Pericyclic Level - 2 - Free MCQ Practice Test with solutions, Chemistry
MCQ Practice Test & Solutions: Test: Pericyclic Level - 2 (30 Questions)
You can prepare effectively for Chemistry Organic Chemistry with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Pericyclic Level - 2". These 30 questions have been designed by the experts with the latest curriculum of Chemistry 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 90 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 3
Detailed Solution: Question 7
Detailed Solution: Question 14
Detailed Solution: Question 15
Detailed Solution: Question 16
The reactivity of compound I-IV with maleic anhydride (V) follows the order:
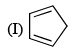
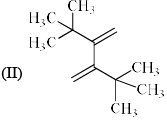
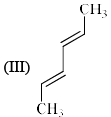
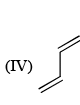
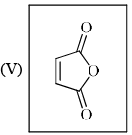
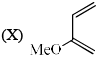
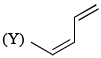
Order of reactivity of the following dienes X, Y and Z in Diels-Alder reaction is:
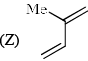
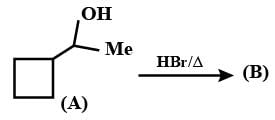
Give the major product and the mechanism involved in the following reaction.

Detailed Solution: Question 20
Amongst the following the compound that does not act as a diene in Diels-Alder reaction is:
In the reaction of Cyclopentadiene with acrylate ester giving diels-Alder reaction products, the interacting frontier orbitals are:
Diels-Alder reaction normally yields endo-adduct as a major product. This is due to:
In the [4 + 2] cycloaddition of 1, 3-butadiene and ethylene:
Detailed Solution: Question 27
The reaction of 1-bromo-2-fluorobenzene with furan in the presence of one equivalent to Mg gives
The diene which undergoes Diels–Alder reaction with maleic anhydride is:
In a Diels–Alder reaction, the most reactive diene amongst the following is:
39 videos|96 docs|46 tests |