All Exams > UPSC > 6 Months Preparation Course for UPSC EPFO > All Questions
All questions of Physics for UPSC CSE Exam
Food gets cooked faster in a pressure cooker because- a)water starts boiling at a lower temperature due to high pressure
- b)water starts boiling at a higher temperature due to high pressure
- c)water boils only at 100°C but the heat content is higher at high pressure
- d)convection currents are set inside the cooker
Correct answer is option 'A'. Can you explain this answer?
Food gets cooked faster in a pressure cooker because
a)
water starts boiling at a lower temperature due to high pressure
b)
water starts boiling at a higher temperature due to high pressure
c)
water boils only at 100°C but the heat content is higher at high pressure
d)
convection currents are set inside the cooker
 | Anjana Choudhary answered |
Because water starts boiling at a lower temperature due to high pressure. According to Gay-Lussac’s law ρ ∝ T.
Which one of the following statements is not true for a person suffering from hypermetropia?- a)The person can see far objects distinctly.
- b)The focal length of the lens is large.
- c)The image of the close object is focussed behind the retina.
- d)A concave lens is used to correct this defect.
Correct answer is option 'D'. Can you explain this answer?
Which one of the following statements is not true for a person suffering from hypermetropia?
a)
The person can see far objects distinctly.
b)
The focal length of the lens is large.
c)
The image of the close object is focussed behind the retina.
d)
A concave lens is used to correct this defect.
 | Janhavi Dey answered |
Hypermetropia is corrected by using a concave lens. Myopia is corrected by convex lens.
An iron needle sinks in water whereas a ship made of iron floats on it because- a)the edge of the needle is pointed
- b)the ship is flat
- c)the ship is driven by powerful engine
- d)specific gravity of the needle is greater than that of water displaced by it.
Correct answer is option 'D'. Can you explain this answer?
An iron needle sinks in water whereas a ship made of iron floats on it because
a)
the edge of the needle is pointed
b)
the ship is flat
c)
the ship is driven by powerful engine
d)
specific gravity of the needle is greater than that of water displaced by it.
 | Kajal Shah answered |
Because the specific gravity of the needle is greater than that of water displaced by it. Specific gravity (or relative density) = Density of the substance / Density of the water at 4°C The average density of iron needle is greater than water, therefore, it sinks.
A fluorescent tube is preferred to an electric bulb because- a)it has a larger light emitting surface.
- b)voltage fluctuations do not affect it.
- c)in a tube electrical energy is almost converted into light.
- d)None of these
Correct answer is option 'C'. Can you explain this answer?
A fluorescent tube is preferred to an electric bulb because
a)
it has a larger light emitting surface.
b)
voltage fluctuations do not affect it.
c)
in a tube electrical energy is almost converted into light.
d)
None of these
 | Simran Sarkar answered |
In a fluorescent tube, electrical energy is almost converted into light. The inner wall of the fluorescent tube is coated with phosphorus which immediately transformed into bright light.
The cloudy nights are warmer than clear nights because- a)clouds prevent escape of radiation of heat from the ground and the air.
- b)absorb sunlight in the day and radiate the same in night.
- c)clouds make the atmosphere damp and generate heat.
- d)clouds obstruct the movement of air which creates heat.
Correct answer is option 'A'. Can you explain this answer?
The cloudy nights are warmer than clear nights because
a)
clouds prevent escape of radiation of heat from the ground and the air.
b)
absorb sunlight in the day and radiate the same in night.
c)
clouds make the atmosphere damp and generate heat.
d)
clouds obstruct the movement of air which creates heat.
| | Jatin Ghosh answered |
The cloudy nights are warmer because clouds prevent escape of radiation of heat from the ground and the air.
What did the early Universe contain before the formation of atoms?- a)Galaxies
- b)Photons and free electrons
- c)Dark matter
- d)Planets
Correct answer is option 'B'. Can you explain this answer?
What did the early Universe contain before the formation of atoms?
a)
Galaxies
b)
Photons and free electrons
c)
Dark matter
d)
Planets
| | Kavita Shah answered |
The early Universe contained photons (light) as elementary particles and free electrons.
What were the majority of atoms produced during the Big Bang?- a)Hydrogen
- b)Helium
- c)Oxygen
- d)Carbon
Correct answer is option 'A'. Can you explain this answer?
What were the majority of atoms produced during the Big Bang?
a)
Hydrogen
b)
Helium
c)
Oxygen
d)
Carbon
| | Rashi Mukherjee answered |
Overview of Big Bang Nucleosynthesis
The Big Bang theory explains the formation of the universe and the primary elements produced in its early moments, known as Big Bang nucleosynthesis. This process occurred within the first few minutes after the Big Bang when the universe was hot and dense.
Production of Light Elements
During this initial phase, the extreme temperatures and pressures facilitated nuclear reactions, leading to the formation of the lightest elements. The predominant atoms produced were:
Why Hydrogen Dominates
The dominance of hydrogen is due to several factors:
Conclusion
In summary, while helium was also produced in significant quantities, the overwhelming majority of atoms formed during the Big Bang were hydrogen. This abundance laid the foundation for later star formation and the synthesis of heavier elements in stellar processes.
