All Exams > JEE > 6 Months Preparation for JEE > All Questions
All questions of d– and f–Block Elements for JEE Exam
Can you explain the answer of this question below:Which one of the following is a diamagnetic ion?- A:CO2+
- B:Cu2+
- C:Mn2+
- D:Sc3+
The answer is d.
Which one of the following is a diamagnetic ion?
A:
CO2+
B:
Cu2+
C:
Mn2+
D:
Sc3+
 | Learners Habitat answered |
Co+2 = [Ar] 3d7
Cu2+ = [Ar] 3d9
Mn+2 = [Ar] 3d5
Sc+3 = [Ar]
We can see that only Sc+3 has no unpaired electron, so it is a diamagnetic ion.
Cu2+ = [Ar] 3d9
Mn+2 = [Ar] 3d5
Sc+3 = [Ar]
We can see that only Sc+3 has no unpaired electron, so it is a diamagnetic ion.
Which form of silver is colourless?- a)Ag2+
- b)Ag
- c)Ag3+
- d)Ag+
Correct answer is option 'D'. Can you explain this answer?
Which form of silver is colourless?
a)
Ag2+
b)
Ag
c)
Ag3+
d)
Ag+
| | Om Desai answered |
ilver in the form of Ag+ is colourless. For transition metal ions to exhibit color, their metal ions must have incompletely filled (n-1)d orbitals.
Ag+ =4d10,5s0
Ag+ has completely filled d orbitals hence is colourless.
Which ion will show more paramagnetic behaviour ?- a)Cu+
- b)Fe2+
- c)Ag+
- d)Fe3+
Correct answer is option 'D'. Can you explain this answer?
Which ion will show more paramagnetic behaviour ?
a)
Cu+
b)
Fe2+
c)
Ag+
d)
Fe3+
| | Lavanya Menon answered |
Since the configuration of Fe3+ ion is (argon ) 3d5,which contains maximum number of unpaired electrons, hence more will be paramagnetic behavior.
Which among the following is colourless?- a)Sc2+
- b)Zn2+
- c)Ti3+
- d)V3+
Correct answer is option 'B'. Can you explain this answer?
Which among the following is colourless?
a)
Sc2+
b)
Zn2+
c)
Ti3+
d)
V3+
| | Krishna Iyer answered |
Zn2+ has completely filled d-orbitals and there are no vacant d-orbitals for the transition of electrons, hence it is colourless.
The first ionization energy of the d-block elements are?- a)Lesser than p-block elements
- b)Between s and p-block elements
- c)Lesser than s-block elements
- d)Higher than p-block elements
Correct answer is option 'B'. Can you explain this answer?
The first ionization energy of the d-block elements are?
a)
Lesser than p-block elements
b)
Between s and p-block elements
c)
Lesser than s-block elements
d)
Higher than p-block elements
| | Rahul Bansal answered |
The first ionization energy of the d-block elements are between s and p-block elements. Thus they are more electropositive than p-block elements and less electropositive than s-block elements.
Brass is an alloy of:- a)Copper and molybdenum
- b)Copper and zinc
- c)Copper and tungsten
- d)Copper and tin
Correct answer is option 'B'. Can you explain this answer?
Brass is an alloy of:
a)
Copper and molybdenum
b)
Copper and zinc
c)
Copper and tungsten
d)
Copper and tin
 | Mrprince 007 answered |
Brass is an alloy of copper and zinc
Which of the following group of elements are not regarded as transition elements?- a)Sc,Y,La
- b)Cu, Ag, Au
- c)Zn, Cd, Hg
- d)Cr ,Mo,W
Correct answer is option 'C'. Can you explain this answer?
Which of the following group of elements are not regarded as transition elements?
a)
Sc,Y,La
b)
Cu, Ag, Au
c)
Zn, Cd, Hg
d)
Cr ,Mo,W
| | Neha Sharma answered |
Zn, Cd & Hg because the d-orbital of these elements are completely filled. So, they don't show the characteristics of transition elements (i.e. the d-orbital of transition elements is incomplete.
The inner transition elements are the elements in which the added electrons go to:- a)(n-1)d-orbitals
- b)(n-1)d-orbitals and (n-1)f-orbitals
- c)(n-1)d-orbitals and ns orbitals
- d)(n-2)f-orbitals
Correct answer is option 'D'. Can you explain this answer?
The inner transition elements are the elements in which the added electrons go to:
a)
(n-1)d-orbitals
b)
(n-1)d-orbitals and (n-1)f-orbitals
c)
(n-1)d-orbitals and ns orbitals
d)
(n-2)f-orbitals
| | Rohit Shah answered |
Lanthanides and actinides are called inner transition elements because they are a group of elements that are shown as the bottom two rows of the periodic table. ... Lanthanides and actinides belong to the f-block elements, which means that they have filled up their f-orbitals with electrons.
The elements which lie between s and p block elements in the long form periodic table are called as:- a)Actinides
- b)d-block elements
- c)Lanthanides
- d)Electropositive elements
Correct answer is option 'B'. Can you explain this answer?
The elements which lie between s and p block elements in the long form periodic table are called as:
a)
Actinides
b)
d-block elements
c)
Lanthanides
d)
Electropositive elements
| | Harsh Desai answered |
D-block elements
The long form periodic table consists of four blocks: s, p, d, and f. The s and p blocks are located on the left and right sides of the periodic table, respectively. The d-block elements are located in the middle of the periodic table, between the s and p blocks. These elements are also known as transition elements.
Explanation:
The d-block elements are characterized by the presence of partially filled d-orbitals in their valence shells. These elements are often referred to as transition elements because they exhibit a transition between the highly reactive s-block elements and the relatively inert p-block elements. The d-block elements are known for their unique chemical and physical properties, such as their ability to form complex ions and their high melting and boiling points.
Examples of d-block elements include titanium, iron, copper, and zinc. These elements are widely used in industry and technology due to their unique properties, such as their strength, durability, and conductivity.
In summary, the elements which lie between s and p block elements in the long form periodic table are called d-block elements or transition elements. These elements exhibit unique chemical and physical properties and are widely used in industry and technology.
