All Exams > CTET & State TET > 2 Months Preparation for CTET Paper 1 > All Questions
All questions of Water for CTET & State TET Exam
To avoid waterborne disease we should do- a)drink water after boiling
- b)add chlorine in water
- c)addition of lime water in water
- d)All of the above
Correct answer is option 'D'. Can you explain this answer?
a)
drink water after boiling
b)
add chlorine in water
c)
addition of lime water in water
d)
All of the above
 | Prof. Sonal Sharma answered |
Boiling kills germs and makes water safe for drinking. Chlorine helps disinfect water by destroying bacteria and microbes. Lime water can also help in cleaning, so all methods are useful.
Heavy water is- a)coolest
- b)moderator
- c)ore
- d)fuel
Correct answer is option 'B'. Can you explain this answer?
a)
coolest
b)
moderator
c)
ore
d)
fuel
 | Skyhigh Academy answered |
Heavy water is used as a neutron moderator in nuclear reactors. It slows down neutrons during nuclear reactions. Therefore, heavy water is called a moderator.
Rise of water level of oceans is due to- a)melting of ice of polar region
- b)excess rain water
- c)acid rain
- d)None of the above
Correct answer is option 'A'. Can you explain this answer?
a)
melting of ice of polar region
b)
excess rain water
c)
acid rain
d)
None of the above
 | Prof. Sonal Sharma answered |
Due to global warming, polar ice caps and glaciers are melting rapidly. This extra water increases the level of oceans and seas. Therefore, melting of polar ice is the main reason.
What is BOD5?- a)Biochemical Oxygen Demand in 5 hours
- b)Biochemical Oxygen Demand in 5 days
- c)Biochemical Oxygen Demand in 5 months
- d)Biochemical Oxygen Demand in 5 minutes
Correct answer is option 'B'. Can you explain this answer?
a)
Biochemical Oxygen Demand in 5 hours
b)
Biochemical Oxygen Demand in 5 days
c)
Biochemical Oxygen Demand in 5 months
d)
Biochemical Oxygen Demand in 5 minutes
 | Skyhigh Academy answered |
BOD5 measures oxygen consumed by microorganisms in 5 days. It is used to check organic pollution in water. Thus, BOD5 means Biochemical Oxygen Demand in 5 days.
Dry ice is- a)solid water
- b)solid CO₂
- c)solid H₂O₂
- d)None of these
Correct answer is option 'B'. Can you explain this answer?
a)
solid water
b)
solid CO₂
c)
solid H₂O₂
d)
None of these
 | Focus Academy answered |
Dry ice is the solid form of carbon dioxide. It changes directly from solid to gas without becoming liquid. Hence, dry ice is solid CO₂.
Pure water is- a)acidic
- b)basic
- c)neutral
- d)All of these
Correct answer is option 'C'. Can you explain this answer?
a)
acidic
b)
basic
c)
neutral
d)
All of these
 | Prof. Sonal Sharma answered |
Pure water has pH value of 7 which indicates neutrality. It is neither acidic nor basic. Hence, pure water is neutral.
Heavy water is used for- a)to form deuterium
- b)to study mechanism of chemical reaction
- c)used in nuclear reactors as moderator
- d)All of the above
Correct answer is option 'D'. Can you explain this answer?
a)
to form deuterium
b)
to study mechanism of chemical reaction
c)
used in nuclear reactors as moderator
d)
All of the above
 | Focus Academy answered |
Heavy water is used to produce deuterium. It is also used as a tracer to study chemical and biochemical reactions. Additionally, it acts as a moderator in nuclear reactors, so all are correct.
After addition of salt in the water- a)boiling point increases
- b)boiling point decreases
- c)no effect on boiling point
- d)freezing point increases
Correct answer is option 'A'. Can you explain this answer?
a)
boiling point increases
b)
boiling point decreases
c)
no effect on boiling point
d)
freezing point increases
 | Skyhigh Academy answered |
Salt raises the boiling point of water by increasing its boiling temperature. This happens because dissolved salt makes it harder for water molecules to escape as vapour. Therefore, boiling point increases.
