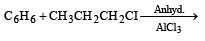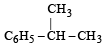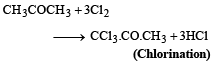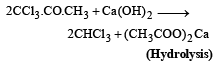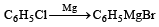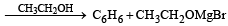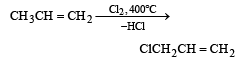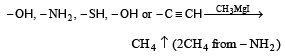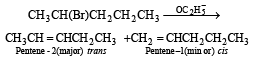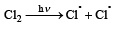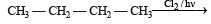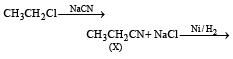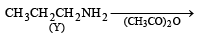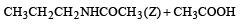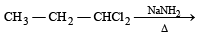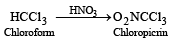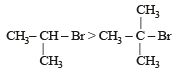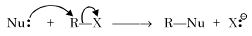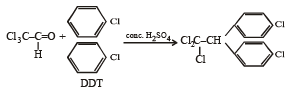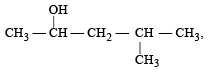31 Year NEET Previous Questions: Haloalkanes & Haloarenes Free MCQ Practice
MCQ Practice Test & Solutions: 31 Year NEET Previous Year Questions: Haloalkanes & Haloarenes (25 Questions)
You can prepare effectively for NEET Chemistry Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "31 Year NEET Previous Year Questions: Haloalkanes & Haloarenes". These 25 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 48 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Phosgene is a common name for [1988]
Detailed Solution: Question 1
Which chloro derivative of benzene among the following would undergo hydrolysis most readily with aqueous sodium hydroxide to furnish the corresponding hydroxy derivative?
Detailed Solution: Question 2
Benzene reacts with n-propyl chloride in the presence of anhydrous AlCl3 to give [1993]
Detailed Solution: Question 3
Industrial preparation of chloroform employs acetone and [1993]
Detailed Solution: Question 4
Chlorobenzene reacts with Mg in dry ether to give a compound (A) which further reacts with ethanol to yield [1993]
Detailed Solution: Question 5
When chlorine is passed through propene at 400°C, which of the following is formed ?[1993]
Detailed Solution: Question 6
Zerevitinov’s determination of active hydrogen in a compound is based upon its reaction with[1994]
Detailed Solution: Question 7
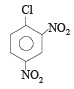
The replacement of chlorine of chlorobenzene to give phenol requires drastic conditions, but the chlorine of 2,4-dinitrochlorobenzene is readily replaced since, [1997]
Detailed Solution: Question 8
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is[1998]
Detailed Solution: Question 9
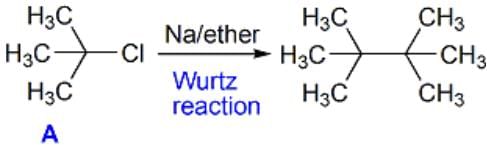
An organic compound A (C4H9Cl) on reaction with Na/diethyl ether gives a hydrocarbon which on monochlorination gives only one chloro derivative, then A is [2001]
Detailed Solution: Question 10
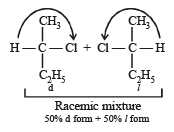
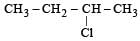 obtained by chlorination of n-butane, will be [2001]
obtained by chlorination of n-butane, will be [2001]
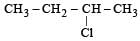 obtained by chlorination of n-butane, will be [2001]
obtained by chlorination of n-butane, will be [2001]Detailed Solution: Question 11
Reactivity order of halides for dehydrohalogenation is [2002]
Detailed Solution: Question 12
Z in the above reaction sequence is [2002]
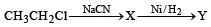

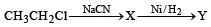

Detailed Solution: Question 13
When CH3CH2CHCl2 is treated with NaNH2, the product formed is [2002]
Detailed Solution: Question 14
Which of the following is responsible for depletion of the ozone layer in the upper strata of the atmosphere? [2004]
Detailed Solution: Question 15
Chloropicrin is obtained by the reaction of
Detailed Solution: Question 16
In a SN2 substitution reaction of the type 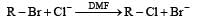 [2008] which one of the following has the highest relative rate ?
[2008] which one of the following has the highest relative rate ?
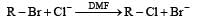 [2008] which one of the following has the highest relative rate ?
[2008] which one of the following has the highest relative rate ?Detailed Solution: Question 17
Which of the following reactions is an example of nucleophilic substitution reaction? [2009]
Detailed Solution: Question 18
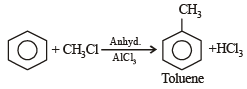
Benzene reacts with CH3Cl in the presence of anhydrous AlCl3 to form: [2009]
Detailed Solution: Question 19
Trichloroacetaldehyde, CCl3CHO reacts with chlorobenzene in presence of sulphuric acid and produces: [2009]
Detailed Solution: Question 20
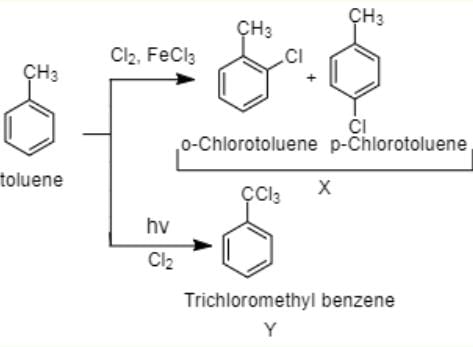
The reaction of toluene with Cl2 in presence of FeCl3 gives ' X' and reaction in presence of light gives ‘Y’. Thus, ‘X’ and ‘Y’ are : [2010]
Detailed Solution: Question 21
Which one is most reactive towards SN1 reaction ?
Detailed Solution: Question 22
Consider the reactions : [2011 M]
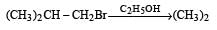
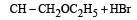
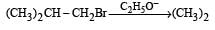
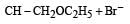
The mechanisms of reactions (i) and (ii) are respectively :
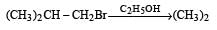
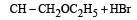
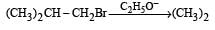
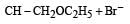
Detailed Solution: Question 23
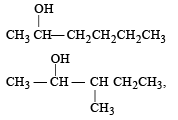
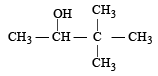
Number of isomeric alcohols of molecular formula C6H14O which give positive iodoform test is [NEET Kar. 2013]
Detailed Solution: Question 24
Which of the following is the correct order of boiling points for the given compounds?
Detailed Solution: Question 25
54 videos|290 docs|74 tests |