Crystalline & Amorphous Solids (Old NCERT) - Free MCQ Practice Test
MCQ Practice Test & Solutions: Test: Crystalline & Amorphous Solids (Old NCERT) (25 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Crystalline & Amorphous Solids (Old NCERT)". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
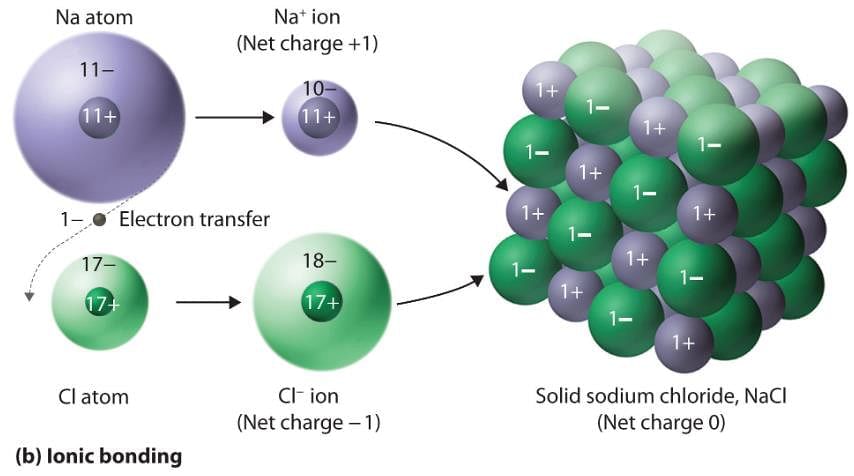
In which of the following solids, ions of opposite charges are held together by strong electrostatic forces of attraction?
Detailed Solution: Question 1
Which of the following crystalline solids have highest melting point?
Detailed Solution: Question 2
Amorphous solids are also known as:
Detailed Solution: Question 3
What are the basic particles of ice crystals?
Detailed Solution: Question 4
Why some of the physical properties of solids show different values when measured along different directions in the same crystals?
Detailed Solution: Question 5
Which of the following properties is generally exhibited by amorphous solids?
Detailed Solution: Question 6
Which of the following is NOT a molecular solid?
Detailed Solution: Question 7
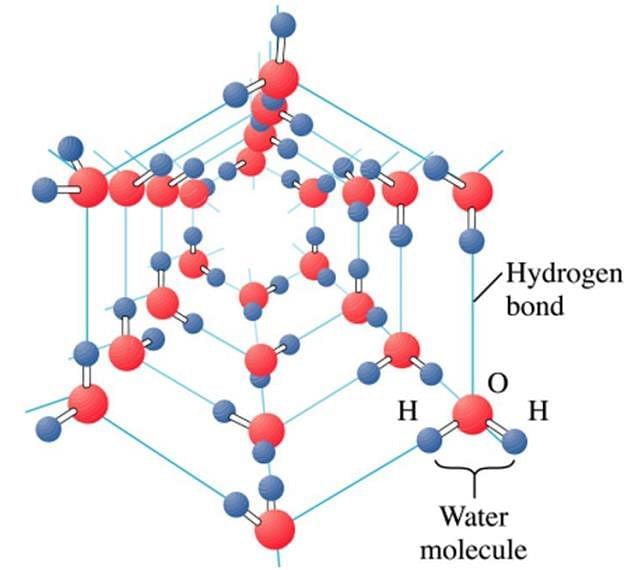
Hydrogen bonding occurs in which type of crystalline solids?
Detailed Solution: Question 8
Which of the following is an amorphous solid?
Detailed Solution: Question 9
Why ancient glass becomes milky?
Detailed Solution: Question 10
Which is not a characteristic of crystalline solids?
Detailed Solution: Question 11
Detailed Solution: Question 12
Which type of solids are held by weak dispersion forces?
Detailed Solution: Question 13
Silicon is found in nature in the forms of ________.
Detailed Solution: Question 14
Which state of matter is incompressible?
Detailed Solution: Question 15
Why do metallic solids conducts electricity?
Detailed Solution: Question 16
Detailed Solution: Question 17
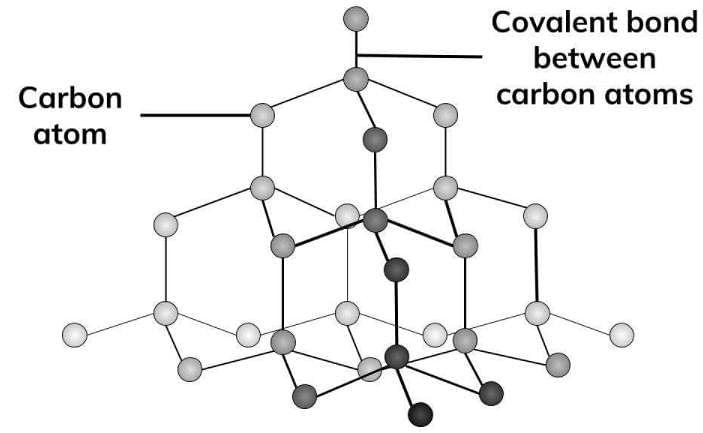
What are the basic constituent particles forming diamond crystals?
Detailed Solution: Question 18
A friend in your chemistry class is struggling to understand why crystalline solids are grouped into four main types: network, molecular, ionic, and metallic. Which explanation below will BEST help him begin to understand why chemists might have these groups?
Detailed Solution: Question 19
Which of the following is a non-conductor in solid state but good conductor in molten state?
Detailed Solution: Question 20
Which of the following properties is NOT exhibited by metallic solids?
Detailed Solution: Question 21
Among the following solids, which one shows the strongest bonding?
Detailed Solution: Question 22
Why ice is a crystalline compound?
Detailed Solution: Question 23
Which of the following is a covalent crystal?
Detailed Solution: Question 24
The points which shows the position of atoms in a crystal are called as _________.
Detailed Solution: Question 25
446 docs|929 tests |