The Big Bang theory explains the formation of the universe and the primary elements produced in its early moments, known as Big Bang nucleosynthesis. This process occurred within the first few minutes after the Big Bang when the universe was hot and dense.
Production of Light Elements
During this initial phase, the extreme temperatures and pressures facilitated nuclear reactions, leading to the formation of the lightest elements. The predominant atoms produced were:
- Hydrogen: Approximately 75% of the universe's baryonic mass is composed of hydrogen. Protons were formed from quarks, and these protons became the basic building blocks of hydrogen.
- Helium: Roughly 25% of the mass was converted into helium, primarily in the form of helium-4 nuclei, as nuclear fusion occurred among protons and neutrons.
- Trace Elements: Minor amounts of lithium and beryllium were formed, but the quantities were significantly less than hydrogen and helium.
Why Hydrogen Dominates
The dominance of hydrogen is due to several factors:
- Simple Structure: Hydrogen, being the simplest element, requires the least energy to form. The fusion process favored its production over heavier elements.
- Stability: Hydrogen nuclei (protons) are stable and do not undergo further fusion under the conditions present at the time, allowing them to remain abundant.
Conclusion
In summary, while helium was also produced in significant quantities, the overwhelming majority of atoms formed during the Big Bang were hydrogen. This abundance laid the foundation for later star formation and the synthesis of heavier elements in stellar processes.
If a rock is brought from the surface of the moon to the earth, then- a)its mass will change.
- b)its weight will change but not its mass.
- c)both mass and weight will change.
- d)both mass and weight will remain the same.
Correct answer is option 'B'. Can you explain this answer?
If a rock is brought from the surface of the moon to the earth, then
a)
its mass will change.
b)
its weight will change but not its mass.
c)
both mass and weight will change.
d)
both mass and weight will remain the same.
 | Anjana Choudhary answered |
If a rock is brought from the surface of the moon to the earth, its weight will change but not its mass. Mass is a invariant physical quantity whereas weight of a body (w = mg) is variable as the value of acceleration due to gravity (g) changes.
What is the significance of studying the Cosmic Background Radiation?- a)To understand the expansion of galaxies.
- b)To learn about dark matter's properties.
- c)To gain insights into the early stages of the Universe.
- d)To study the formation of planets.
Correct answer is option 'C'. Can you explain this answer?
What is the significance of studying the Cosmic Background Radiation?
a)
To understand the expansion of galaxies.
b)
To learn about dark matter's properties.
c)
To gain insights into the early stages of the Universe.
d)
To study the formation of planets.
| | Kavita Shah answered |
Studying the Cosmic Background Radiation provides valuable insights into the early stages of the Universe and its evolution.
The leaning tower of Pisa does not fall because- a)it is tappered at the top.
- b)it covers a large base area.
- c)its centre of gravity remains at the lowest position.
- d)the vertical line through the centre of gravity of the tower falls within the base.
Correct answer is option 'D'. Can you explain this answer?
The leaning tower of Pisa does not fall because
a)
it is tappered at the top.
b)
it covers a large base area.
c)
its centre of gravity remains at the lowest position.
d)
the vertical line through the centre of gravity of the tower falls within the base.
 | Janhavi Dey answered |
Because the vertical line through the centre of gravity of the tower falls within the base.
If the doors of a refrigerator are left open for a few hours, the room temperature will- a)decrease
- b)increase
- c)remain the same
- d)decrease only in the area in the vicinity of the refrigerator
Correct answer is option 'B'. Can you explain this answer?
If the doors of a refrigerator are left open for a few hours, the room temperature will
a)
decrease
b)
increase
c)
remain the same
d)
decrease only in the area in the vicinity of the refrigerator
 | Pallavi Chakraborty answered |
If the doors of a refrigerator are left open for a few hours, the room temperature will increase as the system release heat to the surroundings.
What is the overall shape of the Universe according to scientific observations?- a)Spherical
- b)Cylindrical
- c)Flat
- d)Conical
Correct answer is option 'C'. Can you explain this answer?
What is the overall shape of the Universe according to scientific observations?
a)
Spherical
b)
Cylindrical
c)
Flat
d)
Conical
| | Maheshwar Jain answered |
Flat Shape of the Universe:
The overall shape of the Universe according to scientific observations is considered to be flat. This conclusion is based on a variety of observations and measurements made by astronomers and cosmologists.
Cosmic Microwave Background Radiation:
One of the key pieces of evidence supporting the flat shape of the Universe comes from the Cosmic Microwave Background Radiation (CMB). The fluctuations in the CMB provide valuable information about the geometry of the Universe, and the data collected indicates that the Universe is flat.
Measurements of Large-Scale Structure:
Observations of the large-scale structure of the Universe, including the distribution of galaxies and galaxy clusters, also support the idea that the Universe is flat. These measurements suggest that the overall geometry of the Universe is consistent with a flat shape.
General Relativity:
The theory of General Relativity, proposed by Albert Einstein, also supports the idea of a flat Universe. According to General Relativity, the geometry of the Universe is determined by the distribution of matter and energy, and in a flat Universe, the total energy density is equal to the critical density.
Conclusion:
In conclusion, based on observations of the CMB, measurements of the large-scale structure of the Universe, and the predictions of General Relativity, scientists have determined that the overall shape of the Universe is flat. This has significant implications for our understanding of the cosmos and the fundamental nature of the Universe.