What we can do to make water potable?- a)boiling
- b)add potassium permanganate
- c)addition of bleaching powder
- d)All of the above
Correct answer is option 'D'. Can you explain this answer?
a)
boiling
b)
add potassium permanganate
c)
addition of bleaching powder
d)
All of the above
 | Focus Academy answered |
Boiling kills harmful microorganisms present in water. Potassium permanganate cleans well water and bleaching powder disinfects water. Therefore, all these steps help to make water potable.
Heavy water can be formed by- a)electrolysis of simple water
- b)mixing heavy salt in water
- c)distillation of water
- d)separation of impurities of Ca and Mg
Correct answer is option 'A'. Can you explain this answer?
a)
electrolysis of simple water
b)
mixing heavy salt in water
c)
distillation of water
d)
separation of impurities of Ca and Mg
 | Skyhigh Academy answered |
Heavy water is obtained by electrolysis of normal water. It contains deuterium instead of hydrogen. Hence, electrolysis is the method used to form heavy water.
Density of water is maximum at- a)below 4°C
- b)at 4°C
- c)above 4°C
- d)at 0°C
Correct answer is option 'B'. Can you explain this answer?
a)
below 4°C
b)
at 4°C
c)
above 4°C
d)
at 0°C
 | Focus Academy answered |
Water has a special property of maximum density at 4°C. At this temperature, water occupies least volume and becomes heaviest. This helps aquatic life survive in cold regions.
Fishes are able to live even if lake’s upper layer becomes ice- a)density of water is less
- b)density of ice is more
- c)density of water of lake is more
- d)All of the above
Correct answer is option 'C'. Can you explain this answer?
a)
density of water is less
b)
density of ice is more
c)
density of water of lake is more
d)
All of the above
 | Focus Academy answered |
Water is densest at 4°C and stays at the bottom of lakes. Ice floats on the top layer and forms an insulating cover. This allows aquatic life to survive in the lower liquid water.
Characteristic of pure water is- a)presence of harmful microorganisms
- b)impure and odorous water
- c)presence of chemicals
- d)presence of oxygen in adequate amount
Correct answer is option 'D'. Can you explain this answer?
a)
presence of harmful microorganisms
b)
impure and odorous water
c)
presence of chemicals
d)
presence of oxygen in adequate amount
 | Prof. Sonal Sharma answered |
Pure water is clean, colourless and free from harmful impurities. It should contain adequate dissolved oxygen for supporting aquatic life. Hence, presence of oxygen in adequate amount is correct.
Drinkable water on Earth is- a)97.2%
- b)2.15%
- c)0.65%
- d)0.0%
Correct answer is option 'D'. Can you explain this answer?
a)
97.2%
b)
2.15%
c)
0.65%
d)
0.0%
 | Prof. Sonal Sharma answered |
Most of the Earth’s water is salty and not drinkable. Only a very small part of fresh water is available for human use. Hence, drinkable water is nearly 0.0% when compared to total water.
In summers, wet clothes dry very fastly due to- a)condensation
- b)evaporation
- c)Neither (A) nor (B)
- d)Both (A) and (B)
Correct answer is option 'B'. Can you explain this answer?
a)
condensation
b)
evaporation
c)
Neither (A) nor (B)
d)
Both (A) and (B)
 | Prof. Sonal Sharma answered |
Heat in summer increases the rate of evaporation. Water from wet clothes turns into vapour quickly and escapes into air. Therefore, clothes dry fast due to evaporation.
Consider the following statements
I. Heavy water is radioactive in nature.
II. Heavy water is formed by electrolysis of normal water.
III. Heavy water is not harmful for organisms.
Which statement(s) is/are incorrect?- a)Only II
- b)I and II
- c)I, II and III
- d)III and I
Correct answer is option 'D'. Can you explain this answer?
