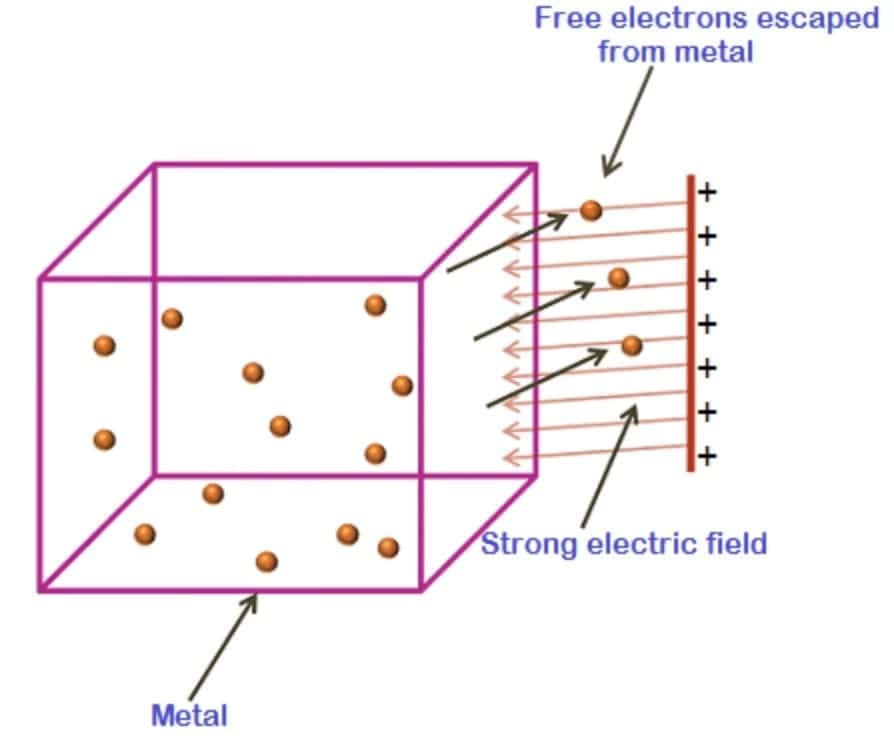
1.2.2 Field Emission
- Application of very strong electric field (≈ 10⁸ V/m) pulls electrons out
- Electric field modifies potential barrier at metal surface
- Used in field emission microscopes and spark plugs
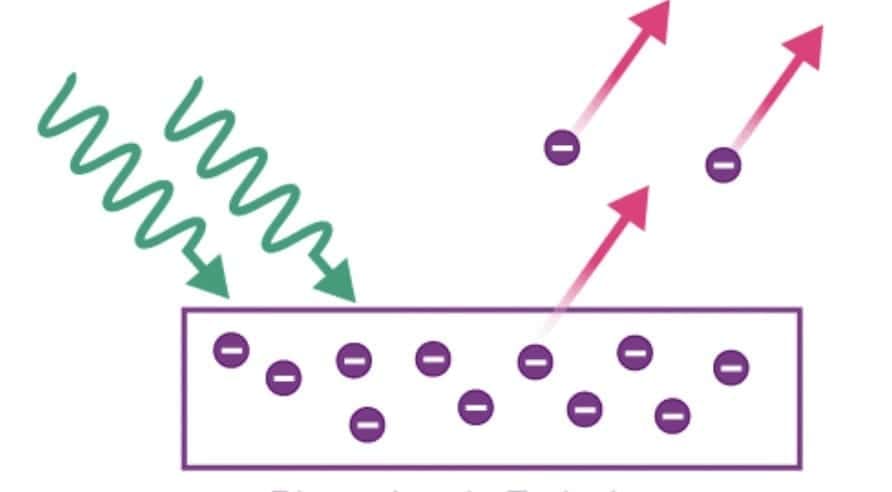
1.2.3 Photoelectric Emission
- Electrons are emitted when light of suitable frequency falls on metal surface
- Emitted electrons are called photoelectrons
- Forms the basis of photoelectric effect
- Most important method for understanding quantum nature of radiation
2. Photoelectric Effect
The phenomenon of emission of electrons from a metal surface when light of suitable frequency illuminates it is called photoelectric effect.