What is the Big Bang Theory?- a)A theory proposing the existence of multiple universes.
- b)A theory explaining the origin of the universe through a massive explosion.
- c)A theory suggesting that the universe has always existed in its current state.
- d)A theory explaining the formation of stars and galaxies.
Correct answer is option 'B'. Can you explain this answer?
What is the Big Bang Theory?
a)
A theory proposing the existence of multiple universes.
b)
A theory explaining the origin of the universe through a massive explosion.
c)
A theory suggesting that the universe has always existed in its current state.
d)
A theory explaining the formation of stars and galaxies.
| | Maitri Singh answered |
The Big Bang Theory:
The Big Bang Theory is a scientific explanation for the origin and evolution of the universe. It proposes that the universe began as an extremely hot and dense point, often referred to as a singularity, and has been expanding ever since. This theory is widely accepted by the scientific community and is supported by various lines of evidence.
Explanation:
1. Origin of the Universe:
The Big Bang Theory suggests that approximately 13.8 billion years ago, all matter and energy in the universe was concentrated in a tiny, infinitely dense point called a singularity. This singularity began to rapidly expand, resulting in the formation of the universe as we know it.
2. Expansion of the Universe:
According to the theory, the universe is constantly expanding. This expansion is evidenced by the observation of distant galaxies moving away from us. The expansion of the universe can be visualized by imagining dots on the surface of a balloon. As the balloon is inflated, the dots move away from each other, just like galaxies in our universe.
3. Background Radiation:
One of the key pieces of evidence supporting the Big Bang Theory is the discovery of cosmic microwave background radiation. This radiation is a faint glow of energy that permeates the entire universe and is thought to be the afterglow of the initial explosion. Its existence and characteristics align with the predictions made by the theory.
4. Abundance of Light Elements:
The Big Bang Theory also explains the abundance of light elements, such as hydrogen and helium, in the universe. It suggests that during the early moments of the universe, the extremely high temperatures and pressures allowed for the formation of these elements. Subsequent processes, such as nuclear fusion in stars, led to the creation of heavier elements.
5. Observable Universe:
The Big Bang Theory provides an explanation for the observed distribution and structure of galaxies in the universe. As the universe expanded, matter began to clump together under the influence of gravity, forming structures like galaxies, stars, and planets. The theory predicts the large-scale structure of the universe, including the existence of galaxy clusters and cosmic filaments.
Overall, the Big Bang Theory is a comprehensive scientific explanation for the origin and evolution of the universe. It provides a framework for understanding the vastness and complexity of the cosmos and is supported by numerous observational and theoretical evidence.
The Big Bang Theory is a scientific explanation for the origin and evolution of the universe. It proposes that the universe began as an extremely hot and dense point, often referred to as a singularity, and has been expanding ever since. This theory is widely accepted by the scientific community and is supported by various lines of evidence.
Explanation:
1. Origin of the Universe:
The Big Bang Theory suggests that approximately 13.8 billion years ago, all matter and energy in the universe was concentrated in a tiny, infinitely dense point called a singularity. This singularity began to rapidly expand, resulting in the formation of the universe as we know it.
2. Expansion of the Universe:
According to the theory, the universe is constantly expanding. This expansion is evidenced by the observation of distant galaxies moving away from us. The expansion of the universe can be visualized by imagining dots on the surface of a balloon. As the balloon is inflated, the dots move away from each other, just like galaxies in our universe.
3. Background Radiation:
One of the key pieces of evidence supporting the Big Bang Theory is the discovery of cosmic microwave background radiation. This radiation is a faint glow of energy that permeates the entire universe and is thought to be the afterglow of the initial explosion. Its existence and characteristics align with the predictions made by the theory.
4. Abundance of Light Elements:
The Big Bang Theory also explains the abundance of light elements, such as hydrogen and helium, in the universe. It suggests that during the early moments of the universe, the extremely high temperatures and pressures allowed for the formation of these elements. Subsequent processes, such as nuclear fusion in stars, led to the creation of heavier elements.
5. Observable Universe:
The Big Bang Theory provides an explanation for the observed distribution and structure of galaxies in the universe. As the universe expanded, matter began to clump together under the influence of gravity, forming structures like galaxies, stars, and planets. The theory predicts the large-scale structure of the universe, including the existence of galaxy clusters and cosmic filaments.
Overall, the Big Bang Theory is a comprehensive scientific explanation for the origin and evolution of the universe. It provides a framework for understanding the vastness and complexity of the cosmos and is supported by numerous observational and theoretical evidence.
What do we call the afterglow of the Big Bang?- a)Cosmic Background Radiation
- b)Galactic Glow
- c)Celestial Aftermath
- d)Stellar Residue
Correct answer is option 'A'. Can you explain this answer?
What do we call the afterglow of the Big Bang?
a)
Cosmic Background Radiation
b)
Galactic Glow
c)
Celestial Aftermath
d)
Stellar Residue
 | Ashwini Mehta answered |
Understanding the Cosmic Background Radiation
The afterglow of the Big Bang, known as Cosmic Background Radiation (CBR), is a crucial concept in cosmology. It represents the remnant heat from the early universe, providing insights into its origins and evolution.
What is Cosmic Background Radiation?
- CBR is the faint microwave radiation that fills the universe.
