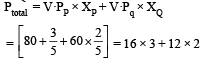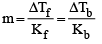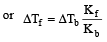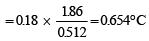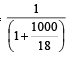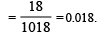31 Years NEET Previous Year Questions: Solutions - 2 - Free MCQ Test
MCQ Practice Test & Solutions: 31 Years NEET Previous Year Questions: Solutions - 2 (30 Questions)
You can prepare effectively for NEET Chemistry Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "31 Years NEET Previous Year Questions: Solutions - 2". These 30 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
An ideal solution is formed when its components [1988]
Detailed Solution: Question 1
All form ideal solution except [1988]
Detailed Solution: Question 2
The relative lowering of the vapour pressure is equal to the ratio between the number of [1991]
Detailed Solution: Question 3
Which of the following aqueous solution has minimum freezing point ? [1991]
Detailed Solution: Question 4
Blood cells retain their normal shape in solution which are [1991]
Detailed Solution: Question 5
Which of the following modes of expressing concentration is independent of temperature ? [1992,1995]
Detailed Solution: Question 6
Which one is a colligative property ? [1992]
Detailed Solution: Question 7
If 0.1 M solution of glucose and 0.1 M solution of urea are placed on two sides of the semipermeable membrane to equal heights, then it will be correct to say that [1992]
Detailed Solution: Question 8
At 25°C, the highest osmotic pressure is exhibited by 0.1 M solution of [1994]
Detailed Solution: Question 9
Which one of the following salts will have the same value of the van’t Hoff factor (i) as that of K4[Fe (CN)6]. [1994]
Detailed Solution: Question 10
The number of moles of oxygen in one litre of air containing 21% oxygen by volume, in standard conditions, is [1995]
Detailed Solution: Question 11
Vapour pressure of benzene at 30°C is 121.8 mm.When 15 g of a non volatile solute is dissolved in 250 g of benzene its vapour pressure decreased to 120.2 mm. The molecular weight of the solute (Mo. wt. of solvent = 78) [1995]
Detailed Solution: Question 12
According to Raoult's law, relative lowering of vapour pressure for a solution is equal to [1995
Detailed Solution: Question 13
The vapour pressure at a given temperature of an ideal solution containing 0.2 mol of a nonvolatile solute and 0.8 mol of solvent is 60 mm of Hg. The vapour pressure of the pure solvent at the same temperature is [1996]
Detailed Solution: Question 14
Which of the following 0.10 m aqueous solutions will have the lowest freezing point ? [1997]
Detailed Solution: Question 15
A 5% solution of cane sugar (mol. wt. =342) is isotonic with 1% solution of a substance X. The molecular weight of X is [1998]
Detailed Solution: Question 16
The vapour pressure of a solvent decreased by 10mm of mercury when a non-volatile solute was added to the solvent. The mole fraction of the solute in the solution is 0.2. What should be the mole fraction of the solvent if the decrease in the vapour pressure is to be 20mm of mercury?
Detailed Solution: Question 17
Which of the following statements, regarding the mole fraction (x) of a component in solution, is incorrect? [1999]
Detailed Solution: Question 18
Which of the following colligative property can provide molar mass of proteins (or polymers or colloids) with greatest precision ? [2000]
Detailed Solution: Question 19
The beans are cooked earlier in pressure cooker, because [2001]
Detailed Solution: Question 20
Molarity of liquid HCl will be, if density of solution is 1.17 gm/cc [2001]
Detailed Solution: Question 21
1 M, 2.5 litre NaOH solution is mixed with another 0.5 M, 3 litre NaOH solution. Then find out the molarity of resultant solution [2002]
Detailed Solution: Question 22
A solution contains non - volatile solute of molecular mass M2. Which of the following can be used to calculate the molecular mass of solute in terms of osmotic pressure ? [2002]
Detailed Solution: Question 23
A solution containing components A and B follows Raoult's law when [2002]
Detailed Solution: Question 24
Formation of a solution from two components can be considered as [2003]
(i) Pure solvent → separated solvent molecules, ΔH1
(ii) Pure solute → separated solute molecules, ΔH2
(iii) Separted solvent & solute molecules → Solution, ΔH3
Solution so formed will be ideal if
(a) ΔHsoln = ΔH1 + ΔH2 - ΔH3
(b) ΔHsoln = ΔH1 + ΔH2 + ΔH3
(c) ΔHsoln = ΔH1 - ΔH2 - ΔH3
(d) ΔHsoln = ΔH3 - ΔH1 - ΔH2
(i) Pure solvent → separated solvent molecules, ΔH1
(ii) Pure solute → separated solute molecules, ΔH2
(iii) Separted solvent & solute molecules → Solution, ΔH3
Solution so formed will be ideal if
Detailed Solution: Question 25
Camphoris often used in molecular mass determination because [2004]
Detailed Solution: Question 26
The vapour pressure of two liquids ‘P’ an d ‘Q’ are 80 and 60 torr, respectively. The total vapour pressure of solution obtained by mixing 3 mole of P and 2 mole of Q would be [2005]
Detailed Solution: Question 27
A solution of urea (m ol. mass 56 g mol-1) boils at 100.18°C at the atmospheric pressure. If Kf and Kb for water are 1.86 and 0.512 K kg mol-1 respectively, the above solution will freeze at [2005]
Detailed Solution: Question 28
The mole fraction of the solute in one molal aqueous solution is: [2 00 5]
Detailed Solution: Question 29
A solution of acetone in ethanol [2006]
Detailed Solution: Question 30
54 videos|290 docs|74 tests |


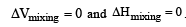

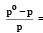 mole fraction of solute
mole fraction of solute 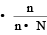

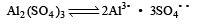
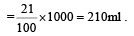
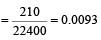
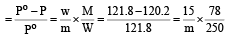
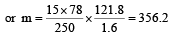
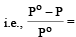 mole fraction of solute
mole fraction of solute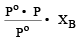
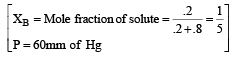
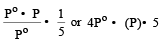
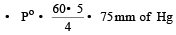
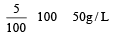
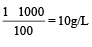
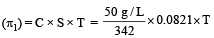
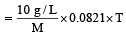
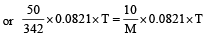
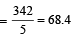
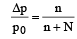 (mole fraction of solute)
(mole fraction of solute)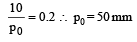
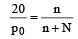 (Mole fraction of solute)
(Mole fraction of solute)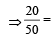 mole fraction of solute
mole fraction of solute
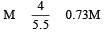

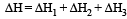 (Accroding to Hess's law) i.e., for ideal solutions there is no change in magnitude of the attractive forces in the two components present.
(Accroding to Hess's law) i.e., for ideal solutions there is no change in magnitude of the attractive forces in the two components present.