Structure of the Atom- Case Based Type Questions - Free with solutions
MCQ Practice Test & Solutions: Test: Structure of the Atom- Case Based Type Questions (12 Questions)
You can prepare effectively for Class 9 Class 9: Additional Practice with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Structure of the Atom- Case Based Type Questions". These 12 questions have been designed by the experts with the latest curriculum of Class 9 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 24 minutes
- - Number of Questions: 12
Sign up on EduRev for free to attempt this test and track your preparation progress.
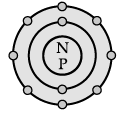
Direction: The electronic configuration of an element ‘X’ is 2, 8, 2 :
Q. How many Valence electrons are there in element X?
Detailed Solution: Question 1
Direction: The electronic configuration of an element ‘X’ is 2, 8, 2 :
Q. The number of electrons present in the atom of element ‘X’ is __________.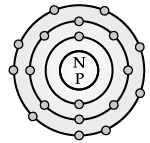

Detailed Solution: Question 2
Direction: The electronic configuration of an element ‘X’ is 2, 8, 2 :
Q. The Valency of element X is:
Detailed Solution: Question 3
Direction: The electronic configuration of an element ‘X’ is 2, 8, 2 :
Q. The element X is:
Detailed Solution: Question 4
Direction: In the following table the mass number and the atomic number of certain elements are given. Study the given data and answer the following questions :
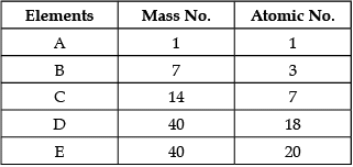
Q. The element which can form a cation:

Detailed Solution: Question 5
Direction: In the following table the mass number and the atomic number of certain elements are given. Study the given data and answer the following questions :
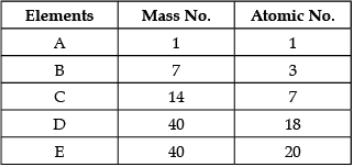
Q. Any two elements are Isobars:

Detailed Solution: Question 6
Direction: In the following table the mass number and the atomic number of certain elements are given. Study the given data and answer the following questions :

Q. Which element will form an anion?

Detailed Solution: Question 7
Direction: In the following table the mass number and the atomic number of certain elements are given. Study the given data and answer the following questions :
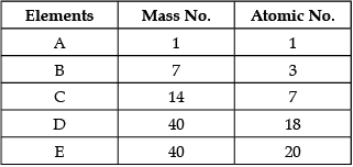
Q. Which element is a noble gas?

Detailed Solution: Question 8
Direction: Read the following passage and answer the following questions.
In order to overcome the objections raised against Rutherford’s model of the atom, Neil Bohr put forward the following postulates about the model of an atom.
Q. Who amended Rutherford’s short comings?
Detailed Solution: Question 9
Direction: Read the following passage and answer the following questions.
In order to overcome the objections raised against Rutherford’s model of the atom, Neil Bohr put forward the following postulates about the model of an atom.
Q. Atoms are made up of ______, _____ and ______.
Detailed Solution: Question 10
Direction: Read the following passage and answer the following questions.
In order to overcome the objections raised against Rutherford’s model of the atom, Neil Bohr put forward the following postulates about the model of an atom.
Q. Atomic mass is the sum of:
Detailed Solution: Question 11
Direction: Read the following passage and answer the following questions.
In order to overcome the objections raised against Rutherford’s model of the atom, Neil Bohr put forward the following postulates about the model of an atom.
Q. The number of electrons that K-shell and L-shell can accommodate:

Detailed Solution: Question 12
4 docs|108 tests |


