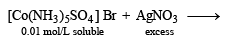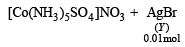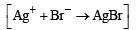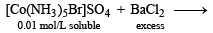JEE Advanced (Single Correct MCQs): Some Basic Concepts of Chemistry Chapter
MCQ Practice Test & Solutions: JEE Advanced (Single Correct MCQs): Some Basic Concepts of Chemistry (28 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Advanced (Single Correct MCQs): Some Basic Concepts of Chemistry". These 28 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 50 minutes
- - Number of Questions: 28
Sign up on EduRev for free to attempt this test and track your preparation progress.
27 g of Al will react completely with how many grams of oxygen?(1978)
Detailed Solution: Question 1
A compound was found to contain nitrogen and oxygen in the ratio 28 gm and 80 gm respectively. The formula of compound is (1978)
Detailed Solution: Question 2
The largest number of molecules is in (1979)
Detailed Solution: Question 3
The total number of electrons in one molecule of carbon dioxide is
Detailed Solution: Question 4
A gaseous mixture contains oxygen and nitrogen in the ratio of 1 : 4 by weight. Therefore the ratio of their number of molecules is (1979)
Detailed Solution: Question 5
2.76 g of silver carbonate on being strongly heated yields a residue weighing (1979)
Detailed Solution: Question 6
M is molecular weight of KMnO4. The equivalent weight of KMnO4 when it is converted into K2MnO4 is (1980)
Detailed Solution: Question 7
If 0.50 mole of BaCl2 is mixed with 0.20 mol of Na3PO4, the maximum number of moles of Ba3(PO4)2 that can be formed is (1981 - 1 Mark)
Detailed Solution: Question 8
One mole of N2H4 loses ten moles of electrons to form a new compound Y. Assuming that all the nitrogen appears in the new compound, what is the oxidation state of nitrogen in Y? (There is no change in the oxidation state of hydrogen). (1981 - 1 Mark)
Detailed Solution: Question 9
The oxidation number of carbon in CH2O is (1982 - 1 Mark)
Detailed Solution: Question 10
A molal solution is one that contains one mole of a solute in: (1986 - 1 Mark)
Detailed Solution: Question 11
The brown ring complex compound is for mulated as [Fe(H2O)5(NO)]SO4. The oxidation state of iron is :
Detailed Solution: Question 12
The equivalent weight of MnSO4 is half of its molecular weight when it is converted to : (1988 - 1 Mark)
Detailed Solution: Question 13
In which mode of expression, the concentration of a solution remains independent of temperature? (1988 - 1 Mark)
Detailed Solution: Question 14
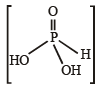
The oxidation number of phosphorus in Ba(H2PO2)2 is : (1990 - 1 Mark)
Detailed Solution: Question 15
The oxidation states of the most electronegative element in the products of the reaction, BaO2 with dil. H2SO4 is (1991 - 1 Mark)
For the redox reaction :
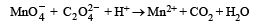
the correct coefficients of the reactants for the balanced reaction are (1992 - 1 Mark)
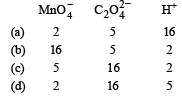
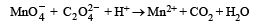
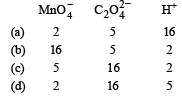
Detailed Solution: Question 17
The normality of 0.3 M phosphorous acid (H3PO3) is, (1999 - 2 Marks)
Detailed Solution: Question 18
The oxidation number of sulph ur in S8 , S2F2, H2 S respectively, are (1999 - 2 Marks)
Detailed Solution: Question 19
Amongst the following identify the species with an atom in +6 oxidation state (2000S)
Detailed Solution: Question 20
The reaction, 3ClO-(aq) → ClO 3- (aq) + 2Cl-(aq), is an example of (2001S)
Detailed Solution: Question 21
An aqueous solution of 6.3 g oxalic acid dihydrate is made up to 250 ml. The volume of 0.1 N NaOH required to completely neutralize 10 ml of this solution is (2001S)
Detailed Solution: Question 22
In the standardization of Na2S2O3 using K2Cr 2O7 by iodometry, the equivalent weight of K2Cr2O7 is (2001S)
Detailed Solution: Question 23
How many moles of electron weigh one kilogram? (2002S)
Detailed Solution: Question 24
Which has maximum number of atoms? (2003S)
Detailed Solution: Question 25
Mixture X = 0.02 mol of [Co(NH3)5SO4]Br and 0.02 mol of [Co(NH3)5Br]SO4 was prepared in 2 litre of solution. (2003S)
1 litre of mixture X + excess AgNO3 —→ Y.
1 litre of mixture X + excess BaCl2 —→ Z
No. of moles of Y and Z are
1 litre of mixture X + excess AgNO3 —→ Y.
1 litre of mixture X + excess BaCl2 —→ Z
No. of moles of Y and Z are
Detailed Solution: Question 26
The pair of the compounds in which both the metals are in the highest possible oxidation state is (2004S)
Detailed Solution: Question 27
Consider a titration of potassium dichromate solution with acidified Mohr's salt solution using diphenylamine as indicator. The number of moles of Mohr's salt required per mole of dichromate is (2007)
Detailed Solution: Question 28
446 docs|929 tests |


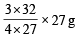
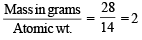
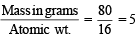

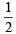 × 6.02 × 1023 molecules of N2O5
× 6.02 × 1023 molecules of N2O5 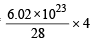 molecules of nitrogen
molecules of nitrogen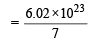 molecules of nitrogen
molecules of nitrogen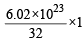
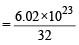 moleculesof oxygen
moleculesof oxygen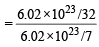 = 7 : 32
= 7 : 32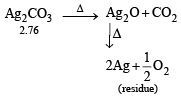
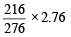 = 2.16g of Ag
= 2.16g of Ag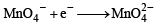
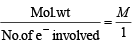 = M [∵ Mol. wt. = M]
= M [∵ Mol. wt. = M]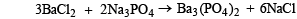
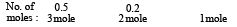
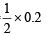 = 0.1 mol.
= 0.1 mol.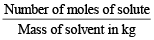
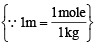
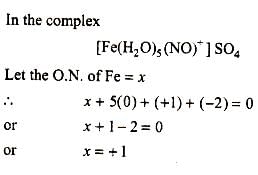
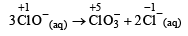
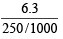 = 25.2 g/L
= 25.2 g/L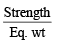
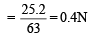
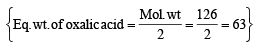
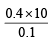 = 40 ml.
= 40 ml.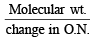
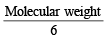
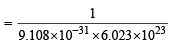
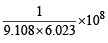
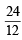 × 6.023 × 1023 = 2 × 6.023 × 1023 atom
× 6.023 × 1023 = 2 × 6.023 × 1023 atom × 6.023 × 1023 = 6.023 × 1023 atom = 1 mole atoms
× 6.023 × 1023 = 6.023 × 1023 atom = 1 mole atoms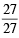 × 6.023 × 1023 = 6.023 × 1023 atom = 1 mole atoms
× 6.023 × 1023 = 6.023 × 1023 atom = 1 mole atoms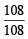 × 6.023 × 1023 = 6.023 × 1023 atom = 1 mole atoms
× 6.023 × 1023 = 6.023 × 1023 atom = 1 mole atoms