2.1 Historical Discovery
- Heinrich Hertz (1887): Discovered that UV light enhanced spark discharge in his electromagnetic wave experiments
- Wilhelm Hallwachs (1888): Observed that negatively charged zinc plate lost charge when illuminated by UV light; uncharged zinc plate became positively charged
- Philipp Lenard (1886-1902): Studied variation of photocurrent with potential, intensity, and frequency systematically
- Key observation: Light causes free electrons (discovered by J.J. Thomson in 1897) to escape from metal surface
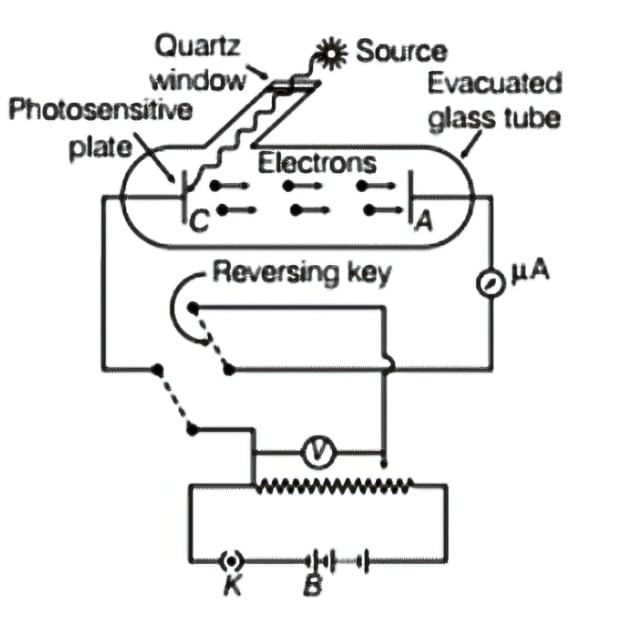
2.2 Experimental Setup
- Components: Evacuated glass/quartz tube, photosensitive emitter plate C, collector plate A, battery, voltmeter, microammeter
- Quartz window: Allows UV radiation to pass through (glass absorbs UV)
- Variable potential: Collector A can be maintained at positive or negative potential relative to emitter C using commutator
- Measurements: Photocurrent measured by microammeter, potential difference by voltmeter
- Control variables: Intensity of light (by changing distance), frequency (by using colored filters), collector potential
2.3 Experimental Observations
2.3.1 Effect of Light Intensity on Photocurrent
- Observation: Photocurrent increases linearly with intensity of incident light
- Conditions: Frequency of radiation and collector potential kept constant
- Interpretation: Number of photoelectrons emitted per second is directly proportional to intensity
- Graph: Straight line passing through origin when photocurrent plotted against intensity