- It originated about 380,000 years after the Big Bang, when the universe cooled enough for protons and electrons to combine and form hydrogen atoms.
Significance of CBR
- Evidence of the Big Bang: CBR supports the Big Bang theory by confirming that the universe was once in a hot, dense state.
- Uniformity: The radiation is remarkably uniform across the sky, indicating that the early universe was homogeneous.
- Temperature: The temperature of CBR is approximately 2.7 Kelvin, which is consistent across the observable universe.
Discovery and Impact
- CBR was discovered in 1965 by Arno Penzias and Robert Wilson, leading to a Nobel Prize in Physics in 1978.
- Its discovery provided strong evidence for the Big Bang theory over alternative models, such as steady-state theories.
Conclusion
Cosmic Background Radiation is a fundamental aspect of our understanding of the universe, acting as a snapshot of the cosmos shortly after its inception. It not only supports the Big Bang theory but also offers a wealth of information about the universe's structure and evolution.
The afterglow of the Big Bang, known as Cosmic Background Radiation (CBR), is a crucial concept in cosmology. It represents the remnant heat from the early universe, providing insights into its origins and evolution.
What is Cosmic Background Radiation?
- CBR is the faint microwave radiation that fills the universe.
- It originated about 380,000 years after the Big Bang, when the universe cooled enough for protons and electrons to combine and form hydrogen atoms.
Significance of CBR
- Evidence of the Big Bang: CBR supports the Big Bang theory by confirming that the universe was once in a hot, dense state.
- Uniformity: The radiation is remarkably uniform across the sky, indicating that the early universe was homogeneous.
- Temperature: The temperature of CBR is approximately 2.7 Kelvin, which is consistent across the observable universe.
Discovery and Impact
- CBR was discovered in 1965 by Arno Penzias and Robert Wilson, leading to a Nobel Prize in Physics in 1978.
- Its discovery provided strong evidence for the Big Bang theory over alternative models, such as steady-state theories.
Conclusion
Cosmic Background Radiation is a fundamental aspect of our understanding of the universe, acting as a snapshot of the cosmos shortly after its inception. It not only supports the Big Bang theory but also offers a wealth of information about the universe's structure and evolution.
Pendulum clocks become slow in summer because- a)days in summer are large.
- b)of the friction in the coil.
- c)the length of the pendulum increases.
- d)the weight of the pendulum changes.
Correct answer is option 'C'. Can you explain this answer?
Pendulum clocks become slow in summer because
a)
days in summer are large.
b)
of the friction in the coil.
c)
the length of the pendulum increases.
d)
the weight of the pendulum changes.
 | Janhavi Dey answered |
Pendulum clocks become slow in summer because the length of the pendulum increases. Increase in length ∆l = lo α ∆T where lo = original length, α = coefficient of linear expansion and ∆T = change in temperature.
Why is hydrogen's presence essential during the Big Bang?- a)Hydrogen is crucial for the formation of stars and galaxies.
- b)Without hydrogen, there would be no planets in the universe.
- c)Hydrogen is the primary building block for water, vital for life's sustenance.
- d)Hydrogen helps maintain the equilibrium of the expanding universe.
Correct answer is option 'C'. Can you explain this answer?
Why is hydrogen's presence essential during the Big Bang?
a)
Hydrogen is crucial for the formation of stars and galaxies.
b)
Without hydrogen, there would be no planets in the universe.
c)
Hydrogen is the primary building block for water, vital for life's sustenance.
d)
Hydrogen helps maintain the equilibrium of the expanding universe.
| | Suresh Reddy answered |
Hydrogen is a primary building block for water, and without it, there would be no water, which is vital for life's sustenance.
Consider the following statements and select the correct answer using the codes given below:
Assertion (A): If ice collects on the freezer, the cooling in the refrigerator is affected adversely.
Reason (R): Ice is a poor conductor.- a)Both A and R are true, and R is the correct explanation of A.
- b)Both A and R are true, but R is not the correct explanation of A.
- c)A is true, but R is false.
- d)A is false, but R is true.
Correct answer is option 'A'. Can you explain this answer?
Consider the following statements and select the correct answer using the codes given below:
Assertion (A): If ice collects on the freezer, the cooling in the refrigerator is affected adversely.
Reason (R): Ice is a poor conductor.
Assertion (A): If ice collects on the freezer, the cooling in the refrigerator is affected adversely.
Reason (R): Ice is a poor conductor.
a)
Both A and R are true, and R is the correct explanation of A.
b)
Both A and R are true, but R is not the correct explanation of A.
c)
A is true, but R is false.
d)
A is false, but R is true.
 | Aniket Mehra answered |
If ice collects on the freezer, the cooling in the refrigerator is affected adversely because ice is a poor conductor of heat.
What force is responsible for the accelerated expansion of the Universe?- a)Gravitational pull of matter
- b)Dark energy
- c)Electromagnetic force
- d)Nuclear force
Correct answer is option 'B'. Can you explain this answer?
What force is responsible for the accelerated expansion of the Universe?
a)
Gravitational pull of matter
b)
Dark energy
c)
Electromagnetic force
d)
Nuclear force
| | Kavita Shah answered |
Dark energy is the force responsible for the accelerated expansion of the Universe.
