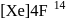F-Block elements - Free MCQ Practice Test with solutions, JEE Chemistry
MCQ Practice Test & Solutions: Test: F-Block elements (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: F-Block elements". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
Statement I : Many trivalent lanthanide ions are coloured both in solid state and in aqueous solution.
Statement II : Colour of these ions is due to the presence of f-electrons.
Detailed Solution: Question 2
The number f-electrons in +3 oxidation state of gadolinium 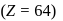 is
is  and in +2 oxidation state of Ytterbium
and in +2 oxidation state of Ytterbium 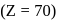 is
is 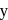 . The sum of
. The sum of 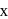 and
and 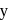 is
is
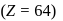 is
is 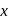 and in +2 oxidation state of Ytterbium
and in +2 oxidation state of Ytterbium 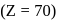 is
is 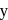 . The sum of
. The sum of 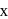 and
and 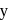 is
isDetailed Solution: Question 3
Detailed Solution: Question 4
Which one of the following elements shows maximum number of different oxidation states in its compounds?
Detailed Solution: Question 5
Detailed Solution: Question 6
Which of the following pairs is expected to form colourless compound?
Detailed Solution: Question 7
Gadolinium (atomic number = 64) is a member of 4f series. It's electronic configuration in +3 oxidation state is [Xe] 4f7. What is the ground state electronic configuration of gadolinium?
Detailed Solution: Question 8
Which of the following trivalent ion has the largest atomic radii in the lanthanide series
Detailed Solution: Question 9
Detailed Solution: Question 10
Which element among the lanthandes has the smallest atomic radius?
Detailed Solution: Question 11
Detailed Solution: Question 12
Which of the following species has maximum tendency to form complexes?
Detailed Solution: Question 13
From the list given below, the number of lanthanides which exhibit +4 state in their oxides is Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy
Detailed Solution: Question 14
Detailed Solution: Question 15
333 videos|699 docs|300 tests |


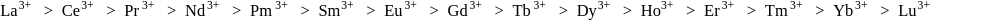
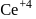 is less stable than
is less stable than 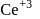 and thus it will readily accept
and thus it will readily accept 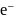 convert into
convert into 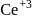 and so it will less basic than
and so it will less basic than 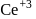 .
.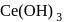 is most basic among all given options.
is most basic among all given options.
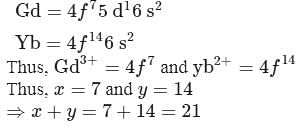
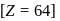 is
is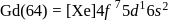
 No. of unpaired electrons
No. of unpaired electrons 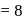
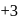 , oxidation state, while Eu shows oxidation state of
, oxidation state, while Eu shows oxidation state of 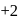 and
and 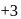 . Am shows
. Am shows 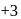 ,
, 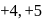 and
and 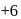 oxidation states. Therefore Americium (Am) has maximum number of oxidation states.
oxidation states. Therefore Americium (Am) has maximum number of oxidation states.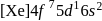
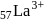 is (4f)" while that of
is (4f)" while that of 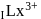 is
is 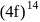 . They are expected to be colomicis.
. They are expected to be colomicis.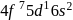
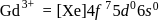
 block elements because
block elements because ". and deu to electronic configuration.
". and deu to electronic configuration. has tendency to go over to
has tendency to go over to 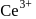 and hence acts as oxidiring agent.
and hence acts as oxidiring agent.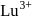 being smallest (due to lanthanide contraction) has maximum tendency to form complexes.
being smallest (due to lanthanide contraction) has maximum tendency to form complexes.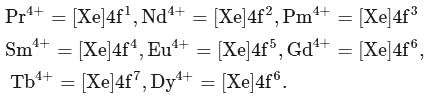
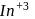 (trivalent lanthanides ions) have E.C.
(trivalent lanthanides ions) have E.C.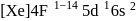
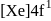 to
to