Consider the following statements
I. Heavy water is radioactive in nature.
II. Heavy water is formed by electrolysis of normal water.
III. Heavy water is not harmful for organisms.
Which statement(s) is/are incorrect?
I. Heavy water is radioactive in nature.
II. Heavy water is formed by electrolysis of normal water.
III. Heavy water is not harmful for organisms.
Which statement(s) is/are incorrect?
a)
Only II
b)
I and II
c)
I, II and III
d)
III and I
 | Focus Academy answered |
Heavy water is not radioactive, so statement I is incorrect. Heavy water can be formed by electrolysis, so statement II is correct. Heavy water is harmful to organisms, so statement III is incorrect.
Which one is correct?- a)Evaporation → Condensation → Cloud → Rain
- b)Cloud → Condensation → Evaporation → Rain
- c)Evaporation → Rain → Condensation → Cloud
- d)Cloud → Rain → Evaporation → Condensation
Correct answer is option 'A'. Can you explain this answer?
a)
Evaporation → Condensation → Cloud → Rain
b)
Cloud → Condensation → Evaporation → Rain
c)
Evaporation → Rain → Condensation → Cloud
d)
Cloud → Rain → Evaporation → Condensation
 | Prof. Sonal Sharma answered |
Water evaporates due to heat and changes into water vapour. Vapour cools and condenses to form clouds and later falls as rain. Thus, the correct sequence is evaporation, condensation, cloud formation and rain.
Consider the following statements about water
I. Water helps aquatic organisms survive because it dissolves nutrients and oxygen.
II. Water allows light to pass through it, due to which aquatic organisms are able to see.
Which statement(s) is/are correct?- a)Only I
- b)Only II
- c)Both I and II
- d)Neither I nor II
Correct answer is option 'C'. Can you explain this answer?
Consider the following statements about water
I. Water helps aquatic organisms survive because it dissolves nutrients and oxygen.
II. Water allows light to pass through it, due to which aquatic organisms are able to see.
Which statement(s) is/are correct?
I. Water helps aquatic organisms survive because it dissolves nutrients and oxygen.
II. Water allows light to pass through it, due to which aquatic organisms are able to see.
Which statement(s) is/are correct?
a)
Only I
b)
Only II
c)
Both I and II
d)
Neither I nor II
 | Prof. Sonal Sharma answered |
Water dissolves oxygen and nutrients needed by aquatic organisms. It also allows sunlight to pass which helps aquatic animals in visibility and survival. Hence, both statements are correct.
Ocean cover is- a)3/4
- b)1/2
- c)1/4
- d)2/3
Correct answer is option 'A'. Can you explain this answer?
a)
3/4
b)
1/2
c)
1/4
d)
2/3
 | Skyhigh Academy answered |
About 71% of Earth’s surface is covered with water. This is nearly equal to three-fourth of the surface. Therefore, ocean cover is 3/4.
Traces of radioactive waste present in water can cause- a)cancer
- b)eye cataract
- c)DNA breakage
- d)All of these
Correct answer is option 'D'. Can you explain this answer?
a)
cancer
b)
eye cataract
c)
DNA breakage
d)
All of these
 | Aspire Academy answered |
Radioactive waste damages cells and tissues in the body. It can cause cancer, cataract and genetic damage like DNA breakage. Hence, all given effects are correct.
Consider the following statements
I. Boiling needs external heat.
II. Evaporation needs external heat.
III. Atmospheric heat is sufficient for evaporation.
IV. Evaporation is highest at boiling point.
Which statements are correct?- a)II, III and IV
- b)I and II
- c)I, III and IV
- d)All of these
Correct answer is option 'C'. Can you explain this answer?