The long form periodic table consists of four blocks: s, p, d, and f. The s and p blocks are located on the left and right sides of the periodic table, respectively. The d-block elements are located in the middle of the periodic table, between the s and p blocks. These elements are also known as transition elements.
Explanation:
The d-block elements are characterized by the presence of partially filled d-orbitals in their valence shells. These elements are often referred to as transition elements because they exhibit a transition between the highly reactive s-block elements and the relatively inert p-block elements. The d-block elements are known for their unique chemical and physical properties, such as their ability to form complex ions and their high melting and boiling points.
Examples of d-block elements include titanium, iron, copper, and zinc. These elements are widely used in industry and technology due to their unique properties, such as their strength, durability, and conductivity.
In summary, the elements which lie between s and p block elements in the long form periodic table are called d-block elements or transition elements. These elements exhibit unique chemical and physical properties and are widely used in industry and technology.
Which of the following is not an interstitial compound?- a)Mn4N
- b)TiC
- c)Fe3H
- d)FeCl3
Correct answer is option 'D'. Can you explain this answer?
Which of the following is not an interstitial compound?
a)
Mn4N
b)
TiC
c)
Fe3H
d)
FeCl3
| | Lavanya Menon answered |
The correct answer is option D
An interstitial compound is a compound that is formed when an atom with a small enough radius sits a hole in a metal lattice
They are usually transitional elements
Eg: Tic, Mn4N, Fe3H, TiH2 etc.
Main Characteristics:-
· High melting points than metals
· They are much harder
· They become chemically inert
· They become less malleable
So, the answer is (a), (b), (c) .
An interstitial compound is a compound that is formed when an atom with a small enough radius sits a hole in a metal lattice
They are usually transitional elements
Eg: Tic, Mn4N, Fe3H, TiH2 etc.
Main Characteristics:-
· High melting points than metals
· They are much harder
· They become chemically inert
· They become less malleable
So, the answer is (a), (b), (c) .
In neutral solution, potassium permanganate forms:- a)Manganese dioxide
- b)Manganous sulphate
- c)Pyrolusite
- d)Potassium manganite
Correct answer is option 'A'. Can you explain this answer?
In neutral solution, potassium permanganate forms:
a)
Manganese dioxide
b)
Manganous sulphate
c)
Pyrolusite
d)
Potassium manganite
 | Prakash answered |
KMno₄ is an oxidising agent (here Mn⁺⁷)in nuetral medium there is a change of 3 in it's oxidation state M⁺⁷-------Mn⁺⁴
The second series of transition element starts with:- a)Scandium
- b)Rhodium
- c)Ytterium
- d)Actinium
Correct answer is option 'C'. Can you explain this answer?
The second series of transition element starts with:
a)
Scandium
b)
Rhodium
c)
Ytterium
d)
Actinium
| | Pooja Mehta answered |
The second series includes the elements yttrium (symbol Y, atomic number 39) to cadmium (symbol Cd, atomic number 48)
Melting point of d block elements across a period:- a)Increases from left to right
- b)Deceases from left to right
- c)Increases to a maximum at d5 and then decreases with increase of atomic number.
- d)Does not change on moving from left to right.
Correct answer is option 'C'. Can you explain this answer?
Melting point of d block elements across a period:
a)
Increases from left to right
b)
Deceases from left to right
c)
Increases to a maximum at d5 and then decreases with increase of atomic number.
d)
Does not change on moving from left to right.
| | Jayant Mishra answered |
The melting and boiling points first increase, reaches maximum and then steadily decrease across any transition series. ... The low melting points of Zn, Cd, and Hg are due to the absence of unpaired d-electrons in their atoms and thus low metallic bonding.
Which of the following is not the condition for the complex formation?- a)High ionic charge density on metal ion.
- b)Smaller size of transition metal ion.
- c)Absence of vacant d-orbitals.
- d)Availability of vacant d-orbitals.
Correct answer is option 'C'. Can you explain this answer?
Which of the following is not the condition for the complex formation?
a)
High ionic charge density on metal ion.
b)
Smaller size of transition metal ion.
c)
Absence of vacant d-orbitals.
d)
Availability of vacant d-orbitals.
| | Krishna Iyer answered |
The correct answer is Option C
Only d orbitals accept the lone pair of electrons during complex formation. Their absence means that the atom will not be able to accept the lone pair of electrons and the complex will not be formed.
Only d orbitals accept the lone pair of electrons during complex formation. Their absence means that the atom will not be able to accept the lone pair of electrons and the complex will not be formed.
Transition metals with highest melting point is- a)Cr
- b)W
- c)Hg
- d)Sc
Correct answer is option 'B'. Can you explain this answer?
Transition metals with highest melting point is
a)
Cr
b)
W
c)
Hg
d)
Sc
 | Pragati Choudhury answered |
W belongs to 5d series and also it have lot of unpaired electrons thus it forms strong metallic bonding.
Ag+ ion is isoelectronic with- a)Zn2+
- b)Cd2+
- c)Pd2+
- d)Cu2+
Correct answer is option 'B'. Can you explain this answer?
Ag+ ion is isoelectronic with
a)
Zn2+
b)
Cd2+
c)
Pd2+
d)
Cu2+
 | Malavika Shah answered |
Ag+ is isoelectronic with Cd2+
When an alkali is added to an orange red solution of dichromate, it forms:- a)Ferrochrome
- b)Chromyl chloride
- c)Chromate
- d)Chromium trioxide
Correct answer is option 'C'. Can you explain this answer?
When an alkali is added to an orange red solution of dichromate, it forms:
a)
Ferrochrome
b)
Chromyl chloride
c)
Chromate
d)
Chromium trioxide
 | Knowledge Hub answered |
The correct answer is C
When an alkali is added to an orange red solution of dichromate it forms chromate.
For example:
K2Cr2O7 + 2KOH ➡ 2K2CrO4 + H2O
When an alkali is added to an orange red solution of dichromate it forms chromate.
For example:
K2Cr2O7 + 2KOH ➡ 2K2CrO4 + H2O
Lucas reagent is- a)ZnCl2 + HCl
- b)MnO2 + H2O
- c)H2SO4 + HCl
- d)NO + H2O
Correct answer is option 'A'. Can you explain this answer?
