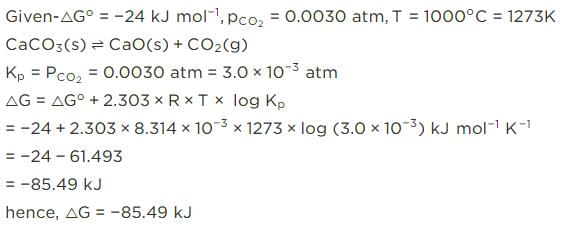EMRS PGT Chemistry Mock Test - 8 Free Online Test 2026
Full Mock Test & Solutions: EMRS PGT Chemistry Mock Test - 8 (150 Questions)
You can boost your EMRS 2026 exam preparation with this EMRS PGT Chemistry Mock Test - 8 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of EMRS 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 180 minutes
- - Total Questions: 150
- - Analysis: Detailed Solutions & Performance Insights
- - Sections covered: Part 1: General Awareness, Part 2: Reasoning Ability, Part 3: Knowledge of ICT, Part 4: Teaching Aptitude, Part 5: Domain Knowledge, Part 6: Language Competency
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
Detailed Solution: Question 1
The new agricultural strategy called 'Green Revolution' was initiated in ________.
Detailed Solution: Question 2
Which of the following is not a department under Ministry of Finance?
Detailed Solution: Question 3
The average pattern of wind speed, temperature, rainfall etc., in place over a long period of time is called ___________.
Detailed Solution: Question 4
From the given alternatives, select the word which CANNOT be formed using the letters of the given word.
FEARLESS
Detailed Solution: Question 5
Which one of the answer figures shall complete the given question figure?
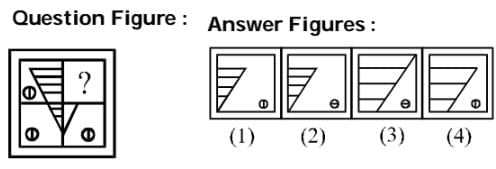

Detailed Solution: Question 6
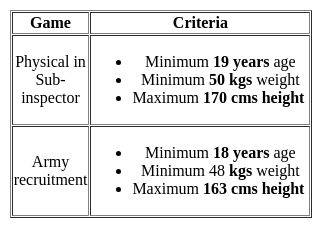
To qualify for Physical in Sub-inspector and Army recruitment, the minimum age required is 19 years and 18 years respectively, the minimum weight required is 50 kgs and 48 kgs respectively, and maximum height allowed is 170 cms and 163 cms respectively. Karan is 17 years old, has a weight of 47 kgs, and has a height of 161 cms. For which of this recruitment is he eligible?
Detailed Solution: Question 7
Picture formats can be recognized by which extensions?
What are the steps taken to open an existing document?
Detailed Solution: Question 9
Which of the following is the most important factor underlying the success of a new teacher?
Detailed Solution: Question 10
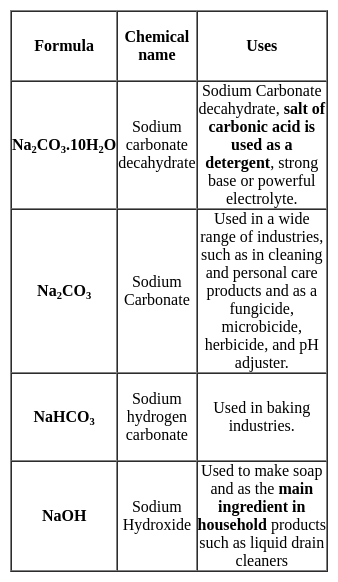
In which of the following pairs both the ions are coloured in aqueous solution? (Atomic number, Sc = 21, Ti = 22, Ni = 28, Cu = 29, Co = 27)
Detailed Solution: Question 11
Direction (Q. Nos. 17-20) This section contains 4 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
Q. Which of the following can be calculated based on Born-Haber cycle of formation of a lattice A+B- (s) from A(s) and 6 (g) ?
Detailed Solution: Question 12
For the reaction, at 1000° C
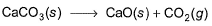
ΔG° = - 24 kJ mol-1, 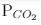 = 0.0030 atm.
= 0.0030 atm.
Q. Hence, ΔG at this temperature is
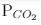 = 0.0030 atm.
= 0.0030 atm.Detailed Solution: Question 13
Detailed Solution: Question 14
Detailed Solution: Question 15
It is observed that the voltage of a galvanic cell using the reaction M(s) + xH+→ Mx+ + X/2H2 varies linearly with the log of the square root of the hydrogen pressure and the cube root of the Mx+ concentration. The value of x is
Detailed Solution: Question 16
The alkali metals are low melting. Which of the following alkali metal is expected to melt if the room temperature rises to 30°C?
