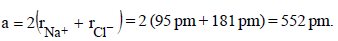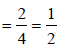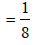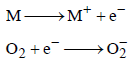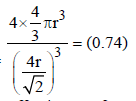MCQs (One or More Correct Option): The Solid State & Surface Chemistry
MCQ Practice Test & Solutions: Test: MCQs (One or More Correct Option): The Solid State & Surface Chemistry | JEE Advanced (8 Questions)
You can prepare effectively for JEE Crack JEE with 35 Years of Previous Year Solved Papers with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: MCQs (One or More Correct Option): The Solid State & Surface Chemistry | JEE Advanced". These 8 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 10 minutes
- - Number of Questions: 8
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following statement(s) is (are) correct?(1998 - 2 Marks)
Detailed Solution: Question 1
The correct statement(s) regarding defects in solids is (are) (2009S)
Detailed Solution: Question 2
The correct statement(s) pertaining to the adsorption of agas on a solid surface is (are) (2011)
Detailed Solution: Question 3
Choose the correct reason(s) for the stability of the lyophobiccolloidal particles. (2012)
The given graphs/data I, II, III and IV represent general trends observed for different physisorption and chemisorption processes under mild conditions of temperature and presure.
Which of the following choice(s) about I, II, III and IV is (are) correct ? (2012)
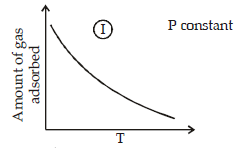
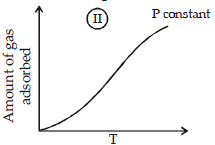
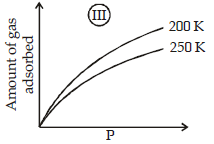

Which of the following choice(s) about I, II, III and IV is (are) correct ? (2012)




Detailed Solution: Question 5
If the unit cell of a mineral has cubic close packed (ccp) array of oxygen atoms with m fraction of octahedral holes occupied by aluminium ions and n fraction of tetrahedral holes occupied by magnesium ions, m and n, respectively, are (JEE Adv. 2015)
Detailed Solution: Question 6
When O2 is adsorbed on a metallic surface, electron transferoccurs from the metal to O2. The true statement(s) regardingthis adsorption is(are) (JEE Adv. 2015)
Detailed Solution: Question 7
The CORRECT statement(s) for cubic close packed (ccp)three dimensional structure is (are) (JEE Adv. 2016)
Detailed Solution: Question 8
347 docs|185 tests |