Lucas reagent is
a)
ZnCl2 + HCl
b)
MnO2 + H2O
c)
H2SO4 + HCl
d)
NO + H2O
 | Arshiya Choudhury answered |
Lucas Test is done to distinguish primary secondary and tertiary alcohols. Lucas reagent is ZnCl2 + HCl
Which one of the following is a diamagnetic ion?- a)CO2+
- b)Cu2+
- c)Mn2+
- d)Sc3+
Correct answer is option 'D'. Can you explain this answer?
Which one of the following is a diamagnetic ion?
a)
CO2+
b)
Cu2+
c)
Mn2+
d)
Sc3+
| | Ameya Pillai answered |
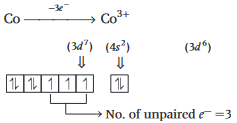
Co2+ (Z = 27) : [Ar]183d7 (3 unpaired electrons)
Cu2+ (Z = 29) : [Ar]183d9 (1 unpaired electrons)
Mn2+ (Z = 25): [Ar]183d5 (5 unpaired electrons)
Sc3+ (Z = 21): [Ar]183d0 (No unpaired electron)
Sc3+ with no unpaired electron will be diamagnetic
Cu2+ (Z = 29) : [Ar]183d9 (1 unpaired electrons)
Mn2+ (Z = 25): [Ar]183d5 (5 unpaired electrons)
Sc3+ (Z = 21): [Ar]183d0 (No unpaired electron)
Sc3+ with no unpaired electron will be diamagnetic
For the four successive transition elements (Cr, Mn, Fe and Co), the stability of + 2 oxidation state will be there in which of the following order?- a)Mn > Cr > Fe > Co
- b)Fe > Mn > Co > Cr
- c)Co > Mn > Fe > Cr
- d)Cr > Mn > Co > Fe
Correct answer is option 'A'. Can you explain this answer?
For the four successive transition elements (Cr, Mn, Fe and Co), the stability of + 2 oxidation state will be there in which of the following order?
a)
Mn > Cr > Fe > Co
b)
Fe > Mn > Co > Cr
c)
Co > Mn > Fe > Cr
d)
Cr > Mn > Co > Fe
 | Mohit Rajpoot answered |
Mn2+ (d5) > Cr2+ (d4) > Fe2+ (d6) > Co2+ (d7) Mn2+ is more stable due to half-filled configuration, i . e . d5.
Transition metals and their compounds have paramagnetic properties due to the presence of unpaired electrons in (n - 1)d-orbitals. The paramagnetic behaviour is expressed in terms of magnetic moment which is because of the spin of the unpaired electrons (n). It is given as
Magnetic moment = 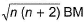
Majority of transition metal compounds are coloured both in solid state as well as in aqueous solution. This is also due to the presence of unpaired electrons in (n - 1) d-orbitals, d-orbitals splitting and d-d transition of electrons absorbing suitable visible light.Q. Which pair of ions are expected to be diamagnetic?- a)Cu+ and Cu2+
- b)Ag+ and Au+
- c)Ag+ and Au3+
- d)Zn2+ and Cu2+
Correct answer is option 'B'. Can you explain this answer?
Transition metals and their compounds have paramagnetic properties due to the presence of unpaired electrons in (n - 1)d-orbitals. The paramagnetic behaviour is expressed in terms of magnetic moment which is because of the spin of the unpaired electrons (n). It is given as
Magnetic moment =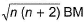
Majority of transition metal compounds are coloured both in solid state as well as in aqueous solution. This is also due to the presence of unpaired electrons in (n - 1) d-orbitals, d-orbitals splitting and d-d transition of electrons absorbing suitable visible light.
Magnetic moment =
Majority of transition metal compounds are coloured both in solid state as well as in aqueous solution. This is also due to the presence of unpaired electrons in (n - 1) d-orbitals, d-orbitals splitting and d-d transition of electrons absorbing suitable visible light.
Q.
Which pair of ions are expected to be diamagnetic?
a)
Cu+ and Cu2+
b)
Ag+ and Au+
c)
Ag+ and Au3+
d)
Zn2+ and Cu2+
 | Ishani Yadav answered |
Ag+ (d10) and Au+(d10) are diamagnetic due to absence of unpaired number of electrons.
Mohr’s salt is a- a)Acidic salt
- b)Double salt
- c)Basic Acidic salt
- d)Normal salt
Correct answer is option 'B'. Can you explain this answer?
Mohr’s salt is a
a)
Acidic salt
b)
Double salt
c)
Basic Acidic salt
d)
Normal salt
 | Amar Pillai answered |
Mohr salt is an example of double salt. Mohr Salt is
FeSO4 . (NH4)2 SO4.6H2O
Which of the following ion has smallest radii?- a)V2+
- b)Ni2+
- c)Mn2+
- d)Ti2+
Correct answer is option 'B'. Can you explain this answer?
Which of the following ion has smallest radii?
a)
V2+
b)
Ni2+
c)
Mn2+
d)
Ti2+
 | Preethi Bose answered |
In period when we move from left to right in a period atomic radii 1st decreases till nickel then starts increasing so Ni2+ has smallest radii.
A reduction in the atomic size with increase in atomic number is characteristic of the elements of- a)high atomic masses
- b)d – block
- c)f – block
- d)radioactive series
Correct answer is option 'C'. Can you explain this answer?
A reduction in the atomic size with increase in atomic number is characteristic of the elements of
a)
high atomic masses
b)
d – block
c)
f – block
d)
radioactive series
 | Nidhi Nambiar answered |
In f block elements with increase in atomic number atomic radii decreases smoothly.
Many copper (I) compounds are unstable in aqueous solution and undergo disproportionation as 2Cu+ → Cu + Cu2+ . This is due to- a)free energy change of reaction is positive
- b)free energy of reaction is negative
- c)second ionisation energy of Cu is higher than first ionisation energy
- d)hydration energy of Cu+ is less than Cu2+
Correct answer is option 'B'. Can you explain this answer?
