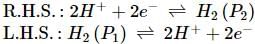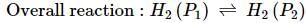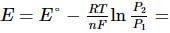JIPMER: Subject wise Tests & Practice JIPMER Chemistry Mock Test - 3 Free
Full Mock Test & Solutions: JIPMER Chemistry Mock Test - 3 (60 Questions)
You can boost your NEET 2026 exam preparation with this JIPMER Chemistry Mock Test - 3 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of NEET 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Total Questions: 60
- - Analysis: Detailed Solutions & Performance Insights
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
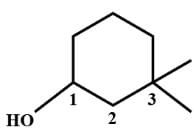
The structural formula of 2-methyl 2-butene is .......
Ethyl bromide on treatment with alocoholic KOH gives
Detailed Solution: Question 4
The first emission line in the atomic spectrum of hydrogen in the Balmer series appears at
The atomic number of an element is 35. What is the total number of electrons present in all the p-orbitals of the ground state atom of the element
Which one of the following is not an isoelectronic pair
A one litre flask is full of brown bromine fumes. The intensity of brown colour of vapour will not decrease appreciably on adding to the flask some
For a given reaction t1/2 = (1/ka). The order of reaction is
The compound with carbon uses only its sp3 hybrid orbitals for bond formation is
The bond dissociation energies of H₂,Cl₂ and HCl are 104, 58 and 103 kcal respectively. The enthalpy of formation of HCl gas will be
Detailed Solution: Question 18
Which pair of atomic numbers represent elements which are both s-block elements
Detailed Solution: Question 19
Which of the following is an example of a double salt?
What will be the e.m.f. of the given cell?
Pt | H2(P1)| H+(aq)|H2(P2)|Pt
Pt | H2(P1)| H+(aq)|H2(P2)|Pt
Detailed Solution: Question 21
At which one of the following temperature-pressure conditions, the deviation of a gas from ideal behaviour is expected to be minimum ?
300 ml of a gas at 27oC is cooled to -3oC at consatnt pressure, the final volume is
The strongest reducing agent of the alkali metal is
3 videos|8 docs|51 tests |
3 videos|8 docs|51 tests |



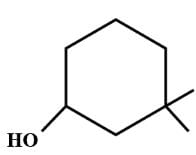 is
is