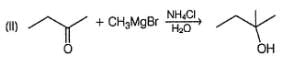Preparation of Alcohols - Free MCQ Practice Test with solutions, NEET Chemistry
MCQ Practice Test & Solutions: Test: Preparation of Alcohols (20 Questions)
You can prepare effectively for NEET Chemistry Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Preparation of Alcohols". These 20 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
Direction (Q. Nos. 1-8) This section contains 8 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q, Which of the following pairs of compounds can be used as starting material in the synthesis of 2-phenyl-2-pentanol?
Detailed Solution: Question 1
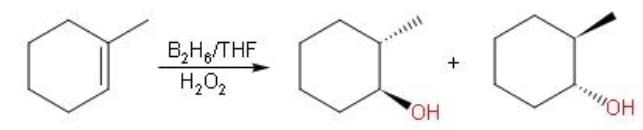
What is the product of the reaction of methyl cyclohexene with B2H6 in THF followed by the oxidation with alkaline H2O2?
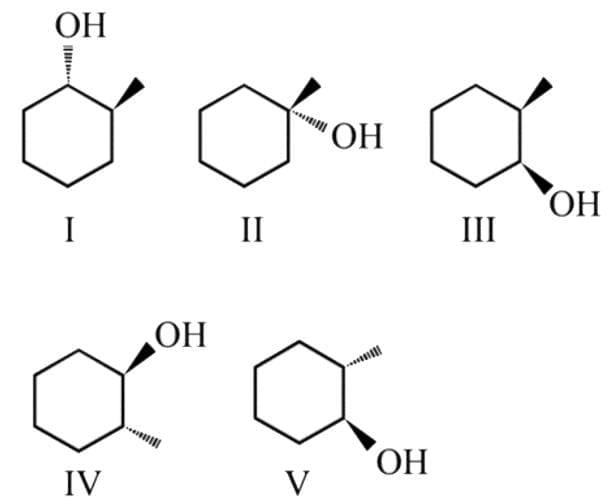

Detailed Solution: Question 2
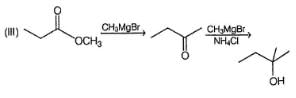
Choose the reagent and reactant that would produce 2-methyl-2-butanol as the major product.
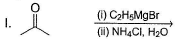
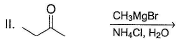
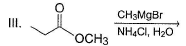
Detailed Solution: Question 3
Detailed Solution: Question 4
What is the correct structure for the major compound produced by the following reaction sequence?
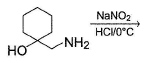
Detailed Solution: Question 5
In the reaction given below,
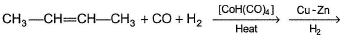
The final major organic product is
Detailed Solution: Question 6
Primary alcohol can easily be prepared from primary alkyl halide via SN2 reaction with aqueous NaOH. However, similar method does not work for the preparation of tertiary alcohol. Which reaction can be used for the efficient preparation of tertiary alcohol (tertiary butanol) from tertiary butyl bromide?
Detailed Solution: Question 7
In the following reaction,
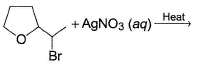
The possible substitution product(s) is/are
Detailed Solution: Question 8
In the following reaction,
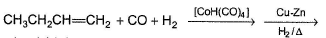
Possible product(s) is/are
Detailed Solution: Question 9
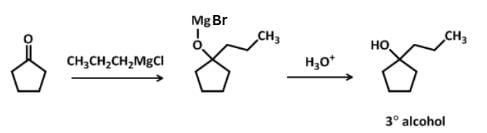
Which of the following Grignard’s synthesis can result into 2-cyclopentyl-2-butanol?
Detailed Solution: Question 10
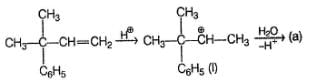
Aliphatic alcohol when treated with dilute H2SO4, undergo isomerisation via reversible reaction. In the following reaction, which of the isomers are expected to be present at equilibrium?
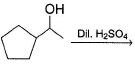
Detailed Solution: Question 11
In the following reaction,
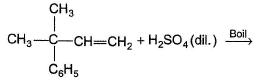
The alcohol(s) formed in significant yield is/are
Detailed Solution: Question 12
In the reaction given below,
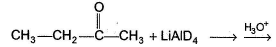
The correct statement regarding the outcome of the above reaction is/are
Detailed Solution: Question 13
Consider the following reaction,
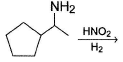
Possible product(s) is/are
Detailed Solution: Question 14
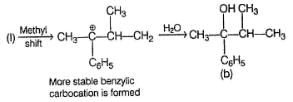
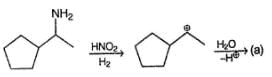
Consider the following sequence of reaction,
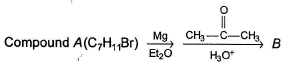
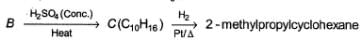
Q.
The structure of compound B is
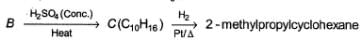
Detailed Solution: Question 15
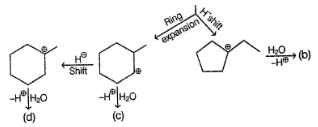
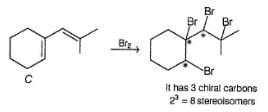
Consider the following sequence of reaction,
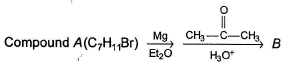
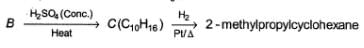
Q.
If C is treated with excess of Br2(l) how many different isomers of bromination product(s) result?
Detailed Solution: Question 16
One Integer Value Correct Type
Direction (Q. Nos. 19-22) This section contains 4 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
In the following reaction,
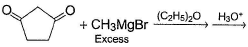
Q.
How many different diols are formed as a result of nucleophilic addition reaction?
Detailed Solution: Question 17
In the reaction given below,
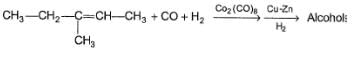
How many different products are expected?
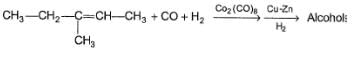
Detailed Solution: Question 18
If 3, 3-dimethyl-2, 4-pentanedione is treated with a Grignard reagent consisting of mixture of CH3MgBr and C2H5MgBr and finally hydrolysing product with dilute H2SO4 results in the formation of how many different diols?
Detailed Solution: Question 19
If a pure enantiomer of 3-methyl-1-pentene is treated with boiling solution of dilute H2SO4, how many different alcohols are expected in principle?
Detailed Solution: Question 20
54 videos|290 docs|74 tests |