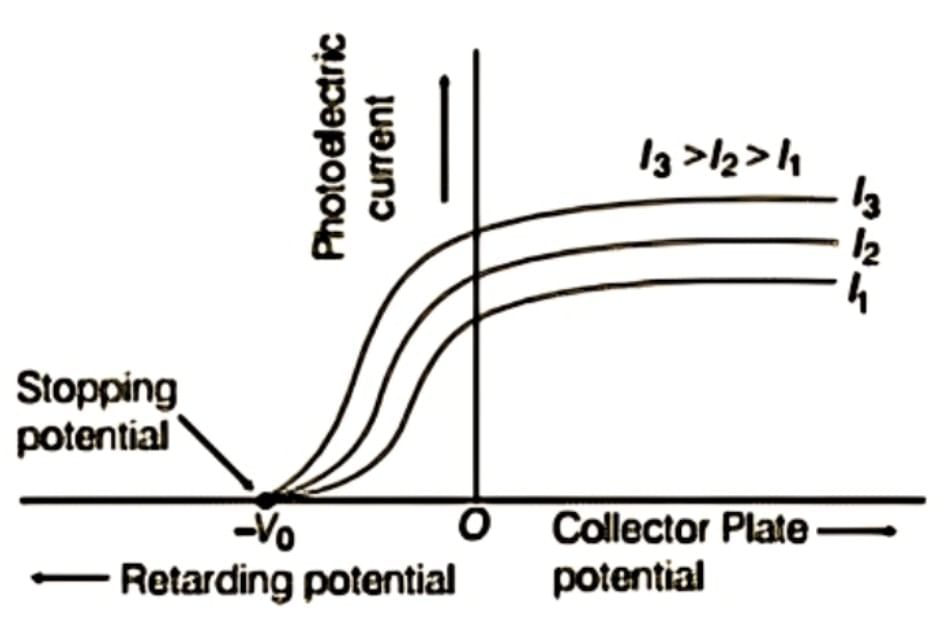
2.3.2 Effect of Collector Potential on Photocurrent
- Positive (accelerating) potential: Photocurrent increases with increasing positive potential on collector A
- Saturation current: Maximum photocurrent achieved when all emitted photoelectrons reach collector; further increase in potential does not increase current
- Negative (retarding) potential: Photocurrent decreases as collector becomes more negative
- Stopping potential (V₀): Minimum negative potential at which photocurrent becomes zero
- Critical finding: For a given frequency, stopping potential is independent of intensity but saturation current increases with intensity
2.3.3 Maximum Kinetic Energy and Stopping Potential
The relationship between stopping potential and maximum kinetic energy is:
- Formula: Kmax = eV₀
- Interpretation: Most energetic photoelectrons are just stopped by retarding potential V₀
- Energy unit: If V₀ is in volts and e = 1.6 × 10⁻¹⁹ C, then Kmax is in joules
- Key point: Not all photoelectrons have same kinetic energy; V₀ corresponds to maximum kinetic energy
2.3.4 Effect of Frequency on Stopping Potential
- Observation: Stopping potential increases linearly with frequency of incident radiation
- Threshold frequency (ν₀): Minimum frequency below which no photoelectric emission occurs regardless of intensity
- Graph: V₀ versus ν is a straight line with positive slope; intercepts frequency axis at ν₀
- Metal dependence: Different metals have different threshold frequencies; slope is same for all metals
