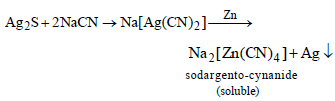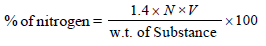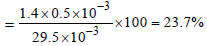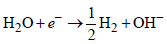JEE Main 35 Year PYQs- General Principles & Processes of Isolation Elements
MCQ Practice Test & Solutions: Test: JEE Main 35 Year PYQs- General Principles & Processes of Isolation of Elements (10 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: JEE Main 35 Year PYQs- General Principles & Processes of Isolation of Elements". These 10 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
Aluminium is extracted by the electrolysis of [2002]
Detailed Solution: Question 1
The metal extracted by leaching with a cyanide is [2002]
Detailed Solution: Question 2
Which one of the following ores is best concentrated byfroth-flotation method ? [2004]
Detailed Solution: Question 3
During the process of electrolytic refining of copper, some metals present as impurity settle as ‘anode mud’. These are [2005]
Detailed Solution: Question 4
Which of the following factors is of no significance forroasting sulphide ores to the oxides and not subjecting thesulphide ores to carbon reduction directly? [2008]
Detailed Solution: Question 5
29.5 mg of an organic compound containing nitrogen wasdigested according to Kjeldahl’s method and the evolvedammonia was absorbed in 20 mL of 0.1 M HCl solution. Theexcess of the acid required 15 mL of 0.1 M NaOH solutionfor complete neutralization. The percentage of nitrogen inthe compound is [2010]
Detailed Solution: Question 6
Which method of purification is represented by the following equation ? [2012]
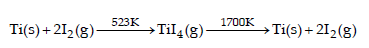
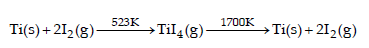
Detailed Solution: Question 7
The metal that cannot be obtained by electrolysis of anaqueous solution of its salts is: [JEE M 2014]
Detailed Solution: Question 8
In the context of the Hall - Heroult process for the extraction of Al, which of the following statements is false ?[JEE M 2015]
Detailed Solution: Question 9
Which one of the following ores is best concentrated byfroth floatation method? [JEE M 2016]
Detailed Solution: Question 10
446 docs|929 tests |