Consider the following statements
I. Boiling needs external heat.
II. Evaporation needs external heat.
III. Atmospheric heat is sufficient for evaporation.
IV. Evaporation is highest at boiling point.
Which statements are correct?
I. Boiling needs external heat.
II. Evaporation needs external heat.
III. Atmospheric heat is sufficient for evaporation.
IV. Evaporation is highest at boiling point.
Which statements are correct?
a)
II, III and IV
b)
I and II
c)
I, III and IV
d)
All of these
 | Prof. Sonal Sharma answered |
Boiling requires extra heat supply, so statement I is correct. Evaporation does not require external heat as atmospheric heat is enough. Evaporation is fastest at boiling point, so III and IV are correct.
Boiling point of water in Fahrenheit scale- a)100°F
- b)0°F
- c)212°F
- d)132°F
Correct answer is option 'C'. Can you explain this answer?
a)
100°F
b)
0°F
c)
212°F
d)
132°F
 | Prof. Sonal Sharma answered |
Water boils at 100°C under normal pressure. This temperature corresponds to 212°F on the Fahrenheit scale. Therefore, 212°F is the boiling point.
Most salty sea is- a)Arab Sea
- b)Red Sea
- c)Dead Sea
- d)Aral Sea
Correct answer is option 'C'. Can you explain this answer?
a)
Arab Sea
b)
Red Sea
c)
Dead Sea
d)
Aral Sea
 | Skyhigh Academy answered |
Dead Sea contains extremely high salt concentration in its water. Due to this, people can float easily without swimming. Hence, Dead Sea is the saltiest.
Which salt is present in maximum amount in seas?- a)Magnesium chloride
- b)Calcium chloride
- c)Magnesium sulphate
- d)Sodium chloride
Correct answer is option 'D'. Can you explain this answer?
a)
Magnesium chloride
b)
Calcium chloride
c)
Magnesium sulphate
d)
Sodium chloride
 | Prof. Sonal Sharma answered |
Sea water contains many dissolved salts but sodium chloride is the highest. This is the common salt used in households. Therefore, sodium chloride is present in maximum amount.
Main causes of acid rain is- a)presence of nitric acid
- b)presence of chloric acid
- c)presence of sulphuric acid
- d)Both (A) and (C)
Correct answer is option 'D'. Can you explain this answer?
a)
presence of nitric acid
b)
presence of chloric acid
c)
presence of sulphuric acid
d)
Both (A) and (C)
 | Focus Academy answered |
Acid rain occurs due to presence of nitric acid and sulphuric acid in rainwater. These acids are formed due to pollution from industries and vehicles. Hence, both nitric and sulphuric acids are the main causes.
Water pollution is- a)presence of undesirable material in water
- b)presence of sewage in water
- c)presence of chemical fertiliser in water
- d)All of the above
Correct answer is option 'D'. Can you explain this answer?
a)
presence of undesirable material in water
b)
presence of sewage in water
c)
presence of chemical fertiliser in water
d)
All of the above
 | Focus Academy answered |
Water pollution includes sewage, chemicals and harmful materials mixing into water. These substances make water unsafe for drinking and use. Hence, all the given options are causes of water pollution.
Consider the following statements
I. Boiling point of water is 273 K.
II. Evaporation takes place at all temperature.
Which of the statement(s) given above is/are correct?- a)Only I
- b)Only II
- c)Both I and II
- d)Neither I nor II
Correct answer is option 'B'. Can you explain this answer?
Consider the following statements
I. Boiling point of water is 273 K.
II. Evaporation takes place at all temperature.
Which of the statement(s) given above is/are correct?
I. Boiling point of water is 273 K.
II. Evaporation takes place at all temperature.
Which of the statement(s) given above is/are correct?
a)
Only I
b)
Only II
c)
Both I and II
d)
Neither I nor II
 | Skyhigh Academy answered |
Boiling point of water is 373 K, not 273 K, so statement I is incorrect. Evaporation can occur at all temperatures but at different rates. Hence, only statement II is correct.