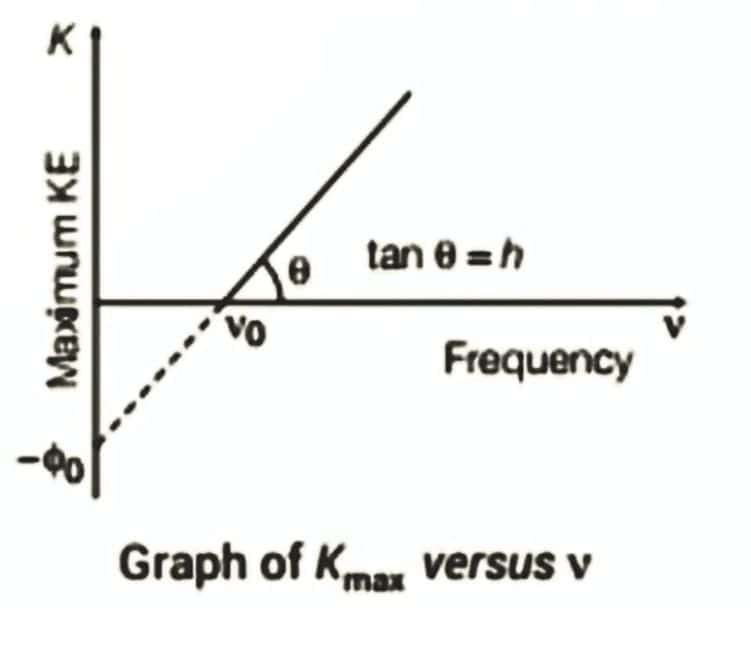
- Implication: Maximum kinetic energy depends on frequency, not intensity
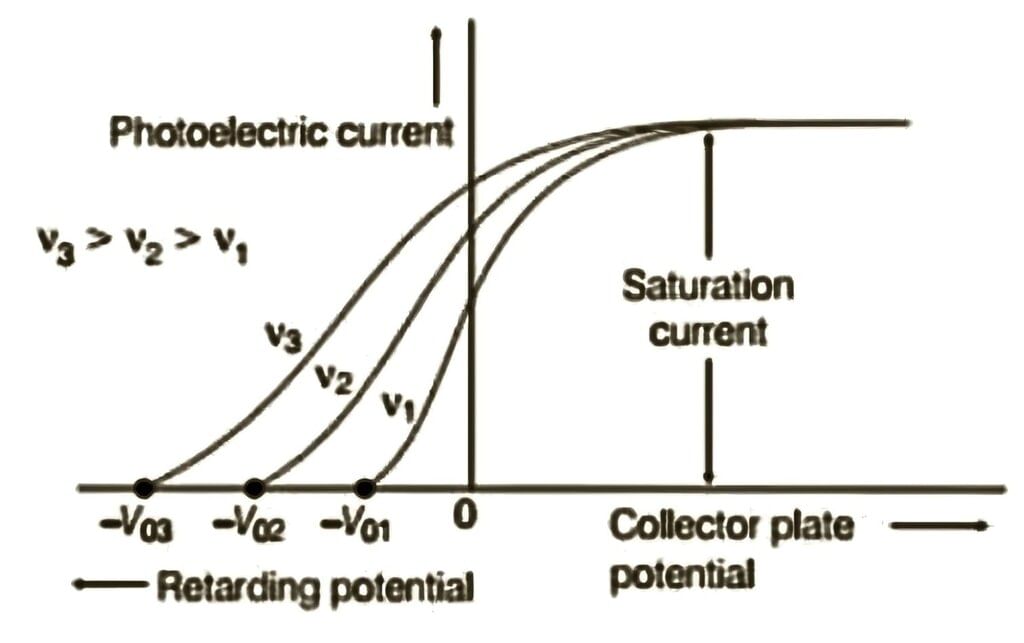 Variation of photoelectric current versus potential for different frequencies but constant intensity of incident radiation
Variation of photoelectric current versus potential for different frequencies but constant intensity of incident radiation
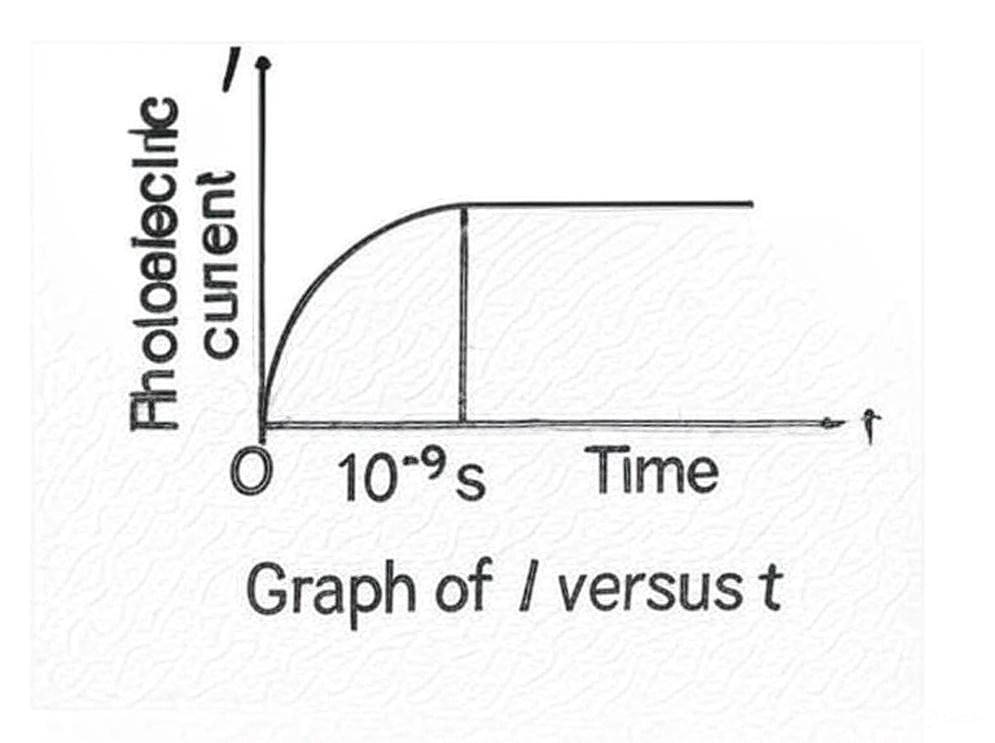
2.3.5 Time Lag
- Observation: Photoelectric emission is instantaneous (≈ 10⁻⁹ s or less)
- Significance: No measurable time lag between illumination and electron emission, even for very dim light
- Condition: Applies only when frequency exceeds threshold frequency
2.4 Summary of Key Experimental Facts
- Intensity effect: Photocurrent (number of photoelectrons) ∝ intensity; Kmax independent of intensity
- Saturation: Saturation current ∝ intensity; stopping potential independent of intensity
- Threshold frequency: No emission below ν₀ regardless of intensity; Kmax increases linearly with ν
- Instantaneous emission: No time lag between illumination and emission
Graphs Related to the Photoelectric Effect
The important experimentally observed graphs include:
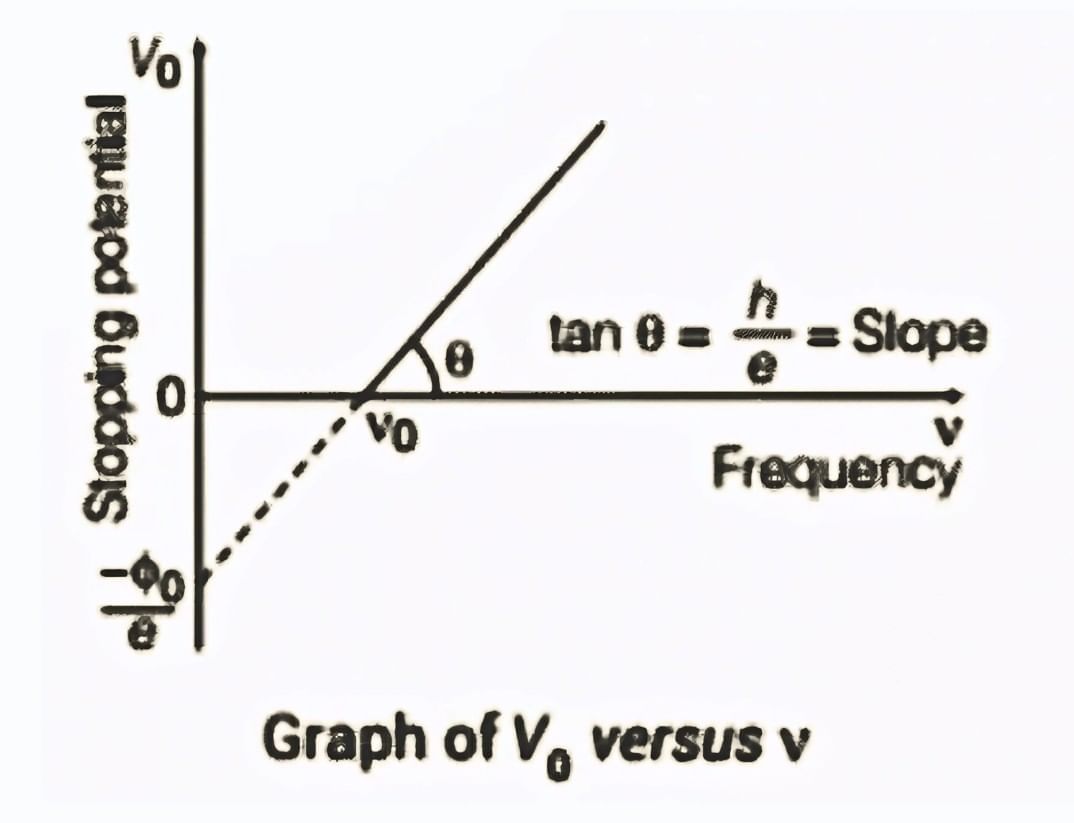
1. Stopping potential V0 vs Frequency ν:
We know that,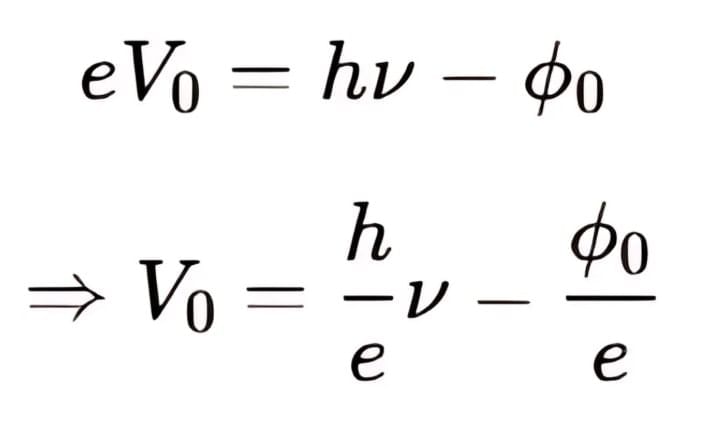 So,
So, 
It can be seen that, Vo versus ν curve is a straight line with slope = h/e and is independent of the nature of the material.
