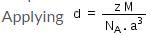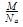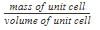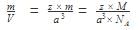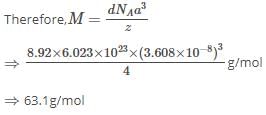Dimensions of Unit Cells (Old NCERT) - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Dimensions of Unit Cells (Old NCERT) (10 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Dimensions of Unit Cells (Old NCERT)". These 10 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
If an element crystallizes as a simple cube, what is the volume of an element provided its density is 1.5 g/cm3 and atomic mass of the element is 63?
Detailed Solution: Question 1
Mass of an atom in an unit cell is calculated by which of the following formula if M is the molar mass:
Detailed Solution: Question 2
In the formula to calculate the density of a unit cell  what is z ?
what is z ?
 what is z ?
what is z ?Detailed Solution: Question 3
Volume of a body centred cubic unit cell is
Detailed Solution: Question 4
Density of a unit cell is equal to mass of unit cell divided by
Detailed Solution: Question 5
Detailed Solution: Question 6
The number of atoms (z) in simple cubic unit cell is
Detailed Solution: Question 7
X-ray diffraction studies show that copper crystallizes in an fcc unit cell with cell edge of 3.608 x 10-8 cm. In a separate experiment, copper is determined to have a density of 8.92 g/cm3, calculate the atomic mass of copper.
Detailed Solution: Question 8
An element with bcc geometry has atomic mass 50 and edge length 290 pm. The density of unit cell will be
Detailed Solution: Question 9
The number of octahedral void(s) per atom present in a cubic close-packed structure is
Detailed Solution: Question 10
446 docs|929 tests |