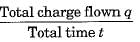RSMSSB Lab Assistant Paper 2 Mock Test - 4 Free Online Test 2026
Full Mock Test & Solutions: RSMSSB Lab Assistant Paper 2 Mock Test - 4 (100 Questions)
You can boost your REET 2026 exam preparation with this RSMSSB Lab Assistant Paper 2 Mock Test - 4 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of REET 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 120 minutes
- - Total Questions: 100
- - Analysis: Detailed Solutions & Performance Insights
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
Which of the following is incorrect about the given graph?
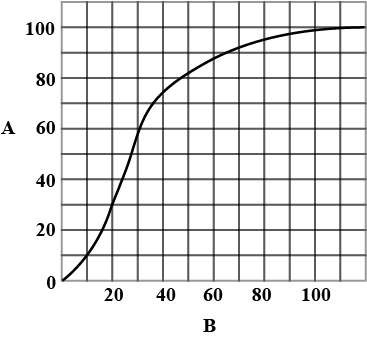

Detailed Solution: Question 1
Four liters of a diatomic ideal gas ( λ =1.4) confined to a cylinder is subject to a closed cycle. Initially, the gas is at 1.00 atm and at 300 K. First, its pressure is tripled under constant volume. Then, it expands adiabatically to its original pressure. Finally, the gas is compressed isobarically to its original volume. Find the temperature at the end of the cycle
Detailed Solution: Question 2
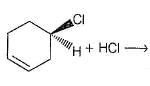
Consider the following reaction,
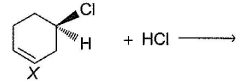
Q.
When a pure enantiomer of X is taken in the above reaction, correct completion regarding the reaction is/are
Detailed Solution: Question 3
Photophosphorylation is the process in which
Detailed Solution: Question 4
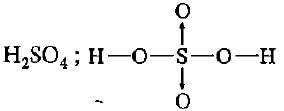
Which of the following molecules contains covalent and coordinate bonds?
Detailed Solution: Question 5
Which of the following is/are not the characteristics of the class Osteichthyes?
(i) Body is streamlined and mouth is terminal.
(ii) Gills are covered by operculum.
(iii) Skin covered with cycloid and placoid scales.
(iv) Many of them are viviparous.
(i) Body is streamlined and mouth is terminal.
(ii) Gills are covered by operculum.
(iii) Skin covered with cycloid and placoid scales.
(iv) Many of them are viviparous.
Detailed Solution: Question 6
Which species has the largest size?Option
Detailed Solution: Question 7
Given below are few mixtures formed by mixing two components. Which of the following binary mixtures will have same composition in liquid and vapour phase?
(i) Ethanol + Chloroform
(ii) Nitric acid + Water
(iii) Benzene + Toluene
(iv) Ethyl chloride + Ethyl bromide
(i) Ethanol + Chloroform
(ii) Nitric acid + Water
(iii) Benzene + Toluene
(iv) Ethyl chloride + Ethyl bromide
Detailed Solution: Question 8
A rectangular conductor PQRS in which the conductor PQ is free to move is kept in a magnetic field B ,which is perpendicular to the PQRS which lies in a single plane. If the conductor PQ has a length l,moves with a speed v, then induced emf is
Detailed Solution: Question 9
Phenols do not respond to which of these tests?
Detailed Solution: Question 10
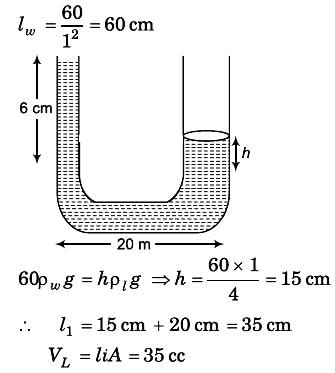
A U-tube having horizontal arm of length 20 cm, has uniform cross-sectional area = 1 cm2. It is filled with water of volume 60 cc. What volume of a liquid of density 4 g/cc should be poured from one side into the U-tube so that no water is left in the horizontal arm of the tube?
Detailed Solution: Question 11
Predict major product of the following reaction.
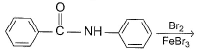
Detailed Solution: Question 12
Consider the following reaction,
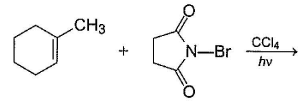
Q.
The expected product(s) is/are
Detailed Solution: Question 13
Which bacterium helps in the production of ‘Swiss cheese’?
Detailed Solution: Question 14
Which of the statements given above is/are correct?
i. The respiratory rhythm centre is primarily located in the medulla region of the brain.
ii. The pneumotaxic centre, found in the pons, enhances the duration of inspiration.
iii. Chemosensitive areas respond to increases in CO2 and hydrogen ions to regulate breathing.
iv. Oxygen plays a significant role in the regulation of respiratory rhythm.
Detailed Solution: Question 15
Topic in NCERT: REGULATION OF RESPIRATIONLine in NCERT: "Respiratory rhythm is maintained by the respiratory centre in the medulla region of brain. A pneumotaxic centre in the pons region of the brain and a chemosensitive area in the medulla can alter respiratory mechanism."
In the reaction N2 + 3H2 ⇌ 2NH3
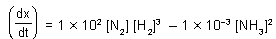 and at some instant if
and at some instant if
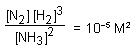
then at this instant value of 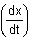 is :
is :
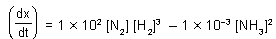 and at some instant if
and at some instant if 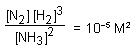
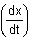 is :
is :Detailed Solution: Question 16
Independent assortment of genes does not take place when
Detailed Solution: Question 17
Four capacitors with capacitances C1 = 1 μF, C2 = 1.5 μF, C3 = 2.5 μF and C4 = 0.5 μF are connected as shown and are connected to 30 volt source. The potential difference between points a and b is
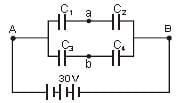

