Solids, Liquids & Gases- 2 - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Solids, Liquids & Gases- 2 (10 Questions)
You can prepare effectively for Class 4 Science for Class 4 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Solids, Liquids & Gases- 2". These 10 questions have been designed by the experts with the latest curriculum of Class 4 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of these is a characteristic property of gases?
Detailed Solution: Question 1
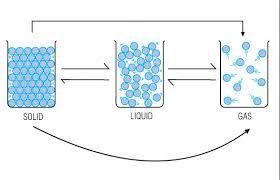
The conversion of matter from solid state to liquid state is called ________.
Detailed Solution: Question 2
Read the statements carefully and choose the correct option.
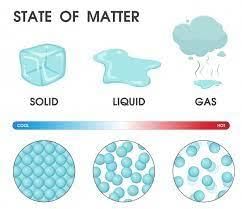
Statement A: In solids the molecules are packed the closest together.
Statement B: In gases the molecules are spread out the most.
Statement A: In solids the molecules are packed the closest together.
Statement B: In gases the molecules are spread out the most.
Detailed Solution: Question 3
Detailed Solution: Question 4
Detailed Solution: Question 5
Which process would be used to separate sugar from a sugar-water solution?
Detailed Solution: Question 6
What happens to the rate of diffusion when a substance changes from a solid to a liquid?
Detailed Solution: Question 7
What is the outcome when a substance undergoes condensation?
Detailed Solution: Question 8
Which of the following is an example of an insoluble substance in water?
Detailed Solution: Question 9
Detailed Solution: Question 10
48 videos|193 docs|34 tests |