Many copper (I) compounds are unstable in aqueous solution and undergo disproportionation as 2Cu+ → Cu + Cu2+ . This is due to
a)
free energy change of reaction is positive
b)
free energy of reaction is negative
c)
second ionisation energy of Cu is higher than first ionisation energy
d)
hydration energy of Cu+ is less than Cu2+
| | Anjana Sharma answered |
Cu+ is unstable in aqueous solution than Cu2+ because although 2nd I.E. of copper is large but hydration enthalpy for Cu2+ is much more negative than that for Cu+ and hence, it is more than compensates for the 2nd I.E. of copper. Therefore, many Cu+ compounds are unstable in aqueous solution and undergo disproportionation.
2Cu+ → Cu2+ + Cu
Which is called chromic acid?- a)Cr2O3
- b)Cr3O4
- c)CrO
- d)H2CrO4
Correct answer is option 'D'. Can you explain this answer?
Which is called chromic acid?
a)
Cr2O3
b)
Cr3O4
c)
CrO
d)
H2CrO4
| | Shraddha Dey answered |
Chromic Acid is a naturally occurring oxide with a formula H2CrO4.
Ferrous sulphate on heating gives- a)SO2 and SO3
- b)SO2 and O2
- c)SO2
- d)SO3
Correct answer is option 'A'. Can you explain this answer?
Ferrous sulphate on heating gives
a)
SO2 and SO3
b)
SO2 and O2
c)
SO2
d)
SO3
 | M. Vishnu answered |
On heating, ferrous sulphate crystals lose water and anhydrous ferrous sulphate (FeSO4) is formed. So their colour changes from light green to white. On furtherheating, anhydrous ferrous sulphate decomposes to form ferric oxide (Fe2O3), sulphur dioxide (SO2) and sulphur trioxide (SO3).
Maximum magnetic moment is shown by- a)d6
- b)d8
- c)d5
- d)d7
Correct answer is option 'C'. Can you explain this answer?
Maximum magnetic moment is shown by
a)
d6
b)
d8
c)
d5
d)
d7
 | Dishani Kulkarni answered |
Magnetic moment is maximum for the element having maximum unpaired electrons.
One or More than One Options Correct TypeDirection (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.Q. Which one of the following arrangements represent the correct order of the property stated against it?- a)V2+ < Cr2+ < Mn2+ < Fe2+ : paramagnetic behaviour
- b)Ni2+ < Co2+ < Fe2+ < Mn2+: ionic size
- c)Co3+ < Fe3+ < Cr3+ < Sc3+ : stability in aqueous solution
- d)Sc < Ti < Cr < Mn : number of oxidation state
Correct answer is option 'B,C,D'. Can you explain this answer?
One or More than One Options Correct Type
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
Which one of the following arrangements represent the correct order of the property stated against it?
a)
V2+ < Cr2+ < Mn2+ < Fe2+ : paramagnetic behaviour
b)
Ni2+ < Co2+ < Fe2+ < Mn2+: ionic size
c)
Co3+ < Fe3+ < Cr3+ < Sc3+ : stability in aqueous solution
d)
Sc < Ti < Cr < Mn : number of oxidation state
 | Arshiya Choudhury answered |

[Ti (H2O)6]3+ absorbs green and yellow region part of visible light. Then the transmitted colour of the compound is- a)purple
- b)green
- c)blue
- d)red
Correct answer is option 'A'. Can you explain this answer?
[Ti (H2O)6]3+ absorbs green and yellow region part of visible light. Then the transmitted colour of the compound is
a)
purple
b)
green
c)
blue
d)
red
 | Ishani Yadav answered |
We know that white light is composed of many different colours. When white light is allowed to fall on [Ti(H2O)6]3+ ion, this ion absorbs the light of green and yellow colour and transmits is almost purple colour.The purple colour of the transmitted light is called complimentary colour of that of absorbed light.
An aqueous solution of [Ti(H2O)6]3+ is:- a)Greenish-yellow in colour
- b)Violet in colour
- c)Blue in colour
- d)Purple in colour
Correct answer is option 'D'. Can you explain this answer?
An aqueous solution of [Ti(H2O)6]3+ is:
a)
Greenish-yellow in colour
b)
Violet in colour
c)
Blue in colour
d)
Purple in colour
| | Arjun Chavan answered |
The aqueous solution of [Ti(H2O)6]3+ is purple in color. This is due to the presence of a transition metal ion, titanium, in the complex ion.
Explanation:
- The complex ion [Ti(H2O)6]3+ is formed by the coordination of six water molecules around a titanium(III) ion. The coordination number of titanium is 6, which means that it is surrounded by six ligands (water molecules) in the complex.
- The color of a complex ion is determined by the d-electrons in the transition metal ion. In the case of [Ti(H2O)6]3+, titanium has one unpaired electron in its d-orbital, which can undergo d-d transitions when it absorbs light.
- The absorption of light causes the electrons to move from a lower energy level to a higher energy level, which results in the color of the complex. In the case of [Ti(H2O)6]3+, the absorption of light causes the complex to appear purple.
- The purple color of [Ti(H2O)6]3+ can be used as a qualitative test for the presence of titanium in a sample. If a sample contains titanium ions, the addition of a small amount of [Ti(H2O)6]3+ will cause the solution to turn purple.
In summary, the aqueous solution of [Ti(H2O)6]3+ is purple in color due to the presence of a transition metal ion, titanium, in the complex ion. The color of the complex is determined by the d-electrons in the transition metal ion and can be used as a qualitative test for the presence of titanium in a sample.
Explanation:
- The complex ion [Ti(H2O)6]3+ is formed by the coordination of six water molecules around a titanium(III) ion. The coordination number of titanium is 6, which means that it is surrounded by six ligands (water molecules) in the complex.
- The color of a complex ion is determined by the d-electrons in the transition metal ion. In the case of [Ti(H2O)6]3+, titanium has one unpaired electron in its d-orbital, which can undergo d-d transitions when it absorbs light.
- The absorption of light causes the electrons to move from a lower energy level to a higher energy level, which results in the color of the complex. In the case of [Ti(H2O)6]3+, the absorption of light causes the complex to appear purple.
- The purple color of [Ti(H2O)6]3+ can be used as a qualitative test for the presence of titanium in a sample. If a sample contains titanium ions, the addition of a small amount of [Ti(H2O)6]3+ will cause the solution to turn purple.
