Chemical Reactions And Equations - MCQ Test, Class 10 Science Solved MCQs
MCQ Practice Test & Solutions: Chemical Reactions And Equations - MCQ Test, Class 10 Science (30 Questions)
You can prepare effectively for Class 10 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Chemical Reactions And Equations - MCQ Test, Class 10 Science". These 30 questions have been designed by the experts with the latest curriculum of Class 10 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following is not a decomposition reacion?
Detailed Solution: Question 1
Which two gases are released on heating lead nitrate?
Detailed Solution: Question 2
What happens when Hydrogen combines with oxygen in the presence of an electric current?
Detailed Solution: Question 3
Match the following with correct response.
(1) Oxidation (A) Gain of electron
(2) Reduction (B) Electron accepting species
(3) Reducing agent (C) Electron losing species
(4) Oxidising agent (D) Loss of electron
Detailed Solution: Question 4
Which of the following represent a double displacement reaction?
Detailed Solution: Question 5
Some crystals of copper sulphate were dissolved in water. The colour of the solution obtained would be
Detailed Solution: Question 6
Mg ribbon burns with a dazzling flame in air (oxygen) and changes into a white substance, Magnesium oxide. Magnesium is
Detailed Solution: Question 7
What happens when dilute HCl is added to iron fillings? Select the correct answer.
Detailed Solution: Question 8
Match the following with correct response.
(1) Chemical change (2) Physical change (3) Exothermic (4) Endothermic
(A) Energy releasing (B) Rusting of iron (C) Energy absorbed (D) Dissolution of sugar or salt in water
Detailed Solution: Question 9
Which statement is correct about the following reaction?
ZnO + CO 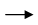 Zn + CO2
Zn + CO2
Detailed Solution: Question 10
The colour of zinc metal is
Detailed Solution: Question 11
The reactions in which more reactive element can displace less reactive element from a compound are called
Detailed Solution: Question 12
Colour of copper sulphate solution changes when an iron nail is dipped in it because of
Detailed Solution: Question 13
Match the following with correct response.
(1) Gain of hydrogen (2) Loss of hydrogen (3) Unpleasant taste and foul small in fatty food (4) Eating away of metal in the presence of air and moisture
(A) Reduction (B) Rancidity (C) Oxidation (D) Corrosion
Detailed Solution: Question 14
Fe2O3 + 2Al 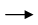 Al2O3 + 2Fe This reaction is an example of –
Al2O3 + 2Fe This reaction is an example of –
Detailed Solution: Question 15
Which of the following is a decomposition reaction?
Detailed Solution: Question 16
Which one of the following will be required to identify the gas evolved when diluted HCl reacts with zinc metal?
Detailed Solution: Question 17
Decomposition of ferrous sulphate into Fe2O3,SO2 and SO3 occurs in the presence of
Detailed Solution: Question 18
What happens when copper metal is added to silver nitrate solution?
Detailed Solution: Question 19
Match the following with correct response.
(1) Reaction in which two substances combine to form new product
(2) substance split to form two or more simple substance
(3) More reactive metal displaces less reactive metal
(4) Loss or gain of electrons occurs
(A) Displacement reaction
(B) Combination Reaction
(C) Redox reaction
(D) Decomposition
(D) Decomposition
Detailed Solution: Question 20
When Iron nails are added to an aquous solution of copper sulphate, a chemical change occurs, which of the following is not true about this reaction?
Detailed Solution: Question 21
Which of the following reactions is a metathesis (double displacement) reaction?
Detailed Solution: Question 22
Which of the following is not a balanced chemical equation?
Detailed Solution: Question 23
The compound formed by Hg+2 and CI- is
Detailed Solution: Question 24
Which gas is produced when calcium carbonate reacts with dilute HCl?
Detailed Solution: Question 25
Match the following with correct response.
(1) Quick lime is added to water (2) Formula for rust (3) Silver tarnishing (4) Pop sound
(A) Ca(OH)2 (B) H2S gas (C) Fe2O3.xH2O (D) H2 gas
Detailed Solution: Question 26
Pieces of Zn metal are added to four different test tubes containing different solutions. In which test tube no change is observed?
Detailed Solution: Question 27
Can we store CuSO4 in iron container and why?
Detailed Solution: Question 28
H2S burns in air to give
Detailed Solution: Question 29
Which of the following statements about the given reaction are correct?
3Fe (s) + 4H2O (g) → Fe3O4 (s) + 4 H2 (g)
(i) Iron metal is getting oxidised
(ii) Water is getting reduced
(iii) Water is acting as reducing agent
(iv) Water is acting as oxidising agent
3Fe (s) + 4H2O (g) → Fe3O4 (s) + 4 H2 (g)
(i) Iron metal is getting oxidised
(ii) Water is getting reduced
(iii) Water is acting as reducing agent
(iv) Water is acting as oxidising agent
Detailed Solution: Question 30