What is the prevailing theory about the universe just before the Big Bang?- a)The universe was filled with stars and galaxies.
- b)The universe was an empty void with no matter or energy.
- c)The universe was filled with an intensely concentrated and unstable form of energy.
- d)The universe existed in a steady state.
Correct answer is option 'C'. Can you explain this answer?
What is the prevailing theory about the universe just before the Big Bang?
a)
The universe was filled with stars and galaxies.
b)
The universe was an empty void with no matter or energy.
c)
The universe was filled with an intensely concentrated and unstable form of energy.
d)
The universe existed in a steady state.
| | Manasa Chakraborty answered |
The prevailing theory about the universe just before the Big Bang is that it was filled with an intensely concentrated and unstable form of energy. This theory is known as the singularity theory or the hot and dense state theory.
Explanation:
- Big Bang Theory: The Big Bang theory is the leading explanation for the origin and evolution of the universe. According to this theory, the universe began as an extremely hot and dense state, and then expanded and cooled over time.
- Singularity: The singularity refers to a point of infinite density and temperature, where the laws of physics as we know them break down. It is believed that the universe existed in this singularity just before the Big Bang.
- Intensely Concentrated Energy: The singularity theory suggests that all the matter and energy of the universe were compressed into an incredibly small and dense region. This energy was so concentrated that the laws of physics as we understand them could not describe its behavior.
- Unstable State: The singularity was an unstable state, meaning that it could not persist indefinitely. The laws of physics dictate that such a highly concentrated and unstable energy state would eventually initiate a rapid expansion, leading to the Big Bang.
- Expansion and Cooling: As the singularity rapidly expanded, the energy began to cool down. This cooling allowed the formation of elementary particles, such as protons, neutrons, and electrons. These particles eventually combined to form atoms and, later on, stars and galaxies.
- Evidence: The singularity theory is supported by various lines of evidence, including the observed expansion of the universe, the abundance of light elements, and the cosmic microwave background radiation.
In conclusion, the prevailing theory suggests that just before the Big Bang, the universe was in an intensely concentrated and unstable state of energy known as the singularity. This theory provides an explanation for the origin and evolution of the universe as we know it today.
Explanation:
- Big Bang Theory: The Big Bang theory is the leading explanation for the origin and evolution of the universe. According to this theory, the universe began as an extremely hot and dense state, and then expanded and cooled over time.
- Singularity: The singularity refers to a point of infinite density and temperature, where the laws of physics as we know them break down. It is believed that the universe existed in this singularity just before the Big Bang.
- Intensely Concentrated Energy: The singularity theory suggests that all the matter and energy of the universe were compressed into an incredibly small and dense region. This energy was so concentrated that the laws of physics as we understand them could not describe its behavior.
- Unstable State: The singularity was an unstable state, meaning that it could not persist indefinitely. The laws of physics dictate that such a highly concentrated and unstable energy state would eventually initiate a rapid expansion, leading to the Big Bang.
- Expansion and Cooling: As the singularity rapidly expanded, the energy began to cool down. This cooling allowed the formation of elementary particles, such as protons, neutrons, and electrons. These particles eventually combined to form atoms and, later on, stars and galaxies.
- Evidence: The singularity theory is supported by various lines of evidence, including the observed expansion of the universe, the abundance of light elements, and the cosmic microwave background radiation.
In conclusion, the prevailing theory suggests that just before the Big Bang, the universe was in an intensely concentrated and unstable state of energy known as the singularity. This theory provides an explanation for the origin and evolution of the universe as we know it today.
What happened during the Big Bang?- a)All matter and energy in the universe were concentrated into an incredibly small, dense point.
- b)The universe reached a state of equilibrium.
- c)The universe expanded, leading to the formation of galaxies.
- d)All planets and stars were formed.
Correct answer is option 'A'. Can you explain this answer?
What happened during the Big Bang?
a)
All matter and energy in the universe were concentrated into an incredibly small, dense point.
b)
The universe reached a state of equilibrium.
c)
The universe expanded, leading to the formation of galaxies.
d)
All planets and stars were formed.
| | Suresh Reddy answered |
During the Big Bang, all matter and energy in the cosmos were concentrated into an incredibly small, dense point, much smaller than an atom.
What is the potential fate of the universe according to some scientists?- a)The universe will continue to expand indefinitely.
- b)The universe will contract back on itself in a phase called the "Big Crunch."
- c)The universe will stabilize and remain in its current state.
- d)The universe will fragment into multiple smaller universes.
Correct answer is option 'B'. Can you explain this answer?
What is the potential fate of the universe according to some scientists?
a)
The universe will continue to expand indefinitely.
b)
The universe will contract back on itself in a phase called the "Big Crunch."
c)
The universe will stabilize and remain in its current state.
d)
The universe will fragment into multiple smaller universes.
| | Akshara Desai answered |
Understanding the Big Crunch
The concept of the "Big Crunch" is one of several theories regarding the ultimate fate of the universe. It suggests a scenario where the universe, currently expanding, may eventually reverse this expansion and begin to contract.
Key Aspects of the Big Crunch Theory:
- Gravitational Forces: The theory is based on the gravitational attraction of matter in the universe. If the density of matter is high enough, the gravitational pull could eventually halt the expansion and pull everything back together.