In summary, the aqueous solution of [Ti(H2O)6]3+ is purple in color due to the presence of a transition metal ion, titanium, in the complex ion. The color of the complex is determined by the d-electrons in the transition metal ion and can be used as a qualitative test for the presence of titanium in a sample.
The catalytic activity of transition metals and their compounds is mainly due to- a)their magnetic behaviour
- b)their ful-filled d-orbitals
- c)their ability to adopt variable oxidation states
- d)their chemical reactivity
Correct answer is option 'C'. Can you explain this answer?
The catalytic activity of transition metals and their compounds is mainly due to
a)
their magnetic behaviour
b)
their ful-filled d-orbitals
c)
their ability to adopt variable oxidation states
d)
their chemical reactivity
 | Nishtha Bose answered |
The variability of oxidation state, a characteristic of transition element arises due to incomplete filling of d-orbitals.
Which of the following pairs of ions have same paramagnetic moment?- a)Cu2+, Ti3+
- b)Ti3+, Ni2+
- c)Ti4+, Cu2+
- d)Mn2+, Cu2+
Correct answer is option 'A'. Can you explain this answer?
Which of the following pairs of ions have same paramagnetic moment?
a)
Cu2+, Ti3+
b)
Ti3+, Ni2+
c)
Ti4+, Cu2+
d)
Mn2+, Cu2+
 | Tejas Desai answered |
Cu2+, Ti3+ will have same unpaired electrons and hence same paramagnetic moment.
The basic character of the transition metal monoxides follows the order (Atomic Nos., Ti = 22, V = 23, Cr = 24, Fe = 26)- a)TiO > VO > CrO > FeO
- b)VO > CrO > TiO > FeO
- c)CrO > VO > FeO > TiO
- d)TiO > FeO > VO > CrO
Correct answer is option 'A'. Can you explain this answer?
The basic character of the transition metal monoxides follows the order (Atomic Nos., Ti = 22, V = 23, Cr = 24, Fe = 26)
a)
TiO > VO > CrO > FeO
b)
VO > CrO > TiO > FeO
c)
CrO > VO > FeO > TiO
d)
TiO > FeO > VO > CrO
| | Tejas Malik answered |
Understanding Transition Metal Monoxides
The transition metal monoxides (TiO, VO, CrO, FeO) display variations in their properties due to changes in metallic character related to their atomic numbers.
Key Factors Influencing Properties
- Atomic Number and Electron Configuration: As we move from Ti (22) to Fe (26), the number of d-electrons increases. The varying d-electron count influences the oxidation states and the bonding character of the oxides.
- Metallic Character: The metallic character decreases across the series from Ti to Fe. This means that TiO will exhibit the most basic character, while FeO will exhibit the least.
- Basicity: Basicity in metal oxides is largely influenced by the ability of the metal to donate electrons. Ti, with its lower oxidation state and higher metallic character, leads to a more basic oxide compared to the others.
Order of Basic Character
Based on the above factors, the order of basic character can be summarized as:
- TiO: Most basic due to the highest metallic character.
- VO: Less basic than TiO, but more basic than CrO and FeO.
- CrO: Intermediate basicity due to its higher oxidation states compared to Ti and V.
- FeO: Least basic, as it has the highest oxidation state and lower metallic character.
Conclusion: Correct Answer
Thus, the correct order of basic character for the transition metal monoxides is:
TiO > VO > CrO > FeO, which corresponds to option 'A'.
The transition metal monoxides (TiO, VO, CrO, FeO) display variations in their properties due to changes in metallic character related to their atomic numbers.
Key Factors Influencing Properties
- Atomic Number and Electron Configuration: As we move from Ti (22) to Fe (26), the number of d-electrons increases. The varying d-electron count influences the oxidation states and the bonding character of the oxides.
- Metallic Character: The metallic character decreases across the series from Ti to Fe. This means that TiO will exhibit the most basic character, while FeO will exhibit the least.
- Basicity: Basicity in metal oxides is largely influenced by the ability of the metal to donate electrons. Ti, with its lower oxidation state and higher metallic character, leads to a more basic oxide compared to the others.
Order of Basic Character
Based on the above factors, the order of basic character can be summarized as:
- TiO: Most basic due to the highest metallic character.
- VO: Less basic than TiO, but more basic than CrO and FeO.
- CrO: Intermediate basicity due to its higher oxidation states compared to Ti and V.
- FeO: Least basic, as it has the highest oxidation state and lower metallic character.
Conclusion: Correct Answer
Thus, the correct order of basic character for the transition metal monoxides is:
TiO > VO > CrO > FeO, which corresponds to option 'A'.
How many unpaired electrons will be present in the ground state of an atom which has valence electronic configuration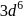 in its +3 oxidation state?
in its +3 oxidation state?- a)1
- b)3
- c)4
- d)7
Correct answer is option 'B'. Can you explain this answer?
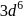 in its +3 oxidation state?
in its +3 oxidation state?a)
1
b)
3
c)
4
d)
7
 | Imk Pathsala answered |
Ground state electronic configuration of the element (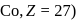 will be
will be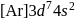 .
.
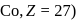 will be
will be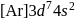 .
.
Which one of the following combines with Fe2+ ion to form a brown complex?- a)N2O
- b)N2O3
- c)N2O5
- d)NO
Correct answer is option 'D'. Can you explain this answer?
Which one of the following combines with Fe2+ ion to form a brown complex?
a)
N2O
b)
N2O3
c)
N2O5
d)
NO
 | Sankar Chakraborty answered |
[Fe(H2O)5NO]2+is brown color complex.
Which among the following transition metal has lowest melting point?- a)Titanium
- b)Cobalt
- c)Mercury
- d)Scandium
Correct answer is option 'C'. Can you explain this answer?
Which among the following transition metal has lowest melting point?
a)
Titanium
b)
Cobalt
c)
Mercury
d)
Scandium
 | Mohit Rajpoot answered |
Mercury is a transition metal that exhibits an unusually low melting point compared to other transition metals. This is due to its unique electron configuration and the relativistic effects that weaken its metallic bonding, resulting in a melting point of -38.83°C.
