Introduction to Transition Metals - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Introduction to Transition Metals (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Introduction to Transition Metals". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Outof [TiF6]2−, [CoF6]3−, Cu2Cl2 and [NiCl4]2− (Z of Ti = 22, Co = 27, Cu = 29, Ni = 28), the colourless species are:
Detailed Solution: Question 1
The basic character of the transition metal monoxides follows the order (Atomic Nos., Ti = 22, V = 23, Cr = 24, Fe = 26)
Detailed Solution: Question 2
Which of the following statement is incorrect?
Detailed Solution: Question 3
On the basis of data given below, 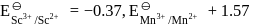
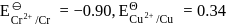
Which of the following statements is incorrect?
Detailed Solution: Question 4
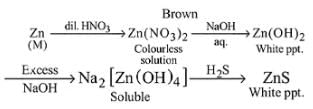
A metal M and its compound can give the following observable changes in a consequence of reactions
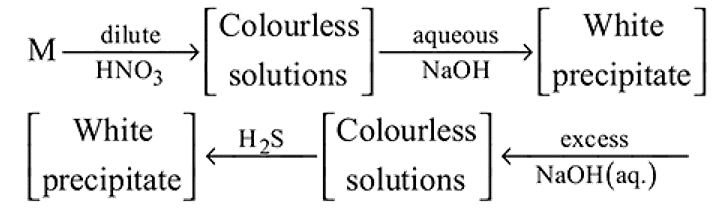

Detailed Solution: Question 5
Detailed Solution: Question 6
The blue complex formed on addition of conc. 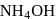 solution to a
solution to a 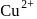 salt solution has the structure?
salt solution has the structure?
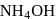 solution to a
solution to a 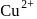 salt solution has the structure?
salt solution has the structure?Detailed Solution: Question 7
When 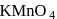 is added to oxalic acid, the decolourisation is slow in the beginning but becomes instantaneous after sometime because
is added to oxalic acid, the decolourisation is slow in the beginning but becomes instantaneous after sometime because
Detailed Solution: Question 8
Number of electrons transfered in each case when 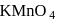 acts as an oxidising agent to give
acts as an oxidising agent to give 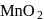 ,
, 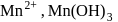 and
and 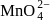 are respectively
are respectively
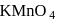 acts as an oxidising agent to give
acts as an oxidising agent to give 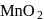 ,
, 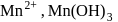 and
and 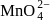 are respectively
are respectivelyDetailed Solution: Question 9
Chloro compound of Vanadium has only spin magnetic moment of 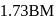 . This Vanadium chloride has the formula:
. This Vanadium chloride has the formula:
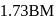 . This Vanadium chloride has the formula:
. This Vanadium chloride has the formula:Detailed Solution: Question 10
Which of the following compounds has colour but no unpaired electrons?
Detailed Solution: Question 11
Which element of the 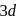 -series has highest third ionisation enthalpy?
-series has highest third ionisation enthalpy?
Detailed Solution: Question 12
In which of the tollowing ions, d-d transition is not possible ?
Detailed Solution: Question 13
Whch of the following pairs involves isoelectronic ions
Detailed Solution: Question 14
In a reaction the ferrous (Fe+2) ion is oxidised to ferric (Fe+3) ion. The equivalent weight of the ion in the above reaction is equal to
Detailed Solution: Question 15
332 videos|699 docs|300 tests |


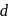 -orbitals which permits the
-orbitals which permits the 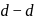 excitation of electrons.
excitation of electrons.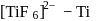 is in
is in 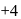 O.S.
O.S. 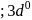 -colourless
-colourless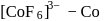 is in
is in 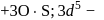 coloured
coloured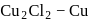 is in
is in 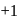 O.S.
O.S. 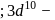 colourless
colourless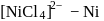 is in
is in 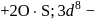 coloured
coloured
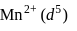 is more stable than
is more stable than 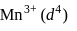 , thus
, thus 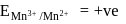
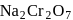 is hygroscopic.
is hygroscopic.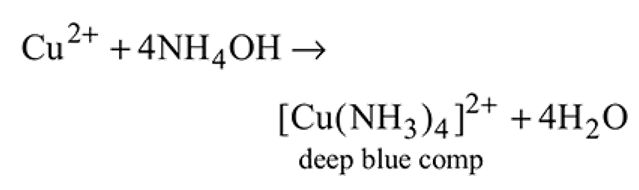
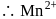 acts as autocatalyst.
acts as autocatalyst.
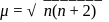
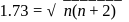
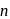 we find
we find 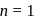 No. of unpaired electrons
No. of unpaired electrons 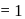 hence its configuration will be
hence its configuration will be 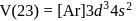
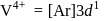
 Its chloride has the formula
Its chloride has the formula 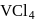
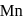 is
is 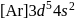 . Being transition metal it has 7 valence electrons and all are involved in bond formation in
. Being transition metal it has 7 valence electrons and all are involved in bond formation in 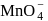 . Hence it has no unpaired electron.
. Hence it has no unpaired electron.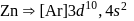
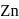 means removal of electron from the stable configuration of
means removal of electron from the stable configuration of 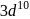 .
.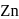 . Hence, the correct option is (b).
. Hence, the correct option is (b).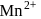 and
and 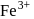 involve isoelectronic ions.
involve isoelectronic ions.
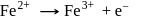
 Equivalent weight
Equivalent weight  Atomic weight
Atomic weight