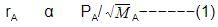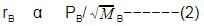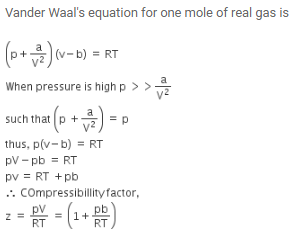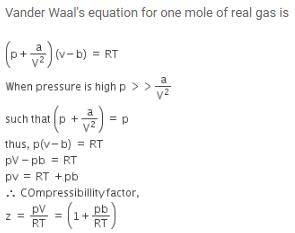JEE Previous Year Questions- States of Matter with solutions - Free MCQ
MCQ Practice Test & Solutions: Test: JEE Previous Year Questions- States of Matter (14 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: JEE Previous Year Questions- States of Matter". These 14 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 14 minutes
- - Number of Questions: 14
Sign up on EduRev for free to attempt this test and track your preparation progress.
According to the kinetic theory of gases, in an ideal gas, between two successive collisions a gas molecule travels –
[AIEEE-2003]
Detailed Solution: Question 1
As the temperature is raised from 20°C to 40°C, the average kinetic energy of neon atoms changes by a factor of which of the following ?
[AIEEE-2004]
Detailed Solution: Question 2
In van der Waals equation of state of the gas law, the constant `b' is a measure of -
[AIEEE-2004]
Detailed Solution: Question 3
Which one of the following statements is NOT true about the effect of an increase in temperature on the distribution of molecular speeds in a gas ?
[AIEEE-2005]
Detailed Solution: Question 4
Equal masses of methane and oxygen are mixed in an empty container at 25ºC. The fraction of the total pressure exerted by oxygen is –
[AIEEE-2007]
Detailed Solution: Question 5
The no. of moles per litre in the equation PV = nRT is expressed by -
[AIEEE-2002]
Detailed Solution: Question 6
The correct value of R is -
[AIEEE-2002]
Detailed Solution: Question 7
If 10-4 dm3 of water is introduced into a 1.0 dm3 flask at 300 K, how many moles of water are in in the vapour phase when equilibrium is established ?
(Given : Vapour pressure of H2O at 300 is 3170 pa; R = 8.314 JK-1 mol)
[AIEEE -2010]
Detailed Solution: Question 8
`a' and `b' are Vander Waals' constant for gases. Chlorine is more easily liquefied than ethane because :
[AIEEE - 2011]
Detailed Solution: Question 9
When r, P and M represent rate of diffusion, pressure and molecular mass, respectively, then the ratio of the rates of diffusion (rA/rB) of two gases A and B, is given as -
[AIEEE - 2011]
Detailed Solution: Question 10
The molecular velocity of any gas is -
[AIEEE - 2011]
Detailed Solution: Question 11
The compressibility factor for a real gas at high pressure is –
[AIEEE-2012]
Detailed Solution: Question 12
For gaseous state, if most probable speed is denoted by C*, average speed by 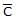 and mean square speed by C, then for a large number of molecules the ratios of these speeds are :
and mean square speed by C, then for a large number of molecules the ratios of these speeds are :
[Jee(Main) 2013, 3/120]
Detailed Solution: Question 13
A gaseous hydrocarbon gives upon combustion 0.72 g of water and 3.08 g. of CO2. The empirical formula of the Hydrocarbon is :
[Jee(Main) 2013, 3/120]
Detailed Solution: Question 14
335 videos|699 docs|300 tests |