Which of the following methods is based on distribution law?- a)Parke’s process
- b)Poling process
- c)Mond’s process
- d)Cupellation process
Correct answer is option 'A'. Can you explain this answer?
Which of the following methods is based on distribution law?
a)
Parke’s process
b)
Poling process
c)
Mond’s process
d)
Cupellation process
 | Saptarshi Ghoshal answered |
Parkes process is a process for removing silver from Pb. It is an example of Liquid liquid extractions
Paramagnetic Curie temperature in Kelvin for iron is equal to :- a)2195
- b)495
- c)895
- d)1043
Correct answer is option 'D'. Can you explain this answer?
Paramagnetic Curie temperature in Kelvin for iron is equal to :
a)
2195
b)
495
c)
895
d)
1043
 | EduRev JEE answered |
The paramagnetic Curie temperature for iron is a critical point in its magnetic behaviour.
- At temperatures above the Curie point, iron loses its ferromagnetic properties.
- The Curie temperature for iron is 1043 Kelvin.
- Above this temperature, iron becomes paramagnetic.
This means it is weakly attracted by a magnetic field and does not retain magnetisation without an external field.
In which of the following the stability of two oxidation states is correctly represented?- a)Ti3+ > Ti4+
- b)Mn2+ > Mn3+
- c)Fe2+ > Fe3+
- d)Cu+ > Cu2+
Correct answer is option 'B'. Can you explain this answer?
In which of the following the stability of two oxidation states is correctly represented?
a)
Ti3+ > Ti4+
b)
Mn2+ > Mn3+
c)
Fe2+ > Fe3+
d)
Cu+ > Cu2+
| | Gauri Datta answered |
Stability of Two Oxidation States
Introduction:
The stability of oxidation states refers to the tendency of an element to exist in a particular oxidation state. Some elements can exist in multiple oxidation states, and the stability of these states depends on various factors such as electron configuration, ionization energy, and the nature of bonding.
Explanation:
In the given options, the correct representation of the stability of two oxidation states is option B, which states Mn2 and Mn3.
Mn2:
- The element manganese (Mn) has an atomic number of 25, and its electron configuration is [Ar] 3d5 4s2.
- Mn2 refers to the +2 oxidation state of manganese. In this state, two electrons are removed from the 4s orbital, leaving behind the electron configuration [Ar] 3d5.
- The 3d5 configuration is a half-filled or near-half-filled configuration, which is relatively stable due to the exchange energy and electron-electron repulsion effects.
- Hence, Mn2 is a stable oxidation state for manganese.
Mn3:
- Mn3 refers to the +3 oxidation state of manganese. In this state, three electrons are removed from the 4s and 3d orbitals, leaving behind the electron configuration [Ar] 3d4.
- The 3d4 configuration is also relatively stable as it is a half-filled configuration, similar to Mn2.
- Additionally, the removal of another electron from the 3d orbital results in the formation of a high-spin d4 configuration, which is energetically favorable.
- Therefore, Mn3 is also a stable oxidation state for manganese.
Other Options:
- Option A (Ti3 and Ti4): Titanium (Ti) has an atomic number of 22, and its electron configuration is [Ar] 3d2 4s2.
- Ti3 would require the removal of all four valence electrons, resulting in an unstable configuration.
- Ti4 would require the removal of all six valence electrons, resulting in an extremely unstable configuration.
- Option C (Fe2 and Fe3): Iron (Fe) has an atomic number of 26, and its electron configuration is [Ar] 3d6 4s2.
- Fe2 would require the removal of all six valence electrons, resulting in an unstable configuration.
- Fe3 would require the removal of all eight valence electrons, resulting in an extremely unstable configuration.
- Option D (Cu and Cu2): Copper (Cu) has an atomic number of 29, and its electron configuration is [Ar] 3d10 4s1.
- Cu is the most stable in its +1 oxidation state (Cu+), where it loses the 4s electron.
- Cu2 would require the removal of all ten valence electrons, resulting in an extremely unstable configuration.
Conclusion:
Based on the electron configurations and stability considerations, the correct representation of the stability of two oxidation states is option B, which states Mn2 and Mn3.
Introduction:
The stability of oxidation states refers to the tendency of an element to exist in a particular oxidation state. Some elements can exist in multiple oxidation states, and the stability of these states depends on various factors such as electron configuration, ionization energy, and the nature of bonding.
Explanation:
In the given options, the correct representation of the stability of two oxidation states is option B, which states Mn2 and Mn3.
Mn2:
- The element manganese (Mn) has an atomic number of 25, and its electron configuration is [Ar] 3d5 4s2.
- Mn2 refers to the +2 oxidation state of manganese. In this state, two electrons are removed from the 4s orbital, leaving behind the electron configuration [Ar] 3d5.
- The 3d5 configuration is a half-filled or near-half-filled configuration, which is relatively stable due to the exchange energy and electron-electron repulsion effects.
- Hence, Mn2 is a stable oxidation state for manganese.
Mn3:
- Mn3 refers to the +3 oxidation state of manganese. In this state, three electrons are removed from the 4s and 3d orbitals, leaving behind the electron configuration [Ar] 3d4.
- The 3d4 configuration is also relatively stable as it is a half-filled configuration, similar to Mn2.
- Additionally, the removal of another electron from the 3d orbital results in the formation of a high-spin d4 configuration, which is energetically favorable.
- Therefore, Mn3 is also a stable oxidation state for manganese.
Other Options:
- Option A (Ti3 and Ti4): Titanium (Ti) has an atomic number of 22, and its electron configuration is [Ar] 3d2 4s2.
- Ti3 would require the removal of all four valence electrons, resulting in an unstable configuration.
- Ti4 would require the removal of all six valence electrons, resulting in an extremely unstable configuration.
- Option C (Fe2 and Fe3): Iron (Fe) has an atomic number of 26, and its electron configuration is [Ar] 3d6 4s2.
- Fe2 would require the removal of all six valence electrons, resulting in an unstable configuration.
- Fe3 would require the removal of all eight valence electrons, resulting in an extremely unstable configuration.
- Option D (Cu and Cu2): Copper (Cu) has an atomic number of 29, and its electron configuration is [Ar] 3d10 4s1.
