Balancing Redox Equations
Balancing of Redox Chemical Equations
Every chemical equation must obey the law of conservation of mass. In a balanced chemical equation, the number of atoms of each element and the total charge (for ionic equations) must be the same on both sides. For oxidation-reduction (redox) reactions, electrons are transferred between species; to balance such equations we must ensure both mass and charge (or electron transfer) are balanced.
Common methods to balance redox reactions
- Oxidation number method
- Half-reaction (ion-electron) method
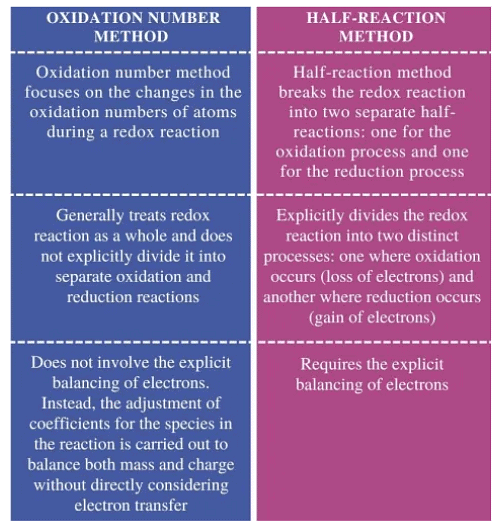 Difference between Oxidation Number Method & Half Reaction Method
Difference between Oxidation Number Method & Half Reaction MethodOxidation Number Method
In the oxidation number method you determine the change in oxidation state of the atoms that undergo redox change and then adjust stoichiometric coefficients so that total increase in oxidation numbers equals total decrease. This method is especially convenient for straightforward reactions where oxidation number changes are easy to track.
Rules for assigning oxidation numbers (brief)
- The oxidation number of an atom in its elemental form is 0.
- For monoatomic ions, the oxidation number equals the ionic charge.
- Oxygen is usually -2 in compounds (except in peroxides where it is -1, and in OF2 where it is +2).
- Hydrogen is usually +1 when bonded to non-metals and -1 when bonded to metals.
- The sum of oxidation numbers in a neutral compound is 0; in a polyatomic ion it equals the ion charge.
Steps for balancing redox equations by the oxidation-number method
- Write the unbalanced (skeletal) equation including all known reactants and products.
- Assign oxidation numbers to all atoms in the equation.
- Identify which elements undergo change in oxidation number and determine whether each is oxidised (increase in oxidation number) or reduced (decrease in oxidation number).
- Calculate the total increase and total decrease in oxidation numbers. For an element that changes on multiple atoms, multiply the change per atom by the number of such atoms.
- Adjust coefficients of species containing the oxidised and reduced elements so that the total increase equals the total decrease in oxidation numbers (i.e., electrons lost = electrons gained).
- Balance the remaining atoms except hydrogen and oxygen.
- Balance hydrogen and oxygen according to the medium:
Acidic solution: add H⁺ to balance hydrogen and H₂O to balance oxygen.
Basic solution: add H₂O to the side deficient in oxygen and add the same number of OH⁻ to the opposite side to balance hydrogen (or first balance as if acidic, then neutralise H⁺ by adding OH⁻ to both sides). - Check and simplify coefficients if possible by dividing by the greatest common divisor; cancel any identical species appearing on both sides.

