Test: Structural of Metals & Alloys - 3 - Mechanical Engineering MCQ
20 Questions MCQ Test - Test: Structural of Metals & Alloys - 3
When atoms are displaced in two separate planes perpendicular to each other, the defect so produced is known as
Atomic packing factor in case of Copper Crystal is
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
The effective number of lattice points in the unit celi of simple cubic, body centered cubic and face centered cubic space lattices respectively are
Which of the following elements is not a metalloid?
Which of the following material is not a ferromagnetic material?
Which one of the following pairs of axis lengths (a, b, c) and inter axial angles (a, β, γ) represents the tetragonal crystal system?
The defect which takes place due to imperfect packing of atoms during crystallization is known as
What is the movement of block of atoms along certain crystallographic phase and directions termed as
A screw dislocation
1. Lies parallel to its Burger’s vector
2. Lies perpendicular to its Burger’s vector
3. Moves in a perpendicular direction to the Burger’s vector
4. Moves in an inclined direction to the Burger’s vector
Q. Select the correct answer using the codes given below:
The set of miller indices of the plane shown in the given figure is

Match List-I (Element) with List-ll (Crystal structure) and select the correct answer using the codes given below the lists:
List-I
A. Alpha Iron
B. Copper
C. Zinc
D. Glass
List-ll
1. Hexagonal closed packed
2. Body-centered cubic
3. Amorphous
4. Face-centered cubic
Codes:
A B C D
(a) 2 3 1 4
(b) 1 4 2 3
(c) 2 4 1 3
(d) 1 3 2 4
What is the planer density of (100) plane in FCC (face-centred cubic) crystal with unit cell side a equa! to?
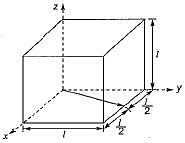
A unit cell of a crystal is shown in given figure. The miller indices of the direction (arrow) shown in the figure is
Match List-I (Miller indices) with List-ll (Denotes) and select the correct answer using the codes given below the lists:
List-I
A. (h, k l)
B. [h k l]
C. {h k l}
D. <h k l>
List-ll
1. direction
2. plane
3. family of direction
4. Family of planes
Codes:
A B C D
(a) 1 2 3 4
(b) 1 2 4 3
(c) 2 1 4 3
(d) 2 1 3 4
The linear density along the direction (110) will be equal to, if lattice constant of copper unit cell is 3.61 Å.
Miller indices of a plane will be whose intercepts are a, b/2 and 3c on x, y and z axes respectively in a simple cubic unit cell?
Consider the following statements about screw dislocation
1. It forms when crystal displaces angulanly over the remaining parts
2. Burgers vector is parallel to screw dislocation line
3. Screw dislocations are symbolically represented by clockwise & anticiock-wise and reffered to negative & positive screw dislocation respectively
Q. Which of these statement are correct?
Ratio of elastic strain energy of an edge dislocation to the screw dislocation is



 .
.




















