Quantum Mechanical Model of an Atom - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Quantum Mechanical Model of an Atom (15 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Quantum Mechanical Model of an Atom". These 15 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Direction (Q. Nos. 1-12) This section contains 12 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q.
Radial wave functio ns (R) of different orbitals are plotted. Which is/are correct graphs?
Detailed Solution: Question 1
For 2s-orbital electron, radial probability density R2 as function of r (distance) is given by
Detailed Solution: Question 2
For an electron in 2p-orbital, radial probability function 4πr2R2 as a function of r is given by
Detailed Solution: Question 3
C onsider the following figures A and B indicating distribution of charge density (electron probability 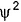 ) with distance r
) with distance r
Select the correct statement(s).
Detailed Solution: Question 4
Detailed Solution: Question 5
Which orbital gives an electron, a greater probability being found close to the nucleus?
Detailed Solution: Question 6
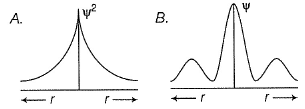
Hamiltonian operator 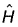 is the sum of two energy operators. These are
is the sum of two energy operators. These are
Detailed Solution: Question 7
Angular nodes in 4s- suborbit is equal to radial nodes in
Detailed Solution: Question 8
Detailed Solution: Question 9
There is formation of in all five nodes (including terminal nodes) in a string of 12 cm. Thus, wavelength of the waves formed is
Detailed Solution: Question 10
For x -axis , wave function (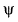 ) can be written as
) can be written as
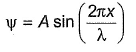
Thus, Schrodinger wave equation along x-axis can be written as :
Detailed Solution: Question 11
The Schrodinger wave equation for H-atom is
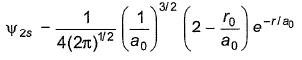
where, a0 is Bohr’s radius. If radial node in 2s is at distance r0, then
Detailed Solution: Question 12
Direction (Q. Nos. 13-15) This section contains 3 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
Q. Which of the following properties can be described by wave function 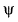 ?
?
Detailed Solution: Question 13
As compared to 1s electron of H-atom in ground state, which of the following properties appear(s) in the radial probability density of 2s electron of H-atom in first excited state?
Detailed Solution: Question 14
Radial probability density in the occupied orbital of a hydrogen atom in the ground state (1s) is given below
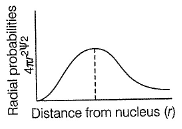
Detailed Solution: Question 15
98 videos|243 docs|71 tests |