2. Maximum kinetic energy vs Frequency:
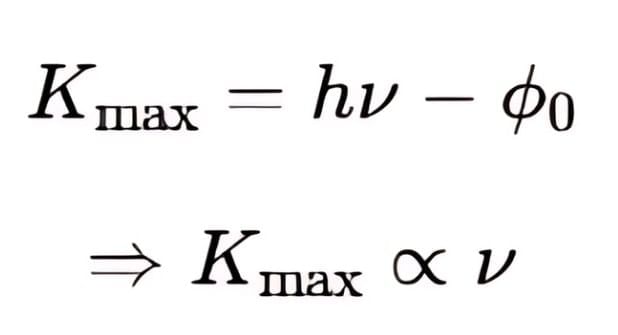
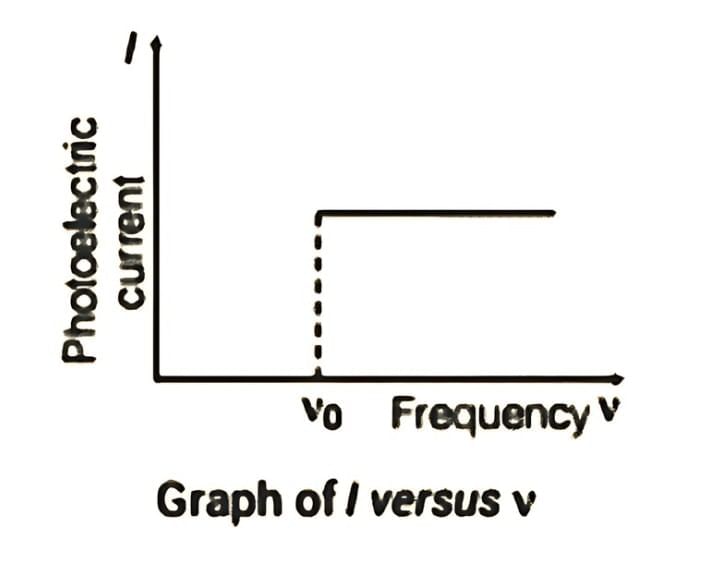
3. Photoelectric current I vs Frequency ν (for fixed intensity):
The saturation current is independent of frequency provided ν ≥ ν0; increasing frequency increases kinetic energy but not the number of photons per unit time for fixed intensity.
4. Intensity vs Stopping Potential V0:
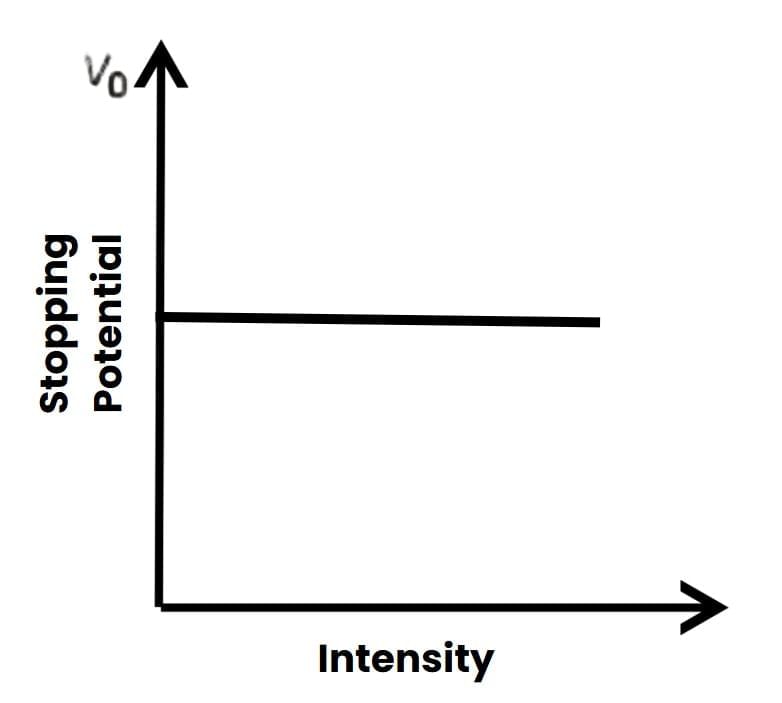 Graph of Vo versus Intensity
Graph of Vo versus Intensity5. Photoelectric Current I vs Time lag t:
The time lag between illumination and emission is negligibly small.
3. Wave Theory Fails to Explain Photoelectric Effect
Classical wave theory of light, successful in explaining interference, diffraction, and polarization, completely fails to explain photoelectric effect observations.
3.1 Predictions of Wave Theory
- Continuous energy absorption: Free electrons should absorb energy continuously from electromagnetic wave
- Intensity dependence: Greater intensity means larger amplitude of electric and magnetic fields, therefore greater energy absorbed per electron
- Kmax expectation: Maximum kinetic energy should increase with intensity
- No threshold: Sufficiently intense beam should eject electrons regardless of frequency
- Time lag: Electrons distributed over wavefront should take considerable time (hours) to accumulate enough energy to escape
3.2 Contradictions with Observations
- Kmax independence: Wave theory predicts Kmax ∝ intensity, but experimentally Kmax is independent of intensity
- Threshold frequency: Wave theory cannot explain why threshold frequency exists
- Instantaneous emission: Wave theory predicts time lag of hours, but emission is instantaneous
- Frequency dependence: Wave theory cannot explain linear dependence of Kmax on frequency
Trap Alert: Students often incorrectly think wave theory can explain some aspects of photoelectric effect. Remember: wave theory completely fails to explain all four key observations.
