Chemistry Exam > Chemistry Test > Organic > Test: Pericyclic Level - 1 - Chemistry MCQ
Pericyclic Level - 1 - Free MCQ Practice Test with solutions, Chemistry
MCQ Practice Test & Solutions: Test: Pericyclic Level - 1 (20 Questions)
You can prepare effectively for Chemistry Organic Chemistry with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Pericyclic Level - 1". These 20 questions have been designed by the experts with the latest curriculum of Chemistry 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
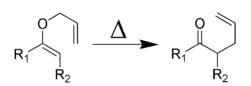
Detailed Solution: Question 1
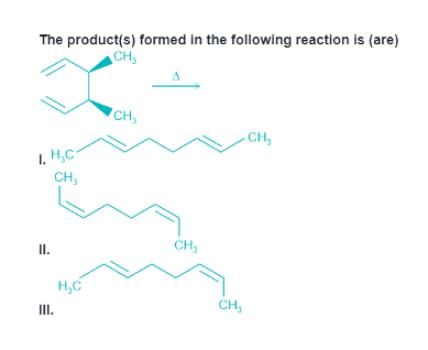
Detailed Solution: Question 13
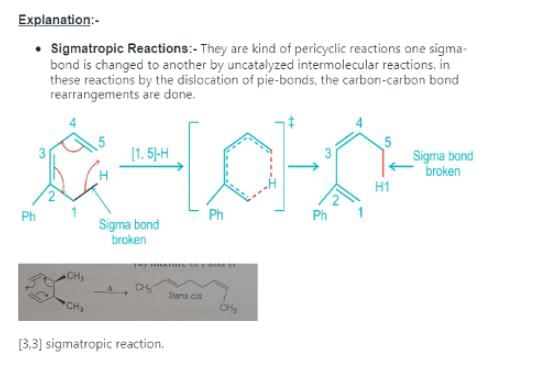
Detailed Solution: Question 16
Test: Pericyclic Level - 1 - Question 19
Which of the following dienes cannot undergo Diels-Alder reactions?
Detailed Solution: Question 19
Detailed Solution: Question 20
39 videos|96 docs|46 tests |
Information about Test: Pericyclic Level - 1 Page
In this test you can find the Exam questions for Test: Pericyclic Level - 1 solved & explained in the simplest way possible. Besides giving Questions and answers for Test: Pericyclic Level - 1, EduRev gives you an ample number of Online tests for practice