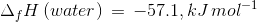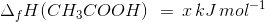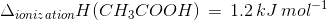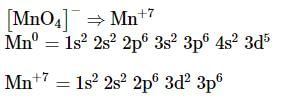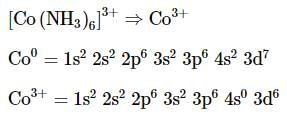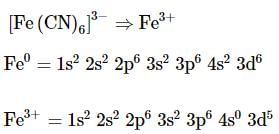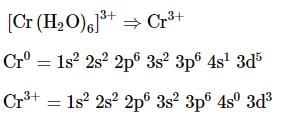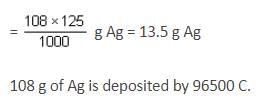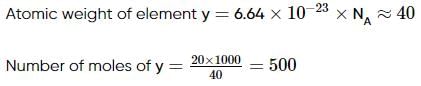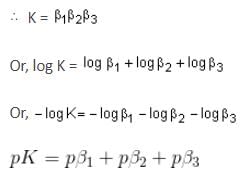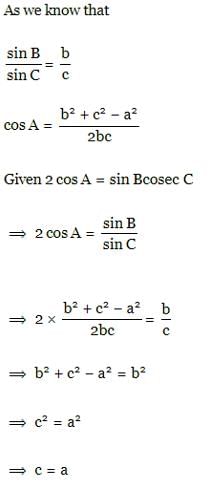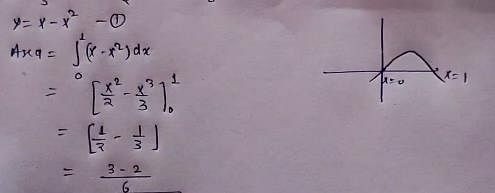AP EAMCET Mock Test - 3 Free Online Test 2026
Full Mock Test & Solutions: AP EAMCET Mock Test - 3 (160 Questions)
You can boost your JEE 2026 exam preparation with this AP EAMCET Mock Test - 3 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of JEE 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 180 minutes
- - Total Questions: 160
- - Analysis: Detailed Solutions & Performance Insights
- - Sections covered: Physics, Chemistry, Mathematics
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
Detailed Solution: Question 1
An alternating voltage is given by 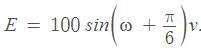 The voltage will be maximum for the first time when is (T = periodic time)
The voltage will be maximum for the first time when is (T = periodic time)
Detailed Solution: Question 2
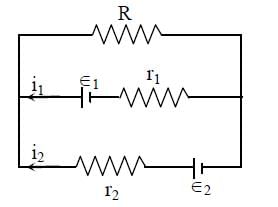
In the given electrical circuit, which one of the following equations is a correct equation?

Detailed Solution: Question 3
A particle is performing U.C.M. along the circumference of a circle of diameter 50 cm with frequency 2 Hz, The acceleration of the particle tn m/s2 is
Detailed Solution: Question 4
A hole is drilled half way to the centre of the earth. A body weighs 300 N on the surface of the earth. How much wil, it weigh at the bottom of the hole?
Detailed Solution: Question 5
The total energy of a simple harmonic oscillator is proportional to
Detailed Solution: Question 6
A lift is tied with thick iron ropes having mass ‘M’. The maximum acceleration of the lit is ‘a’ m/s2 and the maximum safe stress is ‘S’ N/m2. The minimum diameter of the rope is
Detailed Solution: Question 7
A magnetizing field of 5000 A/m produces a magnetic flux of 4 x 10-5 Wb in an iron rod of cross-sectional area 0.4 cm2. The permeability of the rod in Wb/A-m, is
Detailed Solution: Question 8
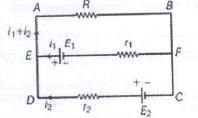
In the network shown cell E has internal resistance r and the galvanometer shows zero deflection. If the cell is replaced by a new cell of 2E and internal resistance 3r keeping ling everything else identical, then
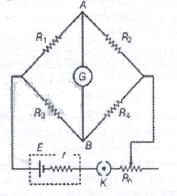