4. Einstein's Photoelectric Equation
In 1905, Albert Einstein proposed revolutionary quantum picture of light to explain photoelectric effect. He extended Planck's quantum hypothesis to electromagnetic radiation itself.
4.1 Photon Concept
- Quantum of radiation: Light consists of discrete packets of energy called photons or quanta
- Energy of photon: E = hν where h = Planck's constant = 6.63 × 10⁻³⁴ J·s, ν = frequency
- Absorption mechanism: Each electron absorbs one complete photon (all-or-nothing process)
- No partial absorption: Electron cannot absorb fraction of photon energy
4.2 Einstein's Photoelectric Equation
The fundamental equation relating photon energy to electron kinetic energy:
- Formula: Kmax = hν - φ₀
- Parameters: Kmax = maximum kinetic energy of photoelectron, hν = photon energy, φ₀ = work function
- Energy conservation: Photon energy is used to overcome work function; remaining energy appears as kinetic energy
- Alternative form: Kmax = h(ν - ν₀) where ν₀ = φ₀/h = threshold frequency
- Using stopping potential: eV₀ = hν - φ₀
4.3 Explanation of Experimental Observations
4.3.1 Independence of Kmax from Intensity
- Kmax = hν - φ₀ depends only on frequency ν, not on number of photons
- Intensity meaning: Number of photons per unit area per unit time
- Effect of intensity: More photons → more electrons emitted → higher photocurrent, but same Kmax
- Each electron-photon interaction is independent; one electron absorbs one photon
4.3.2 Existence of Threshold Frequency
- For emission, Kmax ≥ 0, therefore hν ≥ φ₀
- Threshold condition: ν ≥ ν₀ where ν₀ = φ₀/h
- If ν < ν₀,="" photon="" energy="" insufficient="" to="" overcome="" work="" function="" regardless="" of="">
- No cumulative effect: Photon energy cannot be accumulated by electron over time
4.3.3 Linear Variation with Frequency
- From eV₀ = hν - φ₀, we get V₀ = (h/e)ν - φ₀/e
- Slope: h/e (universal constant, same for all metals)
- Intercept: -φ₀/e on V₀ axis; varies with metal
- Graph of V₀ vs ν is straight line confirming Einstein's equation
4.3.4 Instantaneous Emission
- Single photon-electron interaction is an elementary quantum process
- No time required for energy accumulation; absorption is immediate
- Low intensity means fewer photons, hence fewer electrons, but emission still instantaneous
4.4 Millikan's Verification
- Period: 1906-1916; aimed to disprove Einstein's equation but ended up confirming it
- Method: Precise measurement of stopping potential for various frequencies and metals
- Result: Confirmed linear V₀ vs ν relationship with slope = h/e
- Planck's constant: Calculated h from slope; matched value from blackbody radiation
- Work function: Determined φ₀ for different metals from intercept
- Nobel Prize: Einstein (1921) for photoelectric effect; Millikan (1923) for elementary charge and photoelectric effect
Trap Alert: Students often confuse threshold frequency with cut-off frequency. Both terms mean the same: minimum frequency ν₀ = φ₀/h for photoelectric emission.
5. Photon: Particle Nature of Light
Photoelectric effect provided strong evidence that light behaves as particles (photons) in interaction with matter, complementing its wave nature.
5.1 Properties of Photons
- Energy: E = hν = hc/λ where c = speed of light = 3 × 10⁸ m/s, λ = wavelength
- Momentum: p = E/c = hν/c = h/λ
- Rest mass: Zero (photon cannot exist at rest; always travels at speed c)
- Charge: Electrically neutral; not deflected by electric or magnetic fields
- Speed: Always travels at c in vacuum, regardless of photon energy
- Stability: Photon is stable
- Spin: Photon has spin = 1
5.2 Photon-Matter Interaction
- Conservation laws: Total energy and momentum conserved in photon-particle collisions
- Photon number: Not conserved; photons can be created or absorbed
- All-or-nothing: Photon is either completely absorbed or not absorbed at all
- Compton effect (1924): Scattering of X-rays by electrons confirmed photon momentum
5.3 Intensity and Photon Picture
- Definition: Intensity = (number of photons/area/time) × (energy per photon)
- Same frequency: All photons have same energy hν regardless of intensity
- Increasing intensity: Means increasing number of photons, not energy per photon
- Formula: If N photons per second per unit area, then Intensity I = Nhν
5.4 Dual Nature of Light
- Wave properties: Interference, diffraction, polarization explained by wave theory
- Particle properties: Photoelectric effect, Compton effect explained by photon picture
- Complementarity: Both descriptions are valid; choice depends on experimental context
- Example: Eye lens focuses light (wave optics), retina absorbs light (photon picture)
6. Matter Waves: de Broglie Hypothesis
In 1924, French physicist Louis de Broglie proposed that if radiation has dual nature, material particles should also exhibit wave properties.
