Characteristics of Halo Compounds - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Characteristics of Halo Compounds (10 Questions)
You can prepare effectively for NEET Chemistry Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Characteristics of Halo Compounds". These 10 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which one of the following is likely to give a precipitate with AgNO3 solution?
Detailed Solution: Question 1
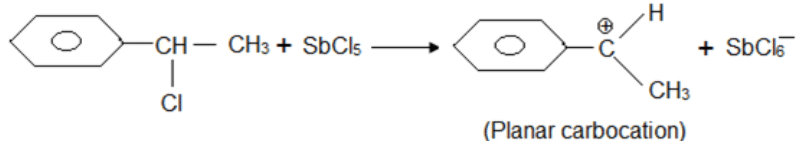
A solution of (–)-1-chloro-1-phenylethane in toluene racemises slowly in the presence of a small amount of SbCl5, due to the formation of
Detailed Solution: Question 2
In SN2 reactions, the correct order of reactivity for the following compounds: CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is
Detailed Solution: Question 3
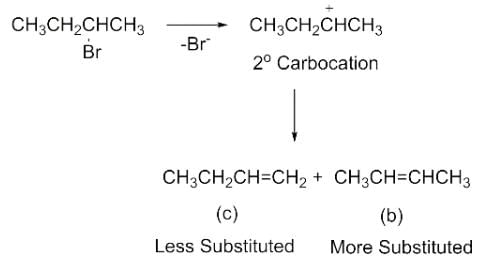
Elimination of bromine from 2-bromobutane results in the formation of
Detailed Solution: Question 4
Which of the following is a tertiary halogenoalkanes?
Detailed Solution: Question 5
Which of the following is not true about optical isomers?
Detailed Solution: Question 6
Which of the following is not an aryl halide?
Detailed Solution: Question 7
Pick up the correct statement about alkyl halides.
Detailed Solution: Question 8
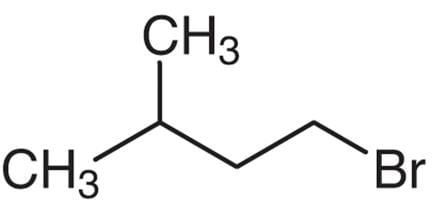
Which one of the following is 1-bromo-3-methyl butane?
Detailed Solution: Question 9
Detailed Solution: Question 10
54 videos|290 docs|74 tests |