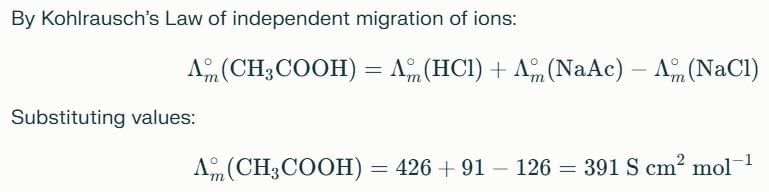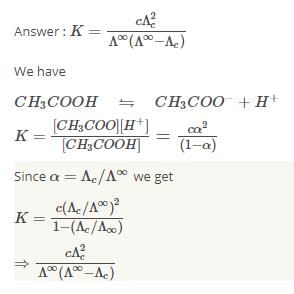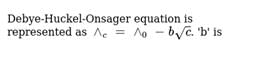Conductance - 1 - Free MCQ Practice Test with solutions, Chemistry Physical
MCQ Practice Test & Solutions: Test: Conductance - 1 (20 Questions)
You can prepare effectively for Chemistry Physical Chemistry with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Conductance - 1". These 20 questions have been designed by the experts with the latest curriculum of Chemistry 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
The cell constant of a conductivity cell is defined as:
Where l = distance between electrodes and A = area of cross-section of each of the electrodes
Detailed Solution: Question 1
Detailed Solution: Question 2
Molar conductivity is defined as:
Where k = conductivity, G = conductance, l = distance between two electrodes and Vm = volume of solution containing 1 mol of electrolyte
Detailed Solution: Question 3
For a dilute solution of a strong electrolyte, the variation of molar conductivity with concentration is given by:
Detailed Solution: Question 4
Which of the following ion is expected to have least value of molar conductivity at infinite dilution in an aqueous solution:
Detailed Solution: Question 5
Detailed Solution: Question 6
The degree of dissociation of a weak electrolyte is given by:
Detailed Solution: Question 7
Which of the following ion has highest molar conductivity at infinite dilution in an aqueous solution:
Detailed Solution: Question 8
The equilibrium constant of acetic acid in an aqueous solution of concentration C is given by
Detailed Solution: Question 9
Equivalent conductivity of Fe2(SO4)3 is related to molar conductivity by the expression:
Detailed Solution: Question 11
Detailed Solution: Question 12
If x is the specific resistance of the electrolyte solution and y is he molarity of the solution, then 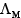 is given by:
is given by:
Detailed Solution: Question 13
When a conductance cell was filled with a 0.0025 M solution of K2SO4, its resistance was 326 Ω. The specific conductance of the solution, if cell constant is 4.
Detailed Solution: Question 14
Equivalent conductance of 0.1 M HA (weak acid) solution is 10 cm2 equivalent–1. The pH of HA solution is(molar conductivity at infinite dilution is 200).
Detailed Solution: Question 15
For HCl solution at 25ºC, the equivalent conductivity at infinite dilution is 425 Ω-1 cm2 eq-1. The specific conductance of a solution of HCl is 3.825 Ω-1 cm-1. If the apparent degree of dissociat ion is 90%, the normality of solution is
Detailed Solution: Question 16
Detailed Solution: Question 17
The correct relation which depicts the Debye-Huckel Onsager Theory for strong electrolytes is:
Detailed Solution: Question 18
The unit of A in Debye-Huckel-Onsager equation for strong electrolytes is:
Detailed Solution: Question 19
Which of the following solutions will show the minimum value of transference number of Cl–1 ions:
Detailed Solution: Question 20
84 videos|147 docs|67 tests |