- Cu is the most stable in its +1 oxidation state (Cu+), where it loses the 4s electron.
- Cu2 would require the removal of all ten valence electrons, resulting in an extremely unstable configuration.
Conclusion:
Based on the electron configurations and stability considerations, the correct representation of the stability of two oxidation states is option B, which states Mn2 and Mn3.
+8 oxidation state is/are shown by- a)Fe
- b)Ru
- c)Os
- d)W
Correct answer is option 'B,C'. Can you explain this answer?
+8 oxidation state is/are shown by
a)
Fe
b)
Ru
c)
Os
d)
W
 | Sinjini Tiwari answered |
The question is asking which elements can exhibit an oxidation state of +8.
Oxidation state is a measure of the degree of oxidation of an atom in a chemical compound, and it indicates the number of electrons that an atom has gained or lost when forming a compound. The oxidation state of an element can vary depending on the compound it is in.
Here are the elements mentioned in the options and their possible oxidation states:
a) Fe (Iron): Iron can exhibit oxidation states ranging from -2 to +6. However, an oxidation state of +8 is not possible for iron.
b) Ru (Ruthenium): Ruthenium can exhibit oxidation states ranging from -2 to +8. Therefore, an oxidation state of +8 is possible for ruthenium.
c) Os (Osmium): Osmium can exhibit oxidation states ranging from -2 to +8. Therefore, an oxidation state of +8 is possible for osmium.
d) W (Tungsten): Tungsten can exhibit oxidation states ranging from -2 to +6. However, an oxidation state of +8 is not possible for tungsten.
Therefore, the correct answer is options B and C, which are Ru and Os.
In summary:
- Fe can exhibit oxidation states from -2 to +6, but not +8.
- Ru can exhibit oxidation states from -2 to +8, including +8.
- Os can exhibit oxidation states from -2 to +8, including +8.
- W can exhibit oxidation states from -2 to +6, but not +8.
Oxidation state is a measure of the degree of oxidation of an atom in a chemical compound, and it indicates the number of electrons that an atom has gained or lost when forming a compound. The oxidation state of an element can vary depending on the compound it is in.
Here are the elements mentioned in the options and their possible oxidation states:
a) Fe (Iron): Iron can exhibit oxidation states ranging from -2 to +6. However, an oxidation state of +8 is not possible for iron.
b) Ru (Ruthenium): Ruthenium can exhibit oxidation states ranging from -2 to +8. Therefore, an oxidation state of +8 is possible for ruthenium.
c) Os (Osmium): Osmium can exhibit oxidation states ranging from -2 to +8. Therefore, an oxidation state of +8 is possible for osmium.
d) W (Tungsten): Tungsten can exhibit oxidation states ranging from -2 to +6. However, an oxidation state of +8 is not possible for tungsten.
Therefore, the correct answer is options B and C, which are Ru and Os.
In summary:
- Fe can exhibit oxidation states from -2 to +6, but not +8.
- Ru can exhibit oxidation states from -2 to +8, including +8.
- Os can exhibit oxidation states from -2 to +8, including +8.
- W can exhibit oxidation states from -2 to +6, but not +8.
Ions having same colour in aqueous solution are- a)Cu2+ and Fe2+
- b)Ni2+ and Fe2+
- c)Fe3+ and Cr3+
- d)V4+ and Cr2+
Correct answer is option 'B,D'. Can you explain this answer?
Ions having same colour in aqueous solution are
a)
Cu2+ and Fe2+
b)
Ni2+ and Fe2+
c)
Fe3+ and Cr3+
d)
V4+ and Cr2+
 | Sinjini Tiwari answered |
Ni2+ and Fe2+ have green colour and V4+ and Cr2+ have blue colour.
Among the following series of transition metal ions, the one where all metal ions have 3d2 electronic configuration is (Atomic number, Ti = 22, V = 23, Cr = 24, Mn = 25) [AIEEE 2004]- a)Ti3+, V2+, Cr3+, Mn4+
- b)Ti+, V4+, Cr6+, Mn7+
- c)Ti4+, V3+, Cr2+, Mn3+
- d)Ti2+, V3+, Cr4+, Mn5+
Correct answer is option 'D'. Can you explain this answer?
Among the following series of transition metal ions, the one where all metal ions have 3d2 electronic configuration is (Atomic number, Ti = 22, V = 23, Cr = 24, Mn = 25)
[AIEEE 2004]
a)
Ti3+, V2+, Cr3+, Mn4+
b)
Ti+, V4+, Cr6+, Mn7+
c)
Ti4+, V3+, Cr2+, Mn3+
d)
Ti2+, V3+, Cr4+, Mn5+
 | Samridhi Bajaj answered |
Ti2+,V3+,Cr4+ and Mn5+ show 1s22s22p63s23p64s03d2 electronic configuration.
Gun metal is an alloy of- a)Cu and Al
- b)Cu and Sn
- c)Cu,Zn and Sn
- d)Cu,Zn and Ni
Correct answer is option 'C'. Can you explain this answer?
Gun metal is an alloy of
a)
Cu and Al
b)
Cu and Sn
c)
Cu,Zn and Sn
d)
Cu,Zn and Ni
 | Sinjini Tiwari answered |
Gun metal is an alloy of Cu, Zn and Sn. It contains 88% Cu 10% Sn and 2% Zn.
In which of the following pairs both the ions are coloured in aqueous solution? (Atomic number, Sc = 21, Ti = 22, Ni = 28, Cu = 29, Co = 27)- a)Sc3+ and Ti3+
- b)Sc3+ and Co2+
- c)Ni2+ and Cu+
- d)Ni2+ and Ti3+
Correct answer is option 'D'. Can you explain this answer?
In which of the following pairs both the ions are coloured in aqueous solution? (Atomic number, Sc = 21, Ti = 22, Ni = 28, Cu = 29, Co = 27)
a)
Sc3+ and Ti3+
b)
Sc3+ and Co2+
c)
Ni2+ and Cu+
d)
Ni2+ and Ti3+
 | Gauri Sharma answered |
Ni2+ and Ti3+ ions are coloured in aqueous solution because they contain unpaired electrons.
