Solids, Liquids & Gases- 1 - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Solids, Liquids & Gases- 1 (10 Questions)
You can prepare effectively for Class 4 Science for Class 4 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Solids, Liquids & Gases- 1". These 10 questions have been designed by the experts with the latest curriculum of Class 4 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
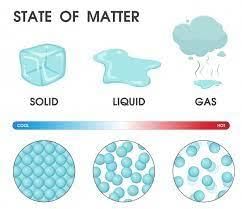
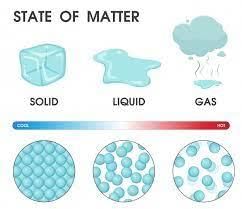
Choose the correct option from the following on the basis of the packing of molecules.
Detailed Solution: Question 1
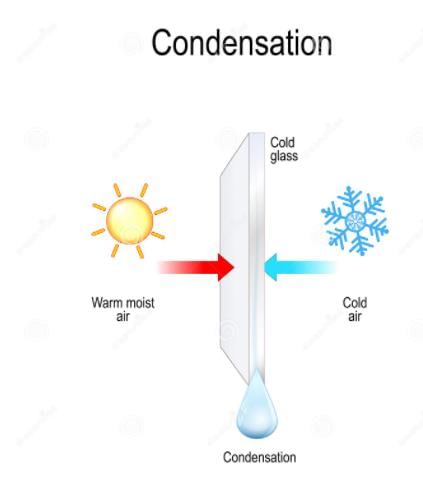
When a gas changes into a liquid, it is called _______.
Detailed Solution: Question 2
State whether the following statement is True or False:
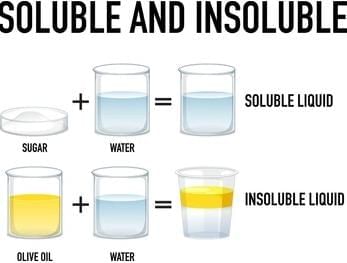
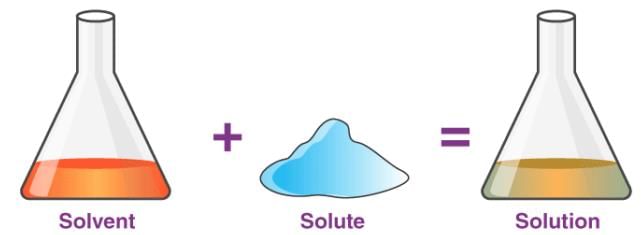
Some solids that dissolve in water are called soluble substances.
Detailed Solution: Question 3
Detailed Solution: Question 4
State whether the following statement is True or False:
The substance that dissolves in a liquid to form a solution is called a solute.
Detailed Solution: Question 5
What is the state of matter that takes the shape of its container?
Detailed Solution: Question 6
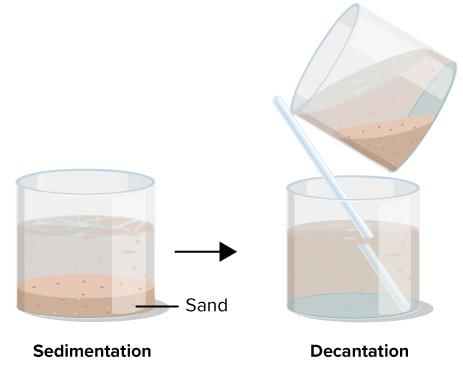
What method is used to separate a mixture of sand and water?
Detailed Solution: Question 7
What changes occur to the molecules of a substance during evaporation?
Detailed Solution: Question 8
Which state of matter is characterized by the highest rate of diffusion?
Detailed Solution: Question 9
What is the main reason why gases are highly compressible compared to liquids and solids?
Detailed Solution: Question 10
48 videos|193 docs|34 tests |