Worked example (oxidation-number method)
Zn + HCl → ZnCl2 + H2
Solution:
Write the unbalanced skeleton equation: Zn + HCl → ZnCl2 + H2
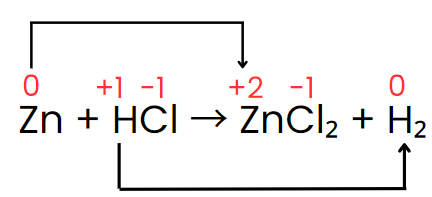
Assign oxidation numbers: Zn(0) → Zn(+2); H(+1) → H(0); Cl(-1) remains unchanged.
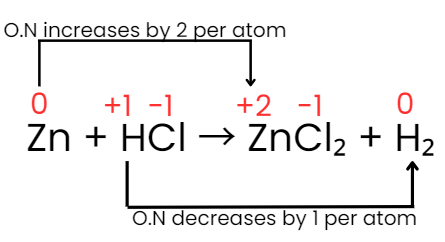
Identify changes: Zn increases from 0 to +2 (loss of 2 electrons), H decreases from +1 to 0 (gain of 1 electron per H).
Total change per Zn atom is +2; each H atom changes by -1. To equalise electron transfer one Zn requires two H atoms to accept 2 electrons in total.
Adjust coefficients accordingly: Zn + 2HCl → ZnCl2 + H2
Verify atoms and charge: Zn:1→1; Cl:2→2; H:2→2. Equation is balanced.
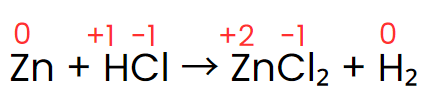
Half-Reaction (Ion-Electron) Method
The half-reaction method balances oxidation and reduction half-reactions separately by balancing atoms and charge (by adding H₂O, H⁺ or OH⁻ and electrons), then combining the half-reactions so electrons cancel. This method is particularly systematic for reactions in acidic or basic solution. The method (often referred to as the ion-electron method) was developed by Jette and Læmer in 1927.
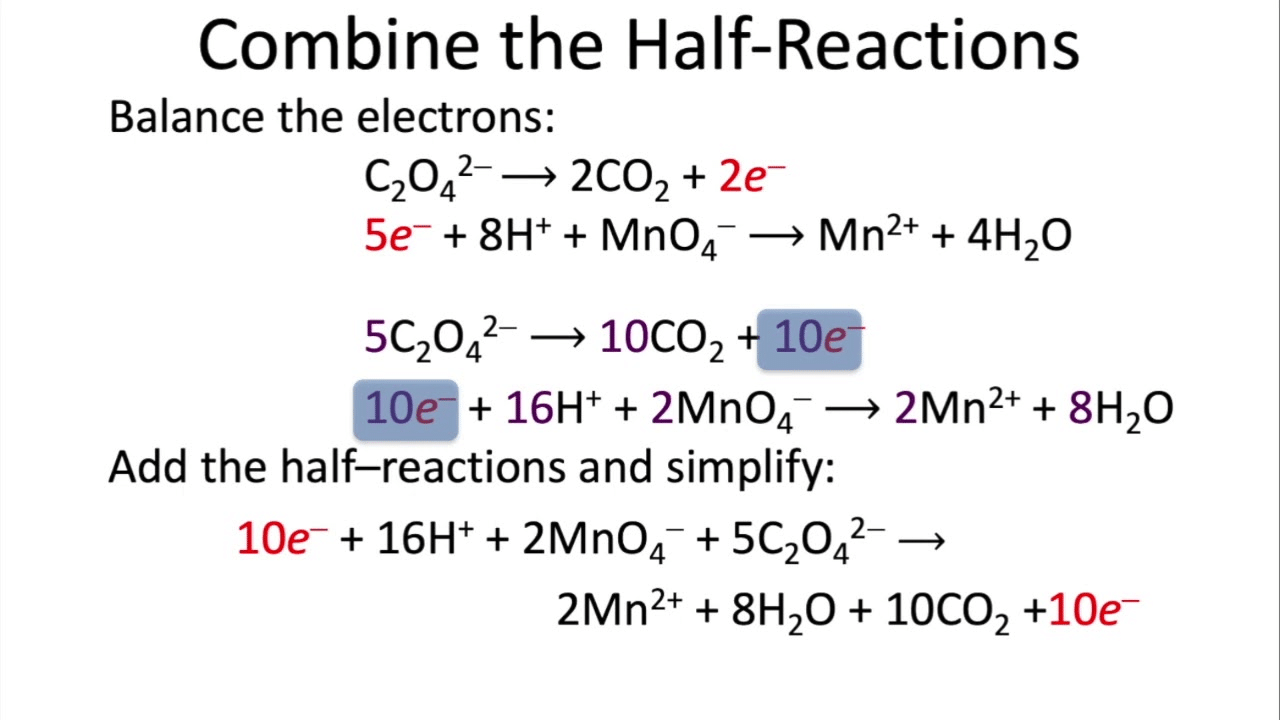
Steps for balancing by the half-reaction method
- Identify the species oxidised and reduced and write separate half-reactions for oxidation and reduction.
- Balance all atoms in each half-reaction except hydrogen and oxygen.
- Balance oxygen by adding H₂O to the side lacking oxygen.
- Balance hydrogen by adding H⁺ (in acidic solution) or by adding H₂O and OH⁻ pairs (in basic solution).
- Balance charge by adding electrons (e⁻) to the more positive side (or the side lacking electrons).
- Multiply the half-reactions by suitable integers so that the number of electrons in both half-reactions is equal.
- Add the half-reactions and cancel electrons and any species common to both sides.
- If the reaction is in basic medium, convert any H⁺ to H₂O + OH⁻ pairs by adding OH⁻ to both sides as required and simplify.
- Convert the net ionic equation back to molecular form by reintroducing spectator ions if a molecular equation is required.
Note: In acidic media include H⁺; in basic media include OH⁻; in neutral media neither H⁺ nor OH⁻ is explicitly used.
Worked example (half-reaction method)
Cu + HNO3 → Cu(NO3)2 + NO + H2O
Solution:
Write the ionic representation of species that change: Cu + H⁺ + NO3- → Cu2+ + NO + H₂O