Number of compounds in which metal has zero oxidation stateWO3,Ni(CO)4, MoO3, Fe(CO)5, Cr(CO)6, [Pt(NH3)2CI2], Co2(CO)8 and Mn2(CO)10
Correct answer is '5'. Can you explain this answer?
Number of compounds in which metal has zero oxidation state
WO3,Ni(CO)4, MoO3, Fe(CO)5, Cr(CO)6, [Pt(NH3)2CI2], Co2(CO)8 and Mn2(CO)10
 | Sinjini Tiwari answered |
Number of compounds in which metal has zero oxidation state
To determine the number of compounds in which the metal has a zero oxidation state, we need to analyze each compound individually and identify the oxidation state of the metal in each case.
1. WO3 (Tungsten trioxide)
In this compound, tungsten is in the +6 oxidation state. Therefore, the metal does not have a zero oxidation state.
2. Ni(CO)4 (Nickel tetracarbonyl)
In this compound, nickel is in the zero oxidation state. The oxidation state of carbon in carbon monoxide is -2, so the oxidation state of nickel must be zero to balance the charges. Therefore, this compound has a metal (nickel) with a zero oxidation state.
3. MoO3 (Molybdenum trioxide)
In this compound, molybdenum is in the +6 oxidation state. Therefore, the metal does not have a zero oxidation state.
4. Fe(CO)5 (Iron pentacarbonyl)
In this compound, iron is in the zero oxidation state. Similar to nickel tetracarbonyl, the oxidation state of carbon in carbon monoxide is -2, so the oxidation state of iron must be zero to balance the charges. Therefore, this compound has a metal (iron) with a zero oxidation state.
5. Cr(CO)6 (Chromium hexacarbonyl)
In this compound, chromium is in the zero oxidation state. Again, the oxidation state of carbon in carbon monoxide is -2, so the oxidation state of chromium must be zero to balance the charges. Therefore, this compound has a metal (chromium) with a zero oxidation state.
6. [Pt(NH3)2Cl2] (Diammine dichloridoplatinum(II))
In this compound, platinum is in the +2 oxidation state. Therefore, the metal does not have a zero oxidation state.
7. Co2(CO)8 (Dicobalt octacarbonyl)
In this compound, cobalt is in the zero oxidation state. The oxidation state of carbon in carbon monoxide is -2, so the oxidation state of cobalt must be zero to balance the charges. Therefore, this compound has a metal (cobalt) with a zero oxidation state.
8. Mn2(CO)10 (Dimanganese decacarbonyl)
In this compound, manganese is in the zero oxidation state. Similar to nickel tetracarbonyl and iron pentacarbonyl, the oxidation state of carbon in carbon monoxide is -2, so the oxidation state of manganese must be zero to balance the charges. Therefore, this compound has a metal (manganese) with a zero oxidation state.
Summary:
Out of the given compounds, the following five compounds have a metal with a zero oxidation state:
- Ni(CO)4 (Nickel tetracarbonyl)
- Fe(CO)5 (Iron pentacarbonyl)
- Cr(CO)6 (Chromium hexacarbonyl)
- Co2(CO)8 (Dicobalt octacarbonyl)
- Mn2(CO)10 (Dimanganese decacarbonyl)
To determine the number of compounds in which the metal has a zero oxidation state, we need to analyze each compound individually and identify the oxidation state of the metal in each case.
1. WO3 (Tungsten trioxide)
In this compound, tungsten is in the +6 oxidation state. Therefore, the metal does not have a zero oxidation state.
2. Ni(CO)4 (Nickel tetracarbonyl)
In this compound, nickel is in the zero oxidation state. The oxidation state of carbon in carbon monoxide is -2, so the oxidation state of nickel must be zero to balance the charges. Therefore, this compound has a metal (nickel) with a zero oxidation state.
3. MoO3 (Molybdenum trioxide)
In this compound, molybdenum is in the +6 oxidation state. Therefore, the metal does not have a zero oxidation state.
4. Fe(CO)5 (Iron pentacarbonyl)
In this compound, iron is in the zero oxidation state. Similar to nickel tetracarbonyl, the oxidation state of carbon in carbon monoxide is -2, so the oxidation state of iron must be zero to balance the charges. Therefore, this compound has a metal (iron) with a zero oxidation state.
5. Cr(CO)6 (Chromium hexacarbonyl)
In this compound, chromium is in the zero oxidation state. Again, the oxidation state of carbon in carbon monoxide is -2, so the oxidation state of chromium must be zero to balance the charges. Therefore, this compound has a metal (chromium) with a zero oxidation state.
6. [Pt(NH3)2Cl2] (Diammine dichloridoplatinum(II))
In this compound, platinum is in the +2 oxidation state. Therefore, the metal does not have a zero oxidation state.
7. Co2(CO)8 (Dicobalt octacarbonyl)
In this compound, cobalt is in the zero oxidation state. The oxidation state of carbon in carbon monoxide is -2, so the oxidation state of cobalt must be zero to balance the charges. Therefore, this compound has a metal (cobalt) with a zero oxidation state.
8. Mn2(CO)10 (Dimanganese decacarbonyl)
In this compound, manganese is in the zero oxidation state. Similar to nickel tetracarbonyl and iron pentacarbonyl, the oxidation state of carbon in carbon monoxide is -2, so the oxidation state of manganese must be zero to balance the charges. Therefore, this compound has a metal (manganese) with a zero oxidation state.
Summary:
Out of the given compounds, the following five compounds have a metal with a zero oxidation state:
- Ni(CO)4 (Nickel tetracarbonyl)
- Fe(CO)5 (Iron pentacarbonyl)
- Cr(CO)6 (Chromium hexacarbonyl)
- Co2(CO)8 (Dicobalt octacarbonyl)
- Mn2(CO)10 (Dimanganese decacarbonyl)
Chapter doubts & questions for d– and f–Block Elements - 6 Months Preparation for JEE 2026 is part of JEE exam preparation. The chapters have been prepared according to the JEE exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for JEE 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of d– and f–Block Elements - 6 Months Preparation for JEE in English & Hindi are available as part of JEE exam. Download more important topics, notes, lectures and mock test series for JEE Exam by signing up for free.
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup



