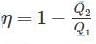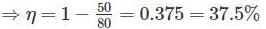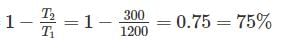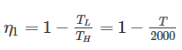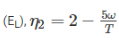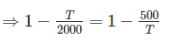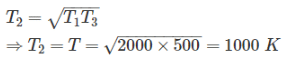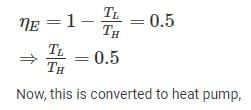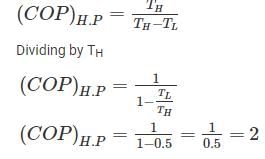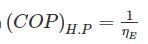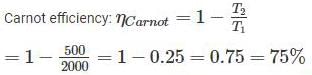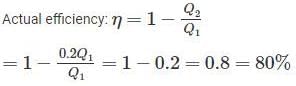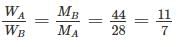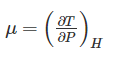SSC JE Mechanical Thermodynamics - 1 Free Online Test 2026
MCQ Practice Test & Solutions: Thermodynamics - 1 (20 Questions)
You can prepare effectively for Mechanical Engineering SSC JE Mechanical Mock Test Series 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Thermodynamics - 1". These 20 questions have been designed by the experts with the latest curriculum of Mechanical Engineering 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 12 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
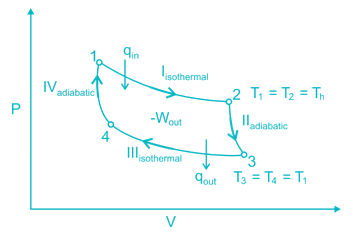
A thermodynamic cycle is composed of four processes. The heat added, and the work done in each process are as follows:
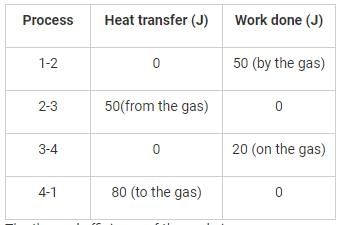
The thermal efficiency of the cycle is

Detailed Solution: Question 1
A heat reservoir is maintained at 927 °C. If the ambient temperature is 27 °C, the availability of heat from the reservoir is limited to
Detailed Solution: Question 2
Which of the following devices complies with the Clausius statement of the second law of thermodynamics?
Detailed Solution: Question 3
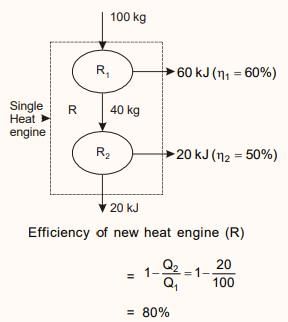
Two reversible engines are connected in series between a heat source and a sink. The efficiencies of these engines are 60% and 50%, respectively. If these two engines are replaced by a single reversible engine, the efficiency of this engine will be
Detailed Solution: Question 4
Detailed Solution: Question 5
The property of a working system which changes as the heat is supplied to the working fluid in a reversible manner is known as ________.
Detailed Solution: Question 6
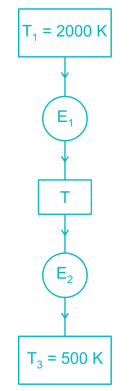
Two reversible heat engines operating between temperatures 2000 K and T K and T K and 500 K respectively. What is the intermediate temperature, if the efficiency of both the cycles is same?
Detailed Solution: Question 7
A Carnot heat engine is working with an efficiency of 50%. If the cycle is converted into a heat pump after reversing, then what is the coefficient of performance of the heat pump?
Detailed Solution: Question 8
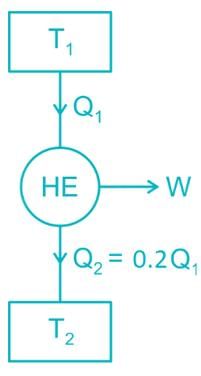
An inventor says, he has invented an engine which will reject 20% of heat absorbed from the source and the engine operates between 2000 K and 500 K. What kind of engine it is?
Detailed Solution: Question 9
In a cyclic process, the work done by the system is 20 kJ, -30 kJ, -5 kJ and 10 kJ. What is the net heat (kJ) for the cyclic process?
Detailed Solution: Question 10
Detailed Solution: Question 11
Detailed Solution: Question 12
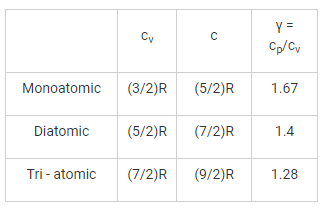
Which gas can attain the highest efficiency for the same compression rise?
Detailed Solution: Question 13
A tank containing air is stirred by a paddle wheel. The work input to the paddle wheel is 9000 kJ and heat transferred to the surroundings from the tank is 3000 kJ. The external work done by the system is:
Detailed Solution: Question 14
If pressure at any point in the liquid approaches the vapor pressure, liquid starts vaporising and creates pockets or bubbles of dissolved gases and vapours. This phenomenon is ________.
Detailed Solution: Question 15
Two gases A and B with their molecular weights 28 and 44 respectively, expand at constant pressures through the same temperature range. The ratio of quantity of work done by the two gases (A : B) is ________.
Detailed Solution: Question 16
A series of operations, which takes place in a certain order and restore the initial conditions at the end, is known as
Detailed Solution: Question 17
Detailed Solution: Question 18
Detailed Solution: Question 19
Detailed Solution: Question 20
3 videos|1 docs|55 tests |