The Ganga Action Plan launched to improve the- a)water quality
- b)water velocity
- c)water quantity
- d)water temperature
Correct answer is option 'A'. Can you explain this answer?
a)
water quality
b)
water velocity
c)
water quantity
d)
water temperature
 | Aspire Academy answered |
The Ganga Action Plan was launched to reduce pollution levels in river Ganga. Its main goal is improving cleanliness and water quality. Hence, the correct answer is water quality.
Due to boiling of the impure or hard water we observe some settled substance, what it may be?- a)bicarbonate of calcium
- b)bicarbonate of magnesium
- c)Both (A) and (B)
- d)Neither (A) nor (B)
Correct answer is option 'C'. Can you explain this answer?
a)
bicarbonate of calcium
b)
bicarbonate of magnesium
c)
Both (A) and (B)
d)
Neither (A) nor (B)
 | Prof. Sonal Sharma answered |
Boiling converts soluble bicarbonates into insoluble carbonates. These carbonates settle at the bottom as deposits. Hence both calcium and magnesium bicarbonates can form the settled substance.
Hard water is impure due to presence of- a)bicarbonates of calcium
- b)chlorides of calcium
- c)bicarbonates of magnesium
- d)All of the above
Correct answer is option 'D'. Can you explain this answer?
a)
bicarbonates of calcium
b)
chlorides of calcium
c)
bicarbonates of magnesium
d)
All of the above
 | Prof. Sonal Sharma answered |
Hard water contains salts of calcium and magnesium. These can be bicarbonates and chlorides which make water hard. Hence, all the given substances cause hardness.
Which country has maximum number of tube-wells in the world?- a)America
- b)Australia
- c)China
- d)India
Correct answer is option 'D'. Can you explain this answer?
a)
America
b)
Australia
c)
China
d)
India
 | Skyhigh Academy answered |
India has extensive use of tube-well irrigation for agriculture. It is considered the highest tube-well user among countries. Hence, India has maximum tube-wells in the world.
Source of fresh water are- a)sea
- b)salty lakes
- c)underground water
- d)oceans
Correct answer is option 'C'. Can you explain this answer?
a)
sea
b)
salty lakes
c)
underground water
d)
oceans
 | Focus Academy answered |
Fresh water is found in rivers, lakes, glaciers and underground sources. Seas and oceans contain saline water which is not suitable for drinking. Therefore, underground water is a correct source of fresh water.
When cold water is poured into the glass, water droplets appear on outer surface due to- a)liquification
- b)evaporation
- c)Both (A) and (B)
- d)Neither (A) nor (B)
Correct answer is option 'A'. Can you explain this answer?
a)
liquification
b)
evaporation
c)
Both (A) and (B)
d)
Neither (A) nor (B)
 | Focus Academy answered |
Cold surface cools the surrounding air and water vapour condenses on the glass. This forms small droplets on the outer surface. This process is called liquification or condensation.
How much percentage of water in the oceans is drinkable?- a)97.2
- b)2.15
- c)0.65
- d)0.0
Correct answer is option 'D'. Can you explain this answer?
a)
97.2
b)
2.15
c)
0.65
d)
0.0
 | Skyhigh Academy answered |
Ocean water contains high salt content and cannot be consumed directly. Therefore, ocean water is not drinkable. Hence, the drinkable percentage is 0.0.
Water conservation can be done by- a)control on soil erosion
- b)reforestation
- c)to stop wastage of water
- d)All of the above
Correct answer is option 'D'. Can you explain this answer?
a)
control on soil erosion
b)
reforestation
c)
to stop wastage of water
d)
All of the above
 | Skyhigh Academy answered |
Soil erosion control helps retain water in the ground. Reforestation improves rainfall and groundwater recharge. Stopping water wastage is also essential, so all methods help in water conservation.