- Collapse of Cosmic Structures: As the universe contracts, galaxies, stars, and planets would move closer together, leading to increased temperatures and densities.
- Ultimate Singularity: The end result of this contraction could be a singularity, where all matter is compressed into an infinitely small and dense point, similar to the conditions believed to have existed at the beginning of the universe during the Big Bang.
- Cyclic Universe Theory: Some scientists propose that this contraction could lead to a cyclical model of the universe, where it expands and contracts repeatedly over vast time scales.
Current Observations and Challenges:
- Recent observations, especially those concerning dark energy, indicate that the universe's expansion is accelerating rather than slowing down. This raises questions about the likelihood of a Big Crunch scenario.
- The fate of the universe remains a subject of intense research, with alternative theories like the Big Freeze and Big Rip also being considered.
In summary, while the Big Crunch presents a fascinating possibility for the universe’s fate, ongoing research and observations continue to shape our understanding of cosmic evolution.
The concept of the "Big Crunch" is one of several theories regarding the ultimate fate of the universe. It suggests a scenario where the universe, currently expanding, may eventually reverse this expansion and begin to contract.
Key Aspects of the Big Crunch Theory:
- Gravitational Forces: The theory is based on the gravitational attraction of matter in the universe. If the density of matter is high enough, the gravitational pull could eventually halt the expansion and pull everything back together.
- Collapse of Cosmic Structures: As the universe contracts, galaxies, stars, and planets would move closer together, leading to increased temperatures and densities.
- Ultimate Singularity: The end result of this contraction could be a singularity, where all matter is compressed into an infinitely small and dense point, similar to the conditions believed to have existed at the beginning of the universe during the Big Bang.
- Cyclic Universe Theory: Some scientists propose that this contraction could lead to a cyclical model of the universe, where it expands and contracts repeatedly over vast time scales.
Current Observations and Challenges:
- Recent observations, especially those concerning dark energy, indicate that the universe's expansion is accelerating rather than slowing down. This raises questions about the likelihood of a Big Crunch scenario.
- The fate of the universe remains a subject of intense research, with alternative theories like the Big Freeze and Big Rip also being considered.
In summary, while the Big Crunch presents a fascinating possibility for the universe’s fate, ongoing research and observations continue to shape our understanding of cosmic evolution.
What did the Wilkinson Microwave Anisotropy Probe (WMAP) mission study?- a)Formation of stars and galaxies.
- b)Cosmic Background Radiation.
- c)The expansion of the universe.
- d)The dark energy accelerating the universe's expansion.
Correct answer is option 'B'. Can you explain this answer?
What did the Wilkinson Microwave Anisotropy Probe (WMAP) mission study?
a)
Formation of stars and galaxies.
b)
Cosmic Background Radiation.
c)
The expansion of the universe.
d)
The dark energy accelerating the universe's expansion.
| | Kavita Shah answered |
The Wilkinson Microwave Anisotropy Probe (WMAP) mission studied the Cosmic Background Radiation in detail.
Consider the following statements and select the correct code:
Assertion (A): A piece of ice added to the drink cools it.
Reason (R): Ice takes latent heat from the drink for melting resulting in the cooling of the drink.- a)Both A and R are true, and R is the correct explanation of A.
- b)Both A and R are true, but R is not the correct explanation of A.
- c)A is true, but R is false.
- d)A is false, but R is true.
Correct answer is option 'A'. Can you explain this answer?
Consider the following statements and select the correct code:
Assertion (A): A piece of ice added to the drink cools it.
Reason (R): Ice takes latent heat from the drink for melting resulting in the cooling of the drink.
Assertion (A): A piece of ice added to the drink cools it.
Reason (R): Ice takes latent heat from the drink for melting resulting in the cooling of the drink.
a)
Both A and R are true, and R is the correct explanation of A.
b)
Both A and R are true, but R is not the correct explanation of A.
c)
A is true, but R is false.
d)
A is false, but R is true.
 | Baishali Patel answered |
A piece of ice added to the drink cool it because ice takes latent heat from the drink for melting resulting in the cooling of the drink.
Of the two bulbs in a house, one glows brighter than the other. Which of the following statements is correct?- a)The brightness does not depend on resistance.
- b)Both the bulbs have the same resistance.
- c)The brighter bulb has larger resistance.
- d)The dimmer bulb has larger resistance.
Correct answer is option 'D'. Can you explain this answer?
Of the two bulbs in a house, one glows brighter than the other. Which of the following statements is correct?
a)
The brightness does not depend on resistance.
b)
Both the bulbs have the same resistance.
c)
The brighter bulb has larger resistance.
d)
The dimmer bulb has larger resistance.
 | Gauri Menon answered |
The dimmer bulb has larger resistance than the brighter one. Power of the bulb ∝ 1 / Resistance Resistance is the obstruction offer to the flow of current.
Electronic motors operating at low voltages tend to burn out because- a)they draw more current which is inversely proportional to the voltage.
- b)they draw more current which is inversely proportional to the square root of the voltage.
- c)they draw heat proportional to V2.
- d)low voltage sets in electrical discharge.
Correct answer is option 'A'. Can you explain this answer?