Detailed Solution: Question 9
A convex lens of focal length ‘f’ is placed In contact with a concave lens of the same focal length. The equivalent focal fengfo of the combination is
Detailed Solution: Question 10
Directions: The following question has four choices out of which ONLY ONE is correct.
The heat of neutralisation of acetic acid by sodium hydroxide is −55.9 kJ/mol, and the heat of neutralisation of a strong acid by a strong base is −57.1 kJ/mol. What is the value of ΔH for the ionisation of CH3COOH?
The heat of neutralisation of acetic acid by sodium hydroxide is −55.9 kJ/mol, and the heat of neutralisation of a strong acid by a strong base is −57.1 kJ/mol. What is the value of ΔH for the ionisation of CH3COOH?
Detailed Solution: Question 11
Two open beakers one containing a solvent and the other containing a mixture of that solvent with a non volatile solute are together sealed in a container. Over time:
Detailed Solution: Question 12
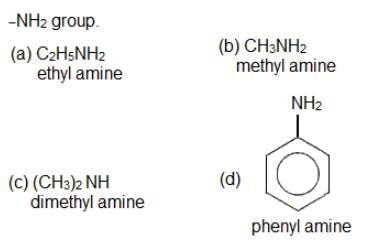
Which of the following amines will not undergo carbylamine reaction?
Detailed Solution: Question 13
The complex ion without 'd' electron in the central metal atom is which of the following?
Detailed Solution: Question 14
What is the time (in sec) required for depositing all the silver present in 125 mL of 1 M AgNO3 solution by passing a current of 241.25 A?
(1F = 96500 coulombs)
(1F = 96500 coulombs)
Detailed Solution: Question 15
One atom of an element y weights 6.64 x 10-23 g. Then, find the numbers of moles of atom in 20 kg
Detailed Solution: Question 16
Directions: Consider the following equilibrium equations (omitting charges).
I. M + Cl → MCl, Keq = β1
II. MCl + Cl → MCl2, Keq = β2
III. MCl2 + Cl → MCl3, Keq = β3
IV. M + 3Cl → MCl3, Keq = K
The relationship(s) between K,β1 ,β2 and β3 is/are
II. MCl + Cl → MCl2, Keq = β2
III. MCl2 + Cl → MCl3, Keq = β3
IV. M + 3Cl → MCl3, Keq = K
Detailed Solution: Question 17
A and B are square matrices of the same order. Which is correct?
In four schools B₁, B₂, B₃, B₄ the percentage of girls students is 12, 20, 13, 17 respectively. From a school selected at random, one student is picked up at random and it is found that the student is a girl. The probability that the school selected is B₂, is
The projection of the vector î - 2 ĵ + k̂ on the vector 4 î - 4 ĵ + 7 k̂ is
In two events P(A∪B) = 5/6, P(A) = 5/6, P(B) = 2/3 then A and B are
The ratio in which the plane r̅.(i̅−2j̅+3k̅)=17 divides the line joining the points -2i̅+4j̅+7k̅ and 3i̅+5j̅+8k is̅
If α , β are two different complex numbers such that | α | = 1, | β | = 1, then the expression | β − α /1 − α β | equals
Detailed Solution: Question 25
The order of differential equation (d2y/dx2)2 = (1+dy/dx)1/2 is
The area of the region bounded by the curve y = x - x2 between x = 0 and x = 1 is
Detailed Solution: Question 29



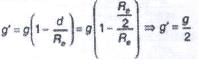
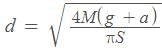
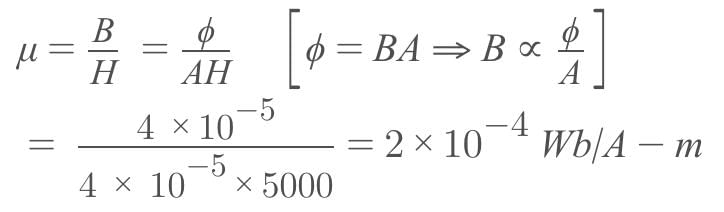
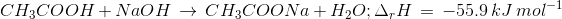
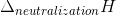 for a strong acid and a strong base =
for a strong acid and a strong base =