6.1 de Broglie Relation
The wavelength associated with a moving particle is given by:
- Formula: λ = h/p = h/(mv)
- Parameters: λ = de Broglie wavelength, h = Planck's constant, p = momentum, m = mass, v = velocity
- Wave-particle duality: Left side (λ) represents wave aspect, right side (p) represents particle aspect
- Universal constant: h connects wave and particle properties
6.2 Verification for Photon
- For photon: E = hν and E = pc (since rest mass = 0)
- Therefore: p = hν/c = h/λ
- This gives λ = h/p, confirming de Broglie relation holds for photons
- Photon wavelength equals electromagnetic wavelength
6.3 Matter Wave Characteristics
- Inverse mass dependence: λ ∝ 1/m; heavier particles have smaller wavelength
- Inverse velocity dependence: λ ∝ 1/v; faster particles have smaller wavelength
- Charge independence: de Broglie wavelength independent of charge or nature of particle
- Universal applicability: Applies to electrons, protons, neutrons, atoms, molecules
6.4 Macroscopic vs Microscopic Objects
6.4.1 Macroscopic Objects
- Example: Ball of mass 0.12 kg moving at 20 m/s has λ = 2.76 × 10⁻³⁴ m
- Atomic spacing: ≈ 10⁻¹⁰ m; de Broglie wavelength is 10²⁴ times smaller
- Conclusion: Wave properties completely negligible and unobservable
- Classical physics: Valid description for macroscopic objects
6.4.2 Microscopic Particles
- Example: Electron (m = 9.11 × 10⁻³¹ kg) at v = 5.4 × 10⁶ m/s has λ ≈ 0.135 nm
- Comparison: Comparable to atomic spacing (≈ 0.1 nm) and X-ray wavelengths
- Observability: Wave properties significant and measurable
- Experiments: Electron diffraction by crystals demonstrates wave nature
6.5 Physical Significance
- Matter waves: Also called de Broglie waves; represent probability amplitude
- Wavelength: Has physical meaning; determines diffraction and interference pattern
- Phase velocity: vₚ = ω/k has no direct physical significance for matter waves
- Group velocity: Equals particle velocity; physically meaningful
- Nobel Prize: de Broglie awarded Nobel Prize in Physics (1929) for discovery of wave nature of electrons
Trap Alert: Students often think de Broglie wavelength depends on particle charge. Remember: λ = h/(mv) depends only on mass and velocity, not charge.
7. Heisenberg Uncertainty Principle
Heisenberg's Uncertainty Principle states that it is impossible to simultaneously determine the exact position and exact momentum of a particle.
If
- Δx = uncertainty in position
- Δp = uncertainty in momentum
then,
Δx Δp ≥ h/4πh
or 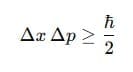
where
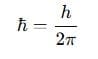
7.1 Physical Meaning
- If position is measured very accurately (Δx small), momentum becomes highly uncertain (Δp large).
- If momentum is measured very accurately (Δp small), position becomes highly uncertain (Δx large).
Thus, both cannot be known exactly at the same time.
7.2 Significance
- Explains why electron cannot have definite orbit in atom.
- Supports wave nature of matter.
- Forms the basis of quantum mechanics.
8. Important Formulae and Constants
8.1 Key Constants
- Planck's constant: h = 6.63 × 10⁻³⁴ J·s
- Speed of light: c = 3 × 10⁸ m/s
- Electron charge: e = 1.6 × 10⁻¹⁹ C
- Electron mass: mₑ = 9.11 × 10⁻³¹ kg
- Energy conversion: 1 eV = 1.6 × 10⁻¹⁹ J
8.2 Photoelectric Effect Formulae
- Einstein's equation: Kmax = hν - φ₀
- Stopping potential: eV₀ = Kmax
- Combined form: eV₀ = hν - φ₀ or V₀ = (h/e)ν - φ₀/e
- Threshold frequency: ν₀ = φ₀/h
- Threshold wavelength: λ₀ = c/ν₀ = hc/φ₀
8.3 Photon Properties
- Energy: E = hν = hc/λ
- Momentum: p = h/λ = hν/c = E/c
- Energy-wavelength: E (in eV) = 1240/λ (in nm) [frequently used approximation]
8.4 Matter Wave Formula
- de Broglie wavelength: λ = h/p = h/(mv)
- For charged particle accelerated through potential V: K.E. = eV = (1/2)mv² = p²/(2m)
- Therefore: λ = h/√(2meV) for electron accelerated through potential V
9. Problem-Solving Strategy
9.1 Photoelectric Effect Problems
- Step 1: Identify given quantities (frequency or wavelength, work function, stopping potential)
- Step 2: Convert wavelength to frequency using c = νλ if needed
- Step 3: Convert work function to same energy units as required (J or eV)
- Step 4: Apply Einstein's equation: Kmax = hν - φ₀ or eV₀ = hν - φ₀
- Step 5: Check if ν > ν₀; if not, no emission occurs
9.2 Matter Wave Problems
- Step 1: Calculate momentum p = mv (if velocity given) or p = √(2mK) if kinetic energy K given
- Step 2: For accelerated charged particle: p = √(2mqV) where q = charge, V = potential
- Step 3: Apply λ = h/p
- Step 4: Check units; momentum in kg·m/s gives wavelength in meters
9.3 Common Numerical Values
- UV light: λ ≈ 200-400 nm, ν ≈ 10¹⁵ Hz
- Visible light: λ ≈ 400-700 nm, ν ≈ 10¹⁴ Hz
- Work functions: Alkali metals (Li, Na, K, Cs) have φ₀ ≈ 2-3 eV; other metals ≈ 4-5 eV
- Electron wavelength: For typical velocities ≈ 10⁶ m/s, λ ≈ 0.1 nm (comparable to atomic spacing)
10. Common Student Mistakes
10.1 Conceptual Errors
- Mistake: Thinking higher intensity light gives electrons more kinetic energy
Correct: Higher intensity gives more electrons, not more energy per electron - Mistake: Believing dim light of high frequency cannot cause emission
