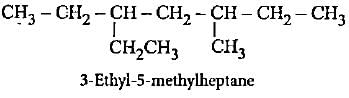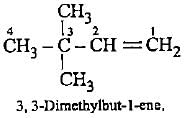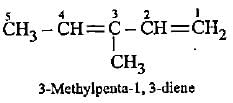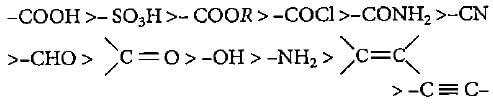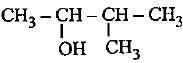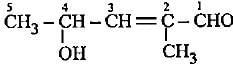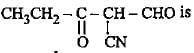Nomenclature of Organic Compounds (NCERT) - Free MCQ Practice Test
MCQ Practice Test & Solutions: Test: Nomenclature of Organic Compounds (NCERT) (20 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Nomenclature of Organic Compounds (NCERT)". These 20 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
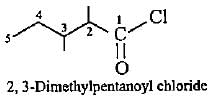
lUPAC names of the given structures are


Detailed Solution: Question 1
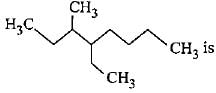
Correct name for the given compound,
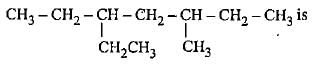
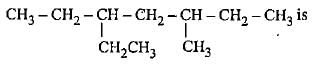
Detailed Solution: Question 2
Detailed Solution: Question 3
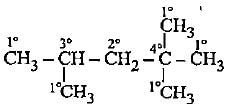
How many primary, secondary, tertiary and quaternary carbon atoms are present in the following compound?
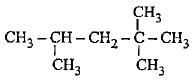
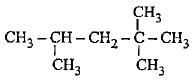
Detailed Solution: Question 4
The lUPAC name of the compound having formula
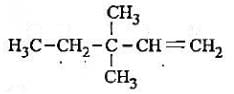

Detailed Solution: Question 5
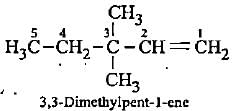
lUPAC name of (CH3)3C - CH = CH2 is
Detailed Solution: Question 6
Which of the following represents 3-methylpenta-1, 3-diene?
Detailed Solution: Question 7
Detailed Solution: Question 8
The correct decreasing order of priority for the functional groups of organic compounds In the lUPAC system of nomenclature Is
Detailed Solution: Question 9
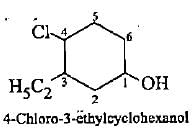
Which of the following compounds Is not correctly matched with Its lUPAC name?
Detailed Solution: Question 10
The correct representation of 4-hydroxy-2-methylpent-2-en-1 -al Is
Detailed Solution: Question 11
Detailed Solution: Question 12
Detailed Solution: Question 13
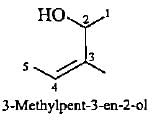
Correct representation of 3-methylpent-3-en-2-ol is
Detailed Solution: Question 14
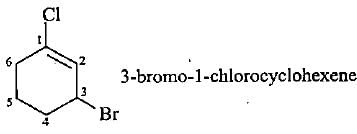
Which of the following lUPAC names is not correctly matched?
Detailed Solution: Question 15
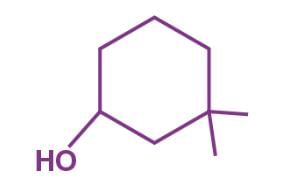
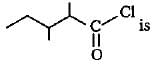
The correct lUPAC name of the compound
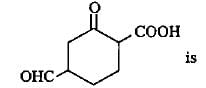

Detailed Solution: Question 16
The correct IUPAC name of the following compound is
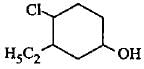

Detailed Solution: Question 17
The lUPAC name of the compound shown below is
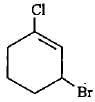

Detailed Solution: Question 18
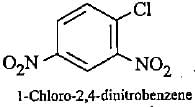
Which ofthe following names of substituted benzene compounds is not correct?
Detailed Solution: Question 19
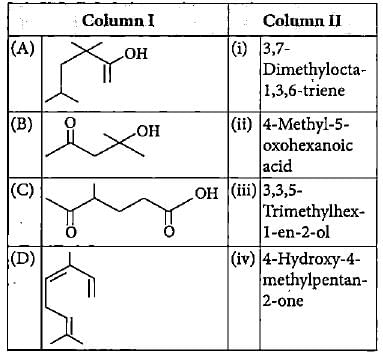
Match the compounds given in column I with the TUPAC names given in column II and mark the appropriate choice.

98 videos|243 docs|71 tests |