Packing Efficiency (Old NCERT) - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Packing Efficiency (Old NCERT) (10 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Packing Efficiency (Old NCERT)". These 10 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
Packing efficiency of body centred cubic unit cell is:
Detailed Solution: Question 1
Packing efficiency in a unit cell is never 100% because constituent particles are assumed to be:
Detailed Solution: Question 2
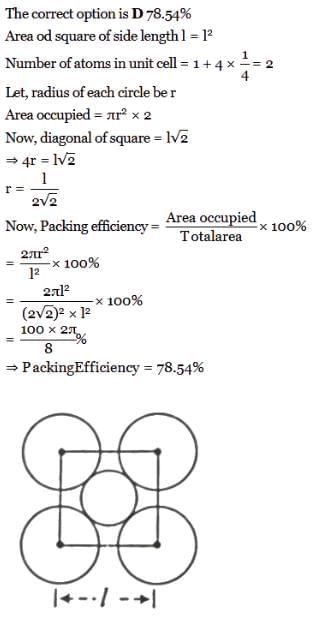
The packing efficiency of the two-dimensional square unit cell shown in the given figure is:
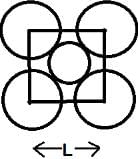

Detailed Solution: Question 3
Packing efficiency in a unit cell is never 100% because constituent particles are assumed to be:
Detailed Solution: Question 4
What is the dimensional formula of packing fraction?
Detailed Solution: Question 5
Arrange the types of arrangement in terms of decreasing packing efficiency.
Detailed Solution: Question 6
Which of the following shows maximum packing efficiency?
Detailed Solution: Question 7
What are the percentages of free space in a CCP and simple cubic lattice?
Detailed Solution: Question 8
If metallic atoms of mass 197 and radius 166 pm are arranged in ABCABC fashion then what is the surface area of each unit cell?
Detailed Solution: Question 9
If copper, density = 9.0 g/cm3 and atomic mass 63.5, bears face-centered unit cells then what is the ratio of surface area to volume of each copper atom?
Detailed Solution: Question 10
335 videos|699 docs|300 tests |



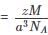
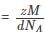
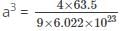
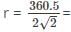 127.46 pm
127.46 pm