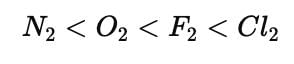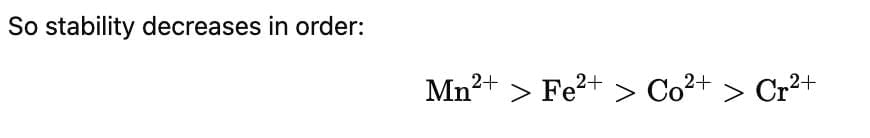Periodic Table & Trends - Free MCQ Practice Test with solutions, NEET Chemistry
MCQ Practice Test & Solutions: Test: Periodic Table & Trends (22 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Periodic Table & Trends". These 22 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 22 minutes
- - Number of Questions: 22
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
The elements charecterised by the filling of 4 f-orbitals, are:
Detailed Solution: Question 2
An element belongs to Group 15 and third period of the periodic table. Its electronic configuration will be
Detailed Solution: Question 3
It is now recognized that the ‘Modern Periodic Law’ is essentially the consequence of the
Detailed Solution: Question 4
Detailed Solution: Question 5
Among the alkali metals cesium is the most reactive because
Detailed Solution: Question 6
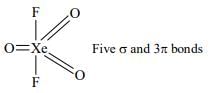
In which one of the given formulae of xenon compounds there are five σ−bonds and three π-bonds in it?
Detailed Solution: Question 7
Detailed Solution: Question 8
Detailed Solution: Question 9
Detailed Solution: Question 10
For alkali metals, which one of the following trends is INCORRECT?
Detailed Solution: Question 11
Dmitri Mendeleev (1834-1907) and the German chemist, Lothar Meyer (1830-1895) proposed arranging elements in
Detailed Solution: Question 12
According to the recommendation of International Union of Pure and Applied Chemistry (IUPAC), the groups in the modern periodic table are numbered from
Detailed Solution: Question 13
Ionization enthalpy increases across a period because
Detailed Solution: Question 14
Choose one of the following in th increasing order of bond length
Detailed Solution: Question 15
For the four successive transition elements (Cr, Mn, Fe and Co), the stability of + 2 oxidation state will be there in which of the following order? (At. nos. Cr = 24, Mn = 25, Fe = 26, Co = 27)
Detailed Solution: Question 16
A transition metal ion exists in its highest oxidation state. It is expected to behave as
Detailed Solution: Question 17
The sequence of ionic mobility in aqueous solution is
Detailed Solution: Question 18
Detailed Solution: Question 19
Generally, the first ionization energy increases along a period. But there are some exceptions. One which is NOT an exception is ______.
Detailed Solution: Question 20
Which of the following will have the most negative electron gain enthalpy and which one the least negative? P, S, Cl, F.
Detailed Solution: Question 21
For Atomic numbers greater than 100 IUPAC has made recommendation that until a new element ‘s discovery is proved, and its name is officially recognized, a systematic nomenclature be derived directly from the atomic number of the element using the numerical roots for 0 and numbers 1-9. The name 'sept' in this scheme corresponds to the digit
Detailed Solution: Question 22
96 videos|243 docs|71 tests |