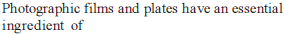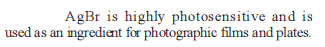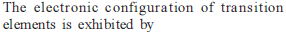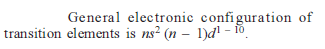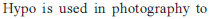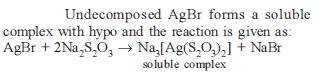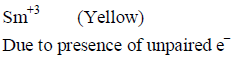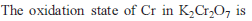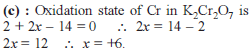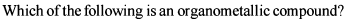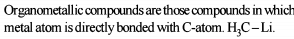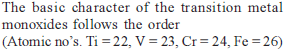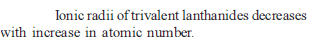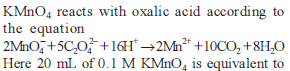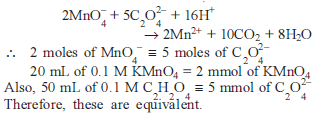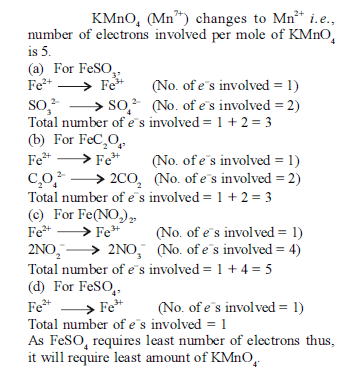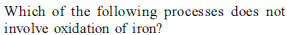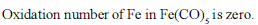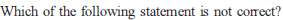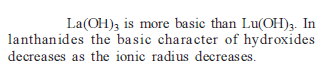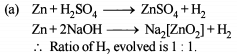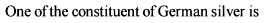d – and f Block Elements - 1 - Free MCQ Test with solutions for JEE Chemistry
MCQ Practice Test & Solutions: d – and f – Block Elements - 1 (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "d – and f – Block Elements - 1". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
What would happen when a solution of potassium chromate is treated with an excess of dilute nitric acid -
[AIEEE-03]
Detailed Solution: Question 1
Excess of KI reacts with CuSO4 solution and then Na2S2O3 solution is added to it. Which of the statements is incorrect for this reaction -
[AIEEE-04]
Detailed Solution: Question 2
Detailed Solution: Question 3
Knowing that the Chemistry of lanthanoids (Ln) is dominated by its +3 oxidation state, which of the following statements is incorrect ?
[AIEEE-09]
Detailed Solution: Question 4
Iron exhibits + 2 and +3 oxidation states. Which of the following statements about iron is incorrect?
[AIEEE-2012]
Detailed Solution: Question 5
Detailed Solution: Question 6
Detailed Solution: Question 7
Detailed Solution: Question 8
Detailed Solution: Question 9
Detailed Solution: Question 10
Detailed Solution: Question 11
Detailed Solution: Question 12
Detailed Solution: Question 13
Detailed Solution: Question 14
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
Detailed Solution: Question 18
Detailed Solution: Question 19
Detailed Solution: Question 20
Detailed Solution: Question 21
Detailed Solution: Question 22
Detailed Solution: Question 23
Detailed Solution: Question 24
Detailed Solution: Question 25
Detailed Solution: Question 26
Detailed Solution: Question 27
Detailed Solution: Question 28
Detailed Solution: Question 29
Detailed Solution: Question 30
332 videos|699 docs|300 tests |