Correct: Even single photon of sufficient frequency can eject electron - Mistake: Assuming photocurrent is proportional to frequency
Correct: Photocurrent is proportional to intensity, not frequency - Mistake: Thinking stopping potential depends on intensity
Correct: V₀ depends only on frequency, independent of intensity
10.2 Calculation Errors
- Mistake: Using λ directly in E = hν formula
Correct: First convert to frequency using ν = c/λ, or use E = hc/λ - Mistake: Mixing units (eV and J) without conversion
Correct: Always convert to consistent units; use 1 eV = 1.6 × 10⁻¹⁹ J - Mistake: Forgetting to check threshold condition
Correct: First verify ν ≥ ν₀ before calculating Kmax - Mistake: Using rest mass of photon in calculations
Correct: Photon has zero rest mass; use E = pc, not E = mc²
10.3 Matter Wave Errors
- Mistake: Thinking de Broglie wavelength depends on particle charge
Correct: λ = h/(mv) is independent of charge - Mistake: Applying λ = h/p to photon and getting wrong answer
Correct: For photon, p = E/c = hν/c, so λ = c/ν (electromagnetic wavelength) - Mistake: Expecting observable wave properties for macroscopic objects
Correct: λ becomes extremely small (≈ 10⁻³⁴ m) for everyday objects
Understanding the dual nature of radiation and matter is fundamental to modern physics. The particle nature of light explains photoelectric effect through photon absorption. The wave nature of matter predicts observable diffraction for microscopic particles like electrons. Both aspects are essential for complete description of physical phenomena at atomic and subatomic scales. Mastery of Einstein's photoelectric equation and de Broglie relation, along with their quantitative applications, is crucial for success in competitive physics examinations.
Some Solved Examples
Example 1. The photoelectric threshold of a certain metal is 2750 Å. Find
(i) The work function for emission of an electron from this metal,
(ii) Maximum kinetic energy of these electrons,
(iii) The maximum velocity of the electrons ejected from the metal by light with a wavelength of 1800 Å.
Sol.
(i) The threshold wavelength is λth = 2750 Å.
Work function φ = h c / λth.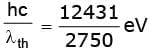
(ii) Energy of incident photon of wavelength 1800 Å (in eV):
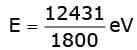
Energy = 6.9 eV.
Maximum kinetic energy Kmax = E - φ = 6.9 eV - 4.52 eV = 2.38 eV.
(iii) If maximum speed is vmax, then (1/2) m vmax² = 2.38 eV.
Solving gives

vmax = 9.15 × 105 m·s-1.
Example 2. Light quanta with energy 4.9 eV eject photoelectrons from a metal with work function 4.5 eV. Find the maximum impulse transmitted to the surface of the metal when each electron flies out.
Sol. By Einstein's equation, the kinetic energy of each ejected electron is K = hν - φ = 4.9 eV - 4.5 eV = 0.4 eV.
Momentum p of the emitted electron (non-relativistic) is p = √(2 m E), where E is kinetic energy in joules.
Substituting values,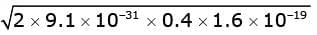
Maximum impulse = 3.45 × 10-25 kg·m·s-1.
Example 3. A tungsten cathode with threshold 2300 Å is irradiated by ultraviolet light of wavelength 1800 Å. Calculate
(i) Maximum energy of emitted photoelectron and
(ii) Work function for tungsten (express results in electron-volts).
Given: h = 6.6 × 10-34 J·s, c = 3.0 × 108 m·s-1, 1 eV = 1.6 × 10-19 J.
Sol. Work function φ = h c / λth (in eV).
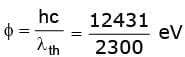
φ = 5.4 eV.
Energy of incident photons (λ = 1800 Å) is
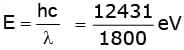
E = 6.9 eV.
Maximum kinetic energy Kmax = E - φ = 6.9 eV - 5.4 eV = 1.5 eV.
Example 4. Light of wavelength 1800 Å ejects photoelectrons from a metal plate whose work function is 2 eV. If a uniform magnetic field of 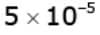 tesla is applied parallel to the plate, what is the radius of the path followed by electrons ejected normally from the plate with maximum energy?
tesla is applied parallel to the plate, what is the radius of the path followed by electrons ejected normally from the plate with maximum energy?
Sol. Energy of incident photons in eV is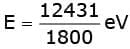
E = 6.9 eV.
Maximum kinetic energy Kmax = E - φ = 6.9 eV - 2.0 eV = 4.9 eV.
Convert to joules: K = 4.9 × 1.6 × 10-19 J.
So (1/2) m v² = 4.9 × 1.6 × 10-19 J.
Solving gives
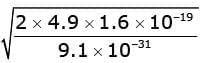
v = 1.31 × 106 m·s-1.
When an electron with speed v enters a magnetic field B normally, it moves in a circle of radius r given by r = m v / (e B).
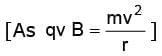
Therefore,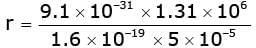
r = 0.149 m.
FAQs on Dual Nature Of Radiation And Matter
| 1. What is electron emission and why is it important in physics? |  |
| 2. What are the main types of electron emission? |  |
| 3. What is the photoelectric effect and how does it demonstrate the particle nature of light? |  |
| 4. What are the laws of photoelectric emission? |  |
| 5. What is Einstein’s photoelectric equation and what does it signify about the dual nature of radiation? |  |







