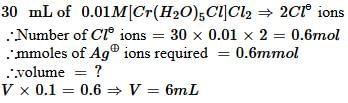Coordination Chemistry- 3 - Chemistry Inorganic Free MCQ Test with solutions
MCQ Practice Test & Solutions: Test: Coordination Chemistry- 3 (30 Questions)
You can prepare effectively for Chemistry Inorganic Chemistry with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Coordination Chemistry- 3". These 30 questions have been designed by the experts with the latest curriculum of Chemistry 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 90 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
The correct order of wavelength (λmax) of the halide to metal charge-transfer band of [Co(NH3)5Cl]2+ (I), [Co(NH3)5Br]2+ (II) and [Co(NH3)5I]2+ (III), is
Detailed Solution: Question 1
Detailed Solution: Question 2
The ground state term symbols for high spin d5s1 and d5 configurations, respectively are:
The volume (in mL) of 0.1 M AgNO3 required for complex precipitation of chloride ions present in 30 mL of 0.01 M solution of [Cr(H2O)5Cl]Cl2, as silver chloride is close to
Detailed Solution: Question 4
The strongest ligand in spectro–chemical series is:
[NiCl2(PPh3)2] is paramagnetic with μeff = 2.9 BM. All the four ligands are monodentate. The geometry of the molecule is:
A solution containing 2.675 g of CoCl3 · 6NH3 (molar mass = 267.5 g mol–1) is passed through a cation exchanger. The chloride ions obtained in solution were treated with excess of AgNO3 to give 4.78 g of AgCl (molar mass = 143.5 g mol–1). The formula of the complex is
(At. Mass of Ag = 108 u)
(At. Mass of Ag = 108 u)
For which of the following ions is the colour in aqueous solution not caused by any d–d transition?
The cyano complex that exhibit highest value of paramagnetism is:
Detailed Solution: Question 11
An excess of AgNO3 is added to 100 mL of a 0.01 M solution of dichlorotetraaquachromium(III) chloride. The number of moles of AgCl precipitated would be
The pair of compounds having metals in their highest oxidation state is
Detailed Solution: Question 14
Correct relat ionship between pairing energy (P) and C.F.S.E (∆0) in complex ion [Ir(H2O)6]3+ is:
In which of the following pairs, both the complexes have the same hybridization?
The value of CFSE (∆0) for complexes given below follow the order:
(I) [Co(NH3)6]3+
(II) [Rh(NH3)6]3+
(III) [Ir(NH3)6]3+
(II) [Rh(NH3)6]3+
(III) [Ir(NH3)6]3+
Cr3+ form for complexes with four different ligands which are [Cr(Cl)6]3–,[Cr(H2O)6]3+, [Cr(NH3)6]3+ and [Cr(CN)6]3–. The order of CFSE (∆0) in these complexes is in the order:
The d–orbital, which are stabilized in an octahedral magnetic field, are:
Total number of geometrical isomers for the complex [RhCl(CO)(PPh3)(NH3)]is
Which of the following is correct arrangement of ligand in terms of the Dq values of their complexes with any particular ‘hard’ metal ion:
The extent of crystal field splitting in octahedral complexes of the given metal with particular weak field ligand are:
A magnetic moment of 1.73 BM will be shown by one among the following
Consider the following two reactions:


According to given information the correct statement (s) is/are:
The value of n for the complex [Fe(CO)4(SiMe3)]n satisfying the 18-electron rule is
Detailed Solution: Question 28
Select correct statement (s) regarding octahedron complex having CFSE = –1.2∆0.
41 videos|93 docs|41 tests |