Detailed Solution: Question 17
In an atom, an electron is moving with a speed of 600 m/s with an accuracy of 0.005%. Certainity with which the position of the electron can be located is (h = 6.6×10-34 kg m2s-1 ,mass of electron, em = 9.1×10-31 kg ) [AIEEE 2009]
Detailed Solution: Question 18
A metal M readily forms water soluble sulphate, and water insoluble hydroxide M(OH)2. Its oxide MO is amphoteric, hard and having high melting point. The alkaline earth metal M must be -
Detailed Solution: Question 19
Comprehension Type
Direction (Q. Nos. 16-18) This section contains a paragraph, describing theory, experiments, data, etc.
Three questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).
Passage
Consider the following sequence of reaction,
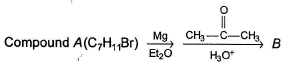
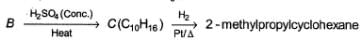
Q.
The structure of compound B is
Three questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).
Detailed Solution: Question 20
In Calgon’s method, one of the following chemical is used to remove hardness of water
Detailed Solution: Question 21
Detailed Solution: Question 22
Which one of the following exists in the oxidation state other than +3?
Detailed Solution: Question 23
H2 gas is adsorbed on the metal surface like tungsten. This follows........ order reaction –
[AIEEE-2002]
Detailed Solution: Question 24
Consider the following reactions,
I. Zn + dil. H2SO4 → ZnSO4 + H2
II. Zn + conc. H2SO4 → ZnSO4+ SO2 + H2O
Oxidising agents in I and II are
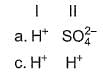
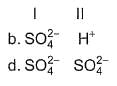
II. Zn + conc. H2SO4 → ZnSO4+ SO2 + H2O
Oxidising agents in I and II are
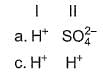

Detailed Solution: Question 25
Directions: In the following question, one part of the sentence may have error(s). Find out the part of the sentence having an error and select the appropriate option. If a sentence is free from error, select 'No error' as your answer.
Q. The rich man (1)/ killed him (2)/ and his own children (3)/ No error (4)
Detailed Solution: Question 26
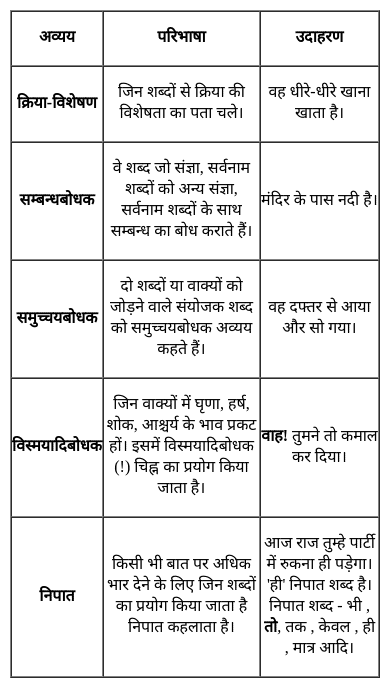
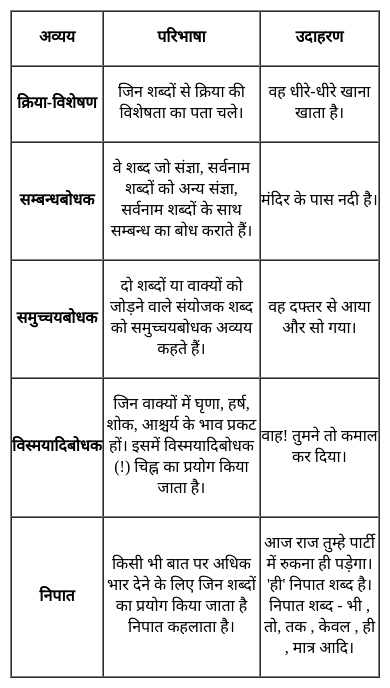
निम्नांकित में 'विस्मयादिबोधक शब्द' का उदाहरण है -
Detailed Solution: Question 27
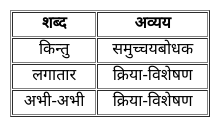
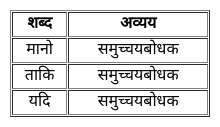
निम्नांकित में से कौन-सा 'समुच्चयबोधक शब्द' नहीं है?
Detailed Solution: Question 28
वह आज तक परीक्षा में सफल नहीं हुआ।- वाक्य के रेखांकित शब्द में निपात है-
Detailed Solution: Question 29
Detailed Solution: Question 30
8 docs|45 tests |