Electronic motors operating at low voltages tend to burn out because
a)
they draw more current which is inversely proportional to the voltage.
b)
they draw more current which is inversely proportional to the square root of the voltage.
c)
they draw heat proportional to V2.
d)
low voltage sets in electrical discharge.
 | Akshara Chavan answered |
Electronic motors draw more current which is inversely proportional to the voltage, therefore, more heat H = I2Rt is generated.
Which one of the following statements is not true about cosmic rays?- a)They have very high frequency.
- b)They have very high wavelength.
- c)They are made of highly energetic charged particles.
- d)They originate from the sun.
Correct answer is option 'B'. Can you explain this answer?
Which one of the following statements is not true about cosmic rays?
a)
They have very high frequency.
b)
They have very high wavelength.
c)
They are made of highly energetic charged particles.
d)
They originate from the sun.
| | Pritam Desai answered |
Understanding Cosmic Rays
Cosmic rays are high-energy particles that travel through space and impact the Earth's atmosphere. Let's analyze the statements regarding cosmic rays to clarify why option 'B' is not true.
Statement A: They have very high frequency.
- Cosmic rays consist of particles such as protons, electrons, and heavier nuclei.
- These particles exhibit very high frequencies in terms of their energy levels, which can reach up to billions of electron volts (GeV).
Statement B: They have very high wavelength.
- This statement is not true because cosmic rays are composed of particles, not electromagnetic waves.
- The concept of wavelength applies to waves, such as light or radio waves, while cosmic rays are primarily particles with mass and thus do not have a wavelength in the traditional sense.
- In fact, high-energy particles have very short wavelengths due to their high momentum according to wave-particle duality, but they are not described in terms of wavelength as electromagnetic radiation.
Statement C: They are made of highly energetic charged particles.
- Cosmic rays indeed consist of highly energetic charged particles, which can include protons and atomic nuclei.
- Their high energy is what allows them to travel vast distances across the universe.
Statement D: They originate from the sun.
- While some cosmic rays do originate from the sun (solar cosmic rays), many others come from outside the solar system, including supernovae and other astronomical events.
Conclusion
In summary, the incorrect statement about cosmic rays is option 'B', as they are not defined by a high wavelength but rather by their nature as energetic charged particles.
Cosmic rays are high-energy particles that travel through space and impact the Earth's atmosphere. Let's analyze the statements regarding cosmic rays to clarify why option 'B' is not true.
Statement A: They have very high frequency.
- Cosmic rays consist of particles such as protons, electrons, and heavier nuclei.
- These particles exhibit very high frequencies in terms of their energy levels, which can reach up to billions of electron volts (GeV).
Statement B: They have very high wavelength.
- This statement is not true because cosmic rays are composed of particles, not electromagnetic waves.
- The concept of wavelength applies to waves, such as light or radio waves, while cosmic rays are primarily particles with mass and thus do not have a wavelength in the traditional sense.
- In fact, high-energy particles have very short wavelengths due to their high momentum according to wave-particle duality, but they are not described in terms of wavelength as electromagnetic radiation.
Statement C: They are made of highly energetic charged particles.
- Cosmic rays indeed consist of highly energetic charged particles, which can include protons and atomic nuclei.
- Their high energy is what allows them to travel vast distances across the universe.
Statement D: They originate from the sun.
- While some cosmic rays do originate from the sun (solar cosmic rays), many others come from outside the solar system, including supernovae and other astronomical events.
Conclusion
In summary, the incorrect statement about cosmic rays is option 'B', as they are not defined by a high wavelength but rather by their nature as energetic charged particles.
Consider the following statements in respect of a jet engine and a rocket:
1. A jet engine uses the surrounding air for its oxygen supply and so is unsuitable for motion in space.
2. A rocket carries its own supply of oxygen in the gas form as a fuel.
Q. Which of the above statement(s) is/are correct?- a)1 only
- b)2 only
- c)Both 1 and 2
- d)Neither 1 nor 2
Correct answer is option 'C'. Can you explain this answer?
Consider the following statements in respect of a jet engine and a rocket:
1. A jet engine uses the surrounding air for its oxygen supply and so is unsuitable for motion in space.
2. A rocket carries its own supply of oxygen in the gas form as a fuel.
Q. Which of the above statement(s) is/are correct?
1. A jet engine uses the surrounding air for its oxygen supply and so is unsuitable for motion in space.
2. A rocket carries its own supply of oxygen in the gas form as a fuel.
Q. Which of the above statement(s) is/are correct?
a)
1 only
b)
2 only
c)
Both 1 and 2
d)
Neither 1 nor 2
 | Saumya Bose answered |
In respect of a jet engine and a rocket, both the given statements are correct.
Why did the Universe appear opaque during its early stages?- a)Due to the absence of photons.
- b)Due to the scattering of photons by free electrons.
- c)Due to the presence of dark energy.
- d)Due to the formation of galaxies.
Correct answer is option 'B'. Can you explain this answer?
Why did the Universe appear opaque during its early stages?
a)
Due to the absence of photons.
b)
Due to the scattering of photons by free electrons.
c)
Due to the presence of dark energy.
d)
Due to the formation of galaxies.
| | Kavita Shah answered |
The Universe appeared opaque during its early stages because photons were scattered by free electrons, making it difficult for light to travel freely.
What caused the Universe to become transparent?- a)The formation of galaxies.
- b)The cooling down of the Universe.
