NCERT Based Test: Electronic Configuration of Elements & The Periodic Table
MCQ Practice Test & Solutions: NCERT Based Test: Electronic Configuration of Elements & The Periodic Table (5 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "NCERT Based Test: Electronic Configuration of Elements & The Periodic Table". These 5 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 5 minutes
- - Number of Questions: 5
Sign up on EduRev for free to attempt this test and track your preparation progress.
Anything that influences the valence electrons will affect the chemistry of the element. Which one of the following factors does not affect the valence shell?
Detailed Solution: Question 1
Which of the following elements shown as pairs with their atomic numbers belong to the same period?
Detailed Solution: Question 2
To which group,an element with atomic number 88 will belong?
Detailed Solution: Question 3
Match the column I with column II and mark the appropriate choice.
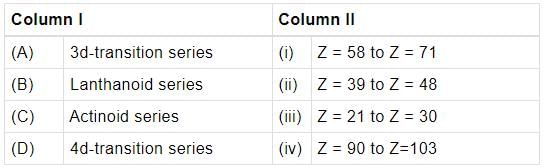

Detailed Solution: Question 4
An element has atomic number 79. Predict the group and period in which the element is placed.
Detailed Solution: Question 5
97 videos|243 docs|71 tests |