Detailed Solution: Question 18
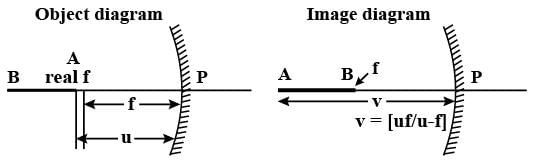
An infinitely long rod lies along the axis of a concave mirror of focal length f. The near end of the rod is at a distance u > f from the mirror. Its image will have a length
Detailed Solution: Question 19
A satellite moves in a circular orbit around earth. The radius of this orbit is one half that of moon’s orbit. The satellite completes one revolution in:
Detailed Solution: Question 20
Study the food web given below and answer the questions that follow.
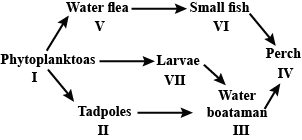
Which of the following organisms in the given food web acts as a secondary consumer?

Detailed Solution: Question 21
Topic in NCERT: Trophic Levels in an Ecosystem
Line in NCERT: "Those animals that depend on the primary carnivores for food are labelled secondary carnivores."
Figure shows plot of PV/T versus P for 1.00x10-3 kg of oxygen gas at two different temperatures. What is the value of PV/T where the curves meet on the y-axis?
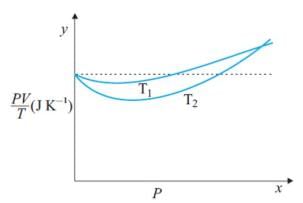

Detailed Solution: Question 22
Identify the alcohol or phenol from the following which is most soluble in water.
Detailed Solution: Question 23
The sound waves which travel in the air are called:
Detailed Solution: Question 24
The magnitude of a physical quantity is 8.5 Ns. The physical quantity is :
Detailed Solution: Question 25
The fertility of soil starts getting badly impacted when pH of soil exceeds:
Detailed Solution: Question 26
The first use of quantum theory to explain the structure of atom was made by –
Detailed Solution: Question 27
Detailed Solution: Question 28
The Indian Parliament passed a Central Legislation named Air Pollution Control Act in the year:
Detailed Solution: Question 29
The correct relation between current 'I' and amount of charge flown 'q' during a time 't' through a conductor is
Detailed Solution: Question 30



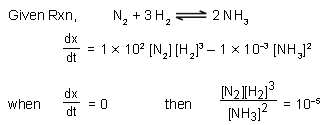

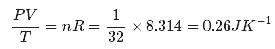 Hence the value of PV/T where the curves meet on the y-axis is 0.26 jK-1
Hence the value of PV/T where the curves meet on the y-axis is 0.26 jK-1