Separate into half-reactions: oxidation half: Cu → Cu2+; reduction half: NO3- → NO
Balance atoms other than H and O: oxidation half is already balanced for Cu. Reduction half is balanced for N.
Balance oxygen by adding H₂O to the side deficient in oxygen; balance hydrogen by adding H⁺ (since medium is acidic): add H₂O and H⁺ as needed for the NO₃⁻ → NO half.
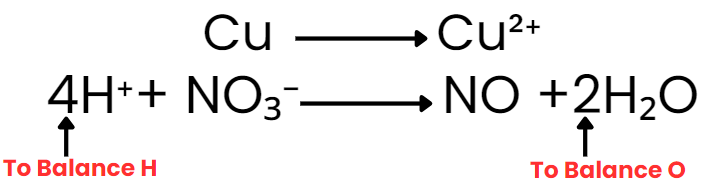
Add electrons to the side deficient in electrons to balance charge: put electrons on the more positive side.
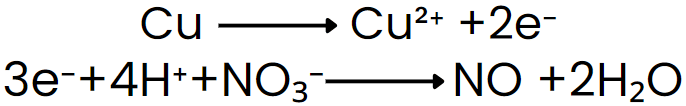
Multiply half-reactions to equalise the number of electrons transferred, then add the half-reactions so electrons cancel.
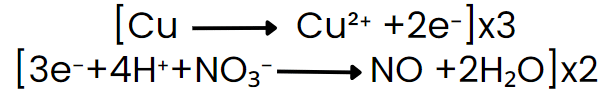
Cancel common species on both sides and combine remaining ions to return to molecular form (if required), adding spectator ions appropriately.
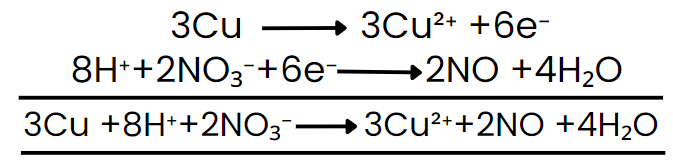
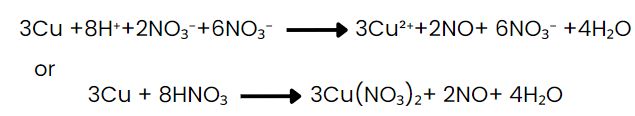
Final balanced molecular equation results after combining and simplifying; check that atoms and charges balance.
Solved Examples
Example Balance the reaction
VO2+ + MnO4- → V(OH)4+ + Mn2+
Solution:
Write the two half-reactions: VO2+ → V(OH)4+ and MnO4- → Mn2+.
Balance atoms except H and O by inspection where possible.
Balance oxygen by adding H₂O and balance hydrogen by adding H⁺ (reaction occurs in acidic medium here): add 3H₂O to the left of the first half to provide O for V(OH)₄⁺ and add H⁺ to balance H.
Add electrons to balance charge in each half-reaction: the VO half requires 1 e⁻ on the product side after balancing; the MnO₄⁻ half requires 5 e⁻ on the reactant side when balanced with H⁺ and H₂O.
Multiply the VO half by 5 so that electrons equal 5 in both half-reactions.
Add the two half-reactions and cancel electrons and any species common to both sides.
Simplify to obtain the balanced ionic equation: 11H₂O + 5VO2+ + MnO₄- → 5V(OH)4+ + Mn2+ + 2H⁺.
Example Balance the redox reaction of pertechnetate with stannous ion in acidic solution:
TcO4- + Sn2+ → Tc4+ + Sn4+
Solution:
Write half-reactions: TcO₄⁻ → Tc⁴⁺ and Sn²⁺ → Sn⁴⁺.
Balance non-H/O atoms (already balanced for Tc and Sn).
Balance oxygen in TcO₄⁻ → Tc⁴⁺ by adding 4 H₂O to the product side; balance hydrogen by adding 8 H⁺ to the reactant side.
Add electrons to balance charge in the Tc half: add 3 e⁻ to the reactant side to reduce TcO₄⁻ to Tc⁴⁺.
Balance the Sn half by adding 2 e⁻ to the product side: Sn²⁺ → Sn⁴⁺ + 2 e⁻.
Multiply the Sn half by 3 and the Tc half by 2 so that total electrons equal 6.
Add the half-reactions and cancel electrons to obtain: 16 H⁺ + 2 TcO₄⁻ + 3 Sn²⁺ → 2 Tc⁴⁺ + 3 Sn⁴⁺ + 8 H₂O.
Example Potassium dichromate reacts with sodium sulphite in acidic medium to form sulphate and Cr(III):
K2Cr2O7 + Na2SO3 (acidic) → SO42- + Cr3+ + ...
Solution:
Write the ionic skeletal equation for the reacting species in acidic medium.

