Hydrogen (Old NCERT) - Free MCQ Practice Test with solutions, JEE Chapter
MCQ Practice Test & Solutions: Test: Hydrogen (Old NCERT) (25 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Hydrogen (Old NCERT)". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
In the earth’s atmosphere, hydrogen exists in the form of
Detailed Solution: Question 1
presence of extensive hydrogen bonding between water molecules leads to
Detailed Solution: Question 2
Temporary hardness It can be removed in boiling by precipitating
Detailed Solution: Question 3
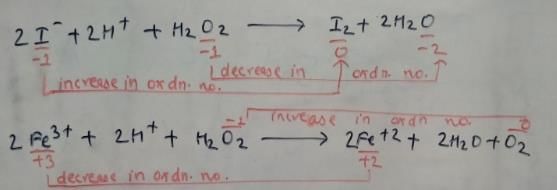
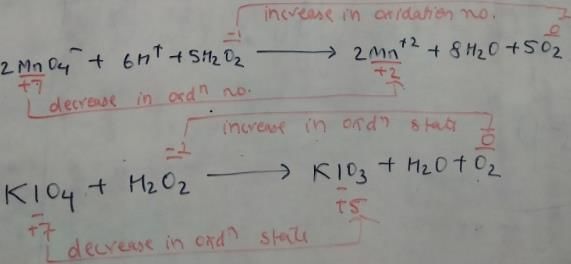
Which of the following equations depict the oxidising nature of H2O2?
Detailed Solution: Question 4
Detailed Solution: Question 5
Detailed Solution: Question 6
For moderation of the climate and body temperature of living beings, the responsible factor is:
Detailed Solution: Question 7
In Calgon’s method, one of the following chemical is used to remove hardness of water
Detailed Solution: Question 8
Which of the following equation depicts reducing nature of H2O2?
Detailed Solution: Question 9
Detailed Solution: Question 10
Approximately what percent of matter in the universe is believed to consist of hydrogen?
Detailed Solution: Question 11
In the gas phase water is a bent molecule with a bond angle of
Detailed Solution: Question 12
The three isotopes of hydrogen in terms of chemical properties show one of the following properties
Detailed Solution: Question 13
Detailed Solution: Question 14
Stoichiometric compounds of dihydrogen are formed with
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
Consider the reactions
(A) H2O2 + 2HI → I2 +2H2O
(B) HOCl + H2O2 → H3O + Cl - - + O2
Which of the following statements is correct about H2O2 with reference to these reactions? Hydrogen perioxide is ________.
(B) HOCl + H2O2 → H3O + Cl - - + O2
Detailed Solution: Question 18
Which of the following statements are not true for hydrogen?
Detailed Solution: Question 19
Detailed Solution: Question 20
Dihydrogen under certain reaction conditions, combines with almost all elements to form binary compounds, called hydrides except with few which are given below Choose one of the options
Detailed Solution: Question 21
How many hydrogen-bonded water molecule(s) are associated in CuSO4 . 5H2O ?
Detailed Solution: Question 22
Detailed Solution: Question 23
Dihydrogen can be prepared on commercial scale by different methods. In its preparation by the action of steam on hydrocarbons, a mixture of CO and H2 gas is formed. It is known as ____________.
Detailed Solution: Question 24
Atoms like N, O and F in hydrides have lower boiling points than those of the subsequent group member hydrides. It is because of
Detailed Solution: Question 25
446 docs|929 tests |