- c)The disappearance of dark energy.
- d)The formation of neutral atoms during recombination.
Correct answer is option 'D'. Can you explain this answer?
What caused the Universe to become transparent?
a)
The formation of galaxies.
b)
The cooling down of the Universe.
c)
The disappearance of dark energy.
d)
The formation of neutral atoms during recombination.
| | Kavita Shah answered |
The Universe became transparent when neutral atoms were formed during recombination, causing the scattering of photons to cease.
Match List-I with List-II and select the correct answer using the codes given below the lists:

- a)A
- b)B
- c)C
- d)D
Correct answer is option 'D'. Can you explain this answer?
Match List-I with List-II and select the correct answer using the codes given below the lists:


a)
A
b)
B
c)
C
d)
D
 | Amrutha Kapoor answered |
Revolver was discovered by Colt. Dynamite was discovered by Alfred Nobel. Law of cooling was discovered by Newton and law of pressure by Pascal.
Match List-I with List-II and select the correct answer using the codes given below the lists:

- a)A
- b)B
- c)C
- d)D
Correct answer is option 'A'. Can you explain this answer?
Match List-I with List-II and select the correct answer using the codes given below the lists:


a)
A
b)
B
c)
C
d)
D
 | Baishali Patel answered |
Wavelength is the distance between two consecutive crests or troughs and 1 angstrom = 10–10m, joule is the unit of energy. Intensity of sound is measured in decibel. Frequency is measured in hertz (Hz).
What was the role of photons in the early Universe?- a)To form galaxies.
- b)To scatter off free electrons, making the Universe opaque.
- c)To create dark energy.
- d)To act as building blocks for matter.
Correct answer is option 'B'. Can you explain this answer?
What was the role of photons in the early Universe?
a)
To form galaxies.
b)
To scatter off free electrons, making the Universe opaque.
c)
To create dark energy.
d)
To act as building blocks for matter.
| | Kavita Shah answered |
Photons in the early Universe scattered off free electrons, making the Universe opaque.
What did Edwin Hubble's observation reveal about galaxies?- a)Galaxies are stationary and do not move.
- b)Galaxies are moving toward each other.
- c)Galaxies are moving away from each other.
- d)Galaxies are constantly changing their positions.
Correct answer is option 'C'. Can you explain this answer?
What did Edwin Hubble's observation reveal about galaxies?
a)
Galaxies are stationary and do not move.
b)
Galaxies are moving toward each other.
c)
Galaxies are moving away from each other.
d)
Galaxies are constantly changing their positions.
| | Suresh Reddy answered |
Edwin Hubble's observation revealed that galaxies are moving away from each other, suggesting an expanding universe.
It is more comfortable to wear white cloths in summer because- a)they reflect heat falling on them.
- b)they radiate heat tansferred from the body.
- c)they absorb perspiration.
- d)they are soothing to the eye.
Correct answer is option 'A'. Can you explain this answer?
It is more comfortable to wear white cloths in summer because
a)
they reflect heat falling on them.
b)
they radiate heat tansferred from the body.
c)
they absorb perspiration.
d)
they are soothing to the eye.
 | Shraddha Mukherjee answered |
The white clothes reflect heat falling on them in summer and they absorb the least from the sun.
What was the purpose of the Planck mission?- a)To study the formation of stars and galaxies.
- b)To understand the evolution of the universe's shape.
- c)To study dark energy and its effects on the universe's expansion.
- d)To create accurate maps of cosmic microwave background radiation.
Correct answer is option 'D'. Can you explain this answer?
What was the purpose of the Planck mission?
a)
To study the formation of stars and galaxies.
b)
To understand the evolution of the universe's shape.
c)
To study dark energy and its effects on the universe's expansion.
d)
To create accurate maps of cosmic microwave background radiation.
| | Kavita Shah answered |
The purpose of the Planck mission was to create accurate maps of cosmic microwave background radiation.
Match List-I with List-II and select the correct answer with the help of codes given below: 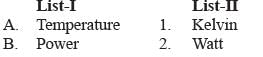


- a)A
- b)B
- c)C
- d)D
Correct answer is option 'C'. Can you explain this answer?
Match List-I with List-II and select the correct answer with the help of codes given below:
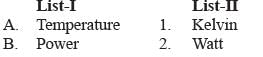


a)
A
b)
B
c)
C
d)
D
 | Akshara Chavan answered |
The unit of temperature is kelvin, power is measured in watt, pressure in pascal and force in newton.
How did the Universe continue to evolve after becoming transparent?- a)It stopped changing and remained static.
- b)Dark energy caused the expansion to slow down.
- c)Atoms formed molecules and then celestial structures.
- d)The rate of expansion remained constant.
Correct answer is option 'C'. Can you explain this answer?
How did the Universe continue to evolve after becoming transparent?
a)
It stopped changing and remained static.
b)
Dark energy caused the expansion to slow down.
c)
Atoms formed molecules and then celestial structures.
d)
The rate of expansion remained constant.
| | Kavita Shah answered |
After becoming transparent, atoms formed molecules, leading to the creation of celestial structures like planets, stars, and galaxies.
What is the inflationary model's proposed explanation for the early Universe's homogeneity problem?- a)The rapid expansion phase shortly after the Big Bang.
- b