Assign oxidation numbers to determine which species are oxidised and which are reduced: Cr changes from +6 in Cr₂O₇²⁻ to +3 in Cr³⁺ (reduction); S in SO₃²⁻ changes from +4 to +6 in SO₄²⁻ (oxidation).

Calculate the change in oxidation number per atom and per formula unit to determine electron transfer magnitudes.

Balance charge by adding H⁺ (acidic medium). Add H⁺ to the appropriate side so ionic charges balance.
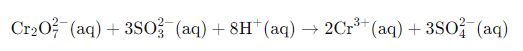
Finally add H₂O molecules to balance oxygen and hydrogen and simplify the equation. Add four H₂O on the right (as required here) to complete balancing.
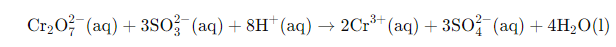
Example Permanganate produces iodine (I2) and manganese dioxide (MnO2) in basic medium:
MnO4- + I⁻ → I2 + MnO2
Solution:
Write the skeletal equation in basic medium.

Find oxidation and reduction half-reactions:
Oxidation half: I⁻ → I₂

Reduction half: MnO₄⁻ → MnO₂

Balance iodine atoms in the oxidation half by placing 2 I⁻ → I₂.

Balance oxygen in the reduction half by adding H₂O where needed and then balance hydrogen by adding H⁺ (initially treat as acidic to balance H and O).
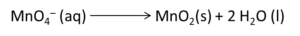
Convert H⁺ to OH⁻ because the medium is basic: add an equal number of OH⁻ to both sides to neutralise H⁺; combine H⁺ + OH⁻ → H₂O and simplify.

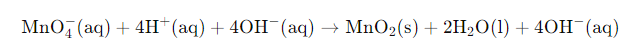
After conversion the half-reactions become balanced for atoms and charge; add electrons where required to balance charge in each half.

Multiply the half-reactions to equalise electron transfer; for example multiply oxidation half by 3 and reduction half by 2 so electrons cancel.


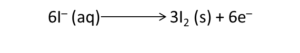

Add the half-reactions and cancel electrons and other species common to both sides to obtain the net balanced equation in basic medium.
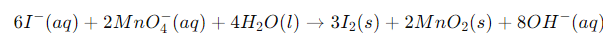
Verify final equation by checking atom counts and net charges on both sides.
Summary
The oxidation-number method is fast when oxidation state changes are obvious. The half-reaction method is systematic and convenient for reactions in acidic or basic media; it explicitly accounts for electrons transferred. Mastery of both methods and careful checking of atoms and charges will ensure correct balancing of any redox equation.
FAQs on Balancing Redox Equations
| 1. What is the oxidation number method for balancing redox equations? |  |
| 2. How do you use the half-reaction method to balance redox equations? |  |
| 3. What are some common mistakes to avoid when balancing redox equations? |  |
| 4. Can you provide an example of a redox reaction and how to balance it using both methods? |  |
| 5. How are redox reactions relevant for NEET and other competitive exams? |  |