Minamata disease is caused by mixing of ______ material in water.- a)cadmium
- b)mercury
- c)iron
- d)sodium
Correct answer is option 'B'. Can you explain this answer?
a)
cadmium
b)
mercury
c)
iron
d)
sodium
 | Skyhigh Academy answered |
Minamata disease is caused by mercury poisoning through contaminated water. It affects the nervous system severely. Hence, mercury is responsible for Minamata disease.
Main pollutants of underground water is- a)arsenic and fluoride
- b)ammonia
- c)nitrates
- d)chromium and chlorides
Correct answer is option 'A'. Can you explain this answer?
a)
arsenic and fluoride
b)
ammonia
c)
nitrates
d)
chromium and chlorides
 | Skyhigh Academy answered |
Underground water is polluted mainly by arsenic and fluoride in many regions. These contaminants make water harmful for human consumption. Therefore, arsenic and fluoride are major pollutants.
City of Lakes in Rajasthan is- a)Jaipur
- b)Jodhpur
- c)Udaipur
- d)Ajmer
Correct answer is option 'C'. Can you explain this answer?
a)
Jaipur
b)
Jodhpur
c)
Udaipur
d)
Ajmer
 | Focus Academy answered |
Udaipur is famous for its beautiful lakes like Pichola and Fateh Sagar. It is also called the "City of Lakes" in Rajasthan. Therefore, Udaipur is the correct answer.
Waterborne disease is- a)dysentery
- b)small pox
- c)anaemia
- d)brain fever
Correct answer is option 'A'. Can you explain this answer?
a)
dysentery
b)
small pox
c)
anaemia
d)
brain fever
 | Aspire Academy answered |
Dysentery spreads through contaminated food and water. It is caused by harmful microbes that enter the digestive system. Therefore, dysentery is a waterborne disease.
Main causes of water pollution- a)pesticides and insecticides used in agricultural practices
- b)industrial waste addition in rivers
- c)accidental oil leakages
- d)All of the above
Correct answer is option 'D'. Can you explain this answer?
a)
pesticides and insecticides used in agricultural practices
b)
industrial waste addition in rivers
c)
accidental oil leakages
d)
All of the above
 | Prof. Sonal Sharma answered |
Agricultural chemicals like pesticides contaminate water sources. Industrial waste releases harmful substances into rivers and lakes. Oil leakage also pollutes marine water, so all options are correct.
Match the following
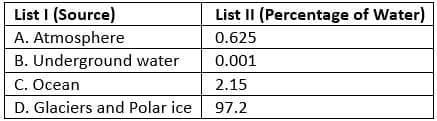
Codes- a)1, 2, 3, 4
- b)4, 3, 2, 1
- c)2, 1, 4, 3
- d)3, 2, 1, 4
Correct answer is option 'C'. Can you explain this answer?
Match the following

Codes

Codes
a)
1, 2, 3, 4
b)
4, 3, 2, 1
c)
2, 1, 4, 3
d)
3, 2, 1, 4
 | Prof. Sonal Sharma answered |
Oceans contain maximum water percentage and atmosphere contains very little water. Underground water and glaciers have smaller percentages compared to oceans. Thus, correct matching corresponds to option C.
Freezing point of water is- a)0°C
- b)32°F
- c)100°F
- d)Both (A) and (B)
Correct answer is option 'D'. Can you explain this answer?
a)
0°C
b)
32°F
c)
100°F
d)
Both (A) and (B)
 | Skyhigh Academy answered |
Water freezes at 0°C on the Celsius scale. The same temperature is 32°F on the Fahrenheit scale. Hence both values correctly represent the freezing point.
For cleaning of water we have to do- a)filtration
- b)boiling
- c)chlorination
- d)All of these
Correct answer is option 'D'. Can you explain this answer?
a)
filtration
b)
boiling
c)
chlorination
d)
All of these
 | Skyhigh Academy answered |
Filtration removes suspended particles from water. Boiling kills microorganisms and makes water safer. Chlorination also kills germs, so all methods are correct.
