EMRS PGT Chemistry Mock Test - 6 Free Online Test 2026
Full Mock Test & Solutions: EMRS PGT Chemistry Mock Test - 6 (150 Questions)
You can boost your EMRS 2026 exam preparation with this EMRS PGT Chemistry Mock Test - 6 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of EMRS 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 180 minutes
- - Total Questions: 150
- - Analysis: Detailed Solutions & Performance Insights
- - Sections covered: Part 1: General Awareness, Part 2: Reasoning Ability, Part 3: Knowledge of ICT, Part 4: Teaching Aptitude, Part 5: Domain Knowledge, Part 6: Language Competency
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
Detailed Solution: Question 1
Direction: Read the following information carefully and answer the questions that follow.
A blacksmith has five iron articles A, B, C, D and E each having a different weight.
I. A weight is twice as much as of B.
II. B weight is four and half times as much as of C.
III. C weight is half times as much as of D.
IV. D weight is half as much as of E.
V. E weight is less than A but more than C.
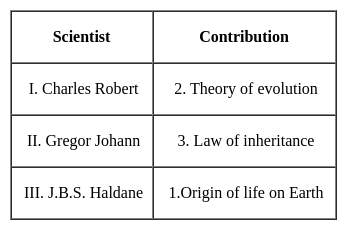
Q. E is lighter in weight than which of the other two articles?
A blacksmith has five iron articles A, B, C, D and E each having a different weight.
I. A weight is twice as much as of B.
II. B weight is four and half times as much as of C.
III. C weight is half times as much as of D.
IV. D weight is half as much as of E.
V. E weight is less than A but more than C.
Detailed Solution: Question 2
Detailed Solution: Question 3
Detailed Solution: Question 4
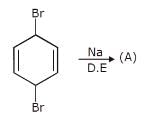
Which is the least reactive halide in both E1 and SN1 reaction?
Detailed Solution: Question 5
When K2O is added to water, the solution becomes basic in nature because it contains a significant concentration of -
Detailed Solution: Question 6
Detailed Solution: Question 7
Detailed Solution: Question 8
Electronic configuration of a transition element X in +3 oxidation state is [Ar] 3d5 and Y in +2 state is [Ar] 3d8. What are the atom ic numbers of the elements?
Detailed Solution: Question 9
[Ti (H2O)6]3+ absorbs green and yellow region part of visible light. Then the transmitted colour of the compound is
Detailed Solution: Question 10
Detailed Solution: Question 11
90% of hydrogen peroxide is used as fuel in ______________
Detailed Solution: Question 12
The most and least reactive electrophiles respectively in a SN1 reaction are


Detailed Solution: Question 13
Presence of particulate matter in polluted air catalyses the oxidation of sulphur dioxide to:
Detailed Solution: Question 14
Which of the following forms a colloidal solution in water?
Detailed Solution: Question 15
Direction:
This section contains 5 questions. When worked out will result in an integer from 0 to 9 {both inclusive).
Q.
Relative decrease in vapour pressure of an aqueous NaCI solution is 0.167. Thus, number of moles of NaCI present in 180 g of H20 is ... .
Detailed Solution: Question 16
A mixture of ethyl alcohol and propyl alcohol has a vapour pressure of 290 mm at 300 K. The vapour pressure of propyl alcohol is 200 mm. If the mole fraction of ethyl alcohol is 0.6, its vapour pressure (in mm) at the same temperature will be -
[AIEEE 2007]
Detailed Solution: Question 17
Elements of which group are known as ore forming elements?
Detailed Solution: Question 18
Detailed Solution: Question 19
Detailed Solution: Question 20
Detailed Solution: Question 21
What kind of support can a school provide to address the individual differences in students?
Detailed Solution: Question 22
Which of the following is an example of scaffolding?
Detailed Solution: Question 23
____is a mental health condition that involves extreme mood swings, including both manic and depressive episodes.
Detailed Solution: Question 24
According to NEP‐2020, National Research Foundation (NRF) may be established to
Detailed Solution: Question 25
In the following question, out of the four alternatives, select the alternative which is the best substitute for the phrase.
Q. A story that can be interpreted to reveal a hidden meaning.
Detailed Solution: Question 26
In the following question, four words are given out of which one word is incorrectly spelled. Find the incorrectly spelled word.
Detailed Solution: Question 27
In the following question, an idiomatic expression and its four possible meanings are given. Find out the correct meaning of the idiom.
Giving a slap on the wrist.
Detailed Solution: Question 28
इनमें से कौन-सा व्यंजन 'संयुक्त व्यंजन' का उदाहरण है?
Detailed Solution: Question 29
Detailed Solution: Question 30
8 docs|45 tests |