MCQs (One or More Correct Option): The d- and f-Block Elements & Coordination
MCQ Practice Test & Solutions: Test: MCQs (One or More Correct Option): The d- and f-Block Elements & Coordination Compounds | JEE Advanced (15 Questions)
You can prepare effectively for JEE Crack JEE with 35 Years of Previous Year Solved Papers with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: MCQs (One or More Correct Option): The d- and f-Block Elements & Coordination Compounds | JEE Advanced". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Potassium manganate (K2MnO4) is formed when
Detailed Solution: Question 1
The aqueous solutions of the following salts will be coloured in the case of
Detailed Solution: Question 2
Among the following ions which one has the highest paramagnetism?
Detailed Solution: Question 3
Which of the following alloys contains(s) Cu and Zn?
Detailed Solution: Question 4
In nitroprusside ion the iron and NO exist as FeII and NO+ rather than FeIII and NO. These forms can be differentiated by
Detailed Solution: Question 5
Addition of high proportions of manganese makes steel useful in making rails of railroads, because manganese
Detailed Solution: Question 6
If the bond length of CO bond in carbon monoxide is 1.128Å, then what is the value of CO bond length in Fe(CO)5?
Detailed Solution: Question 7
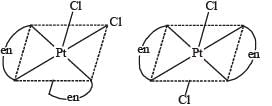
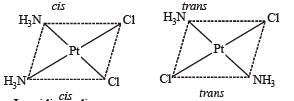
The compound(s) that exhibit(s) geometrical isomerism is (are)
Detailed Solution: Question 8
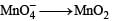
Reduction of the metal centre in aqueous permanganate ion involves
Detailed Solution: Question 9
Which of the following ions can push the reaction 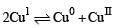 in the backward direction by forming stable complexes with Cu2+?
in the backward direction by forming stable complexes with Cu2+?
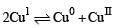 in the backward direction by forming stable complexes with Cu2+?
in the backward direction by forming stable complexes with Cu2+?Detailed Solution: Question 10
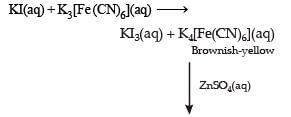
For the given aqueous reactions, which of the statement (s) is (are) true?
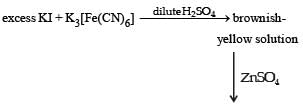



Detailed Solution: Question 11
The pair(s) of coordination complexes/ions exhibiting the same kind of isomerism is(are)
Detailed Solution: Question 12
The pair (s) of reagents that yield paramagnetic species is/are
Detailed Solution: Question 13
The correct statement(s) about Cr2+ and Mn3+ is(are) [Atomic numbers of Cr = 24 and Mn = 25]
Detailed Solution: Question 14
Fe3+ is reduced to Fe2+ by using
347 docs|185 tests |


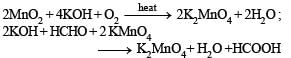

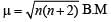
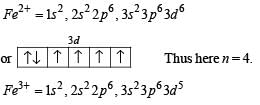


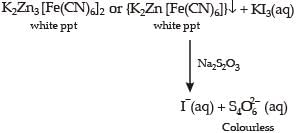
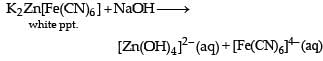
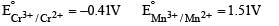 Above E° values explains reducing nature of Cr2+ and oxidizing behaviour of Mn3+.
Above E° values explains reducing nature of Cr2+ and oxidizing behaviour of Mn3+.