Heavy water is- a)D₂O
- b)H₂O
- c)N₂O
- d)P₂O
Correct answer is option 'A'. Can you explain this answer?
a)
D₂O
b)
H₂O
c)
N₂O
d)
P₂O
 | Focus Academy answered |
Heavy water is written as D₂O where D represents deuterium. It is different from normal water which is H₂O. Hence, D₂O is the correct formula of heavy water.
In hilly areas, water pipelines burst during winter because- a)water expands after freezing
- b)water squeezes after freezing
- c)pipelines squeeze at freezing
- d)None of the above
Correct answer is option 'A'. Can you explain this answer?
a)
water expands after freezing
b)
water squeezes after freezing
c)
pipelines squeeze at freezing
d)
None of the above
 | Prof. Sonal Sharma answered |
When water freezes, it expands in volume. This expansion puts pressure on pipelines and may cause them to burst. Hence, the reason is expansion of water after freezing.
Water in earthen pot remains cool due to- a)condensation
- b)evaporation
- c)sedimentation
- d)sublimation
Correct answer is option 'B'. Can you explain this answer?
a)
condensation
b)
evaporation
c)
sedimentation
d)
sublimation
 | Aspire Academy answered |
Earthen pots have tiny pores through which water slowly seeps out. This water evaporates from the surface and takes heat from the pot. Therefore, water remains cool due to evaporation.
Main cause of water level depletion is- a)urbanisation
- b)industrialisation
- c)global warming
- d)acid rain
Correct answer is option 'A'. Can you explain this answer?
a)
urbanisation
b)
industrialisation
c)
global warming
d)
acid rain
 | Focus Academy answered |
Urbanisation increases groundwater use and reduces natural recharge areas. Concrete surfaces prevent rainwater from entering soil. Hence, urbanisation is the main cause of water level depletion.
71% Earth is covered by- a)land
- b)air
- c)water
- d)coal
Correct answer is option 'C'. Can you explain this answer?
a)
land
b)
air
c)
water
d)
coal
 | Aspire Academy answered |
Earth is known as the blue planet due to abundance of water. Around 71% of its surface is covered by oceans, seas and other water sources. Hence, the correct answer is water.
BOD is- a)Biological Optimal Demand
- b)Biological Optimal Decide
- c)Biological Oxygen Demand
- d)None of the above
Correct answer is option 'C'. Can you explain this answer?
a)
Biological Optimal Demand
b)
Biological Optimal Decide
c)
Biological Oxygen Demand
d)
None of the above
 | Focus Academy answered |
BOD stands for Biological Oxygen Demand. It measures oxygen used by microorganisms to decompose organic matter in water. Higher BOD indicates more polluted water.
Salt, which absorb water is called- a)hygroscopic salt
- b)hydrophilic salt
- c)anhydrous salt
- d)hydrophobic salt
Correct answer is option 'B'. Can you explain this answer?
a)
hygroscopic salt
b)
hydrophilic salt
c)
anhydrous salt
d)
hydrophobic salt
 | Prof. Sonal Sharma answered |
Hydrophilic substances are those that attract water molecules. Such salts absorb water due to their nature of bonding with water. Therefore, salt that absorbs water is called hydrophilic salt.
Chapter doubts & questions for Water - 2 Months Preparation for CTET Paper 1 2026 is part of CTET & State TET exam preparation. The chapters have been prepared according to the CTET & State TET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for CTET & State TET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Water - 2 Months Preparation for CTET Paper 1 in English & Hindi are available as part of CTET & State TET exam. Download more important topics, notes, lectures and mock test series for CTET & State TET Exam by signing up for free.
2 Months Preparation for CTET Paper 1218 videos|569 docs|217 tests |
Top Courses CTET & State TET
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup

