Mechanical Engineering Mock Test: SSC JE (ME)- 1 Free Online Test 2026
Full Mock Test & Solutions: Mock Test: SSC JE (ME)- 1 (100 Questions)
You can boost your Mechanical Engineering 2026 exam preparation with this Mock Test: SSC JE (ME)- 1 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of Mechanical Engineering 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Total Questions: 100
- - Analysis: Detailed Solutions & Performance Insights
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
An ideal gas at 27ºC is heated at constant pressure till its volume becomes three times. What would be then the temperature of gas?
Detailed Solution: Question 1
Measurement of temperature is based on which law of thermo dynamics?
Detailed Solution: Question 2
Hot coffee stored in a well insulated thermos flask is an example of
Detailed Solution: Question 3
Which one of the following expresses the reversible work done by the system (steady flow)
between states 1 and 2?
between states 1 and 2?
A closed system receives 60 kJ heat but its internal energy decreases by 30 kJ. Then the work done by the system is
Detailed Solution: Question 5
Detailed Solution: Question 6
Detailed Solution: Question 8
The temperature at which the volume of a gas becomes zero is called
Detailed Solution: Question 9
The molecular mass expressed in gram (i.e. 1 gmole) of all gases, at N. T. P., occupies a volume of
Detailed Solution: Question 10
The sum of internal energy (U) and the product of pressure and volume (p.v) is known as
Detailed Solution: Question 11
Detailed Solution: Question 12
The C.O.P. of a refrigerator working on a reversed Carnot cycle is where
T1 = Lowest absolute temperature, and
T2 = Highest absolute temperature
T1 = Lowest absolute temperature, and
T2 = Highest absolute temperature
The leakage in a refrigeration system using freon is detected by
Detailed Solution: Question 15
The COP of a heat pump βhp and the COP of a refrigerator βRef are related as
Detailed Solution: Question 17
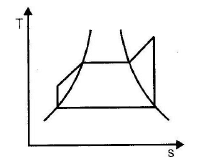
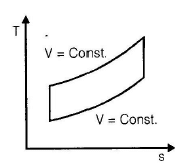
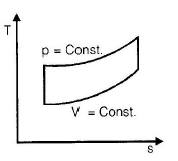
The correct sequence of the given four cycles on T-s plane in figure 1, 2, 3, 4 is
(1)
(2) 
(3) 
(4) 
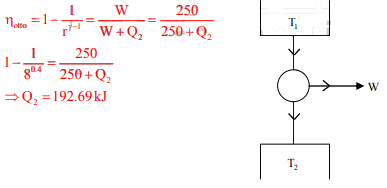
An Otto cycle has a compression ratio of 8. If 250 kJ of work is extracted from the cycle, the heat rejected by the cycle is
Detailed Solution: Question 19
Detailed Solution: Question 20
The stoichiometric air-fuel ratio for petrol is 15 :1. What is the air-fuel ratio regarded for maximum power ?
Detailed Solution: Question 21
Match List-I (SI Engine Operating Mode) with List –II (Appromimate A/F Ratio) and select the correct answer using the code given below the lists
List - I (SI Engine Operational Mode)
A. Idling
B. Cruising
C. Maximum Power
D. Cold starting
List-II (A/F Ratio Supplied by the Carburetor)
1. 3
2. 10
3. 13
4. 16
5. 20
Detailed Solution: Question 22
Detailed Solution: Question 23
Detailed Solution: Question 24
Detailed Solution: Question 25
Match List-I (Type of boiler) with List-II (Classification of boiler) select the correct answer
using the codes given below the lists:

using the codes given below the lists:
Benson boiler is one of the high pressure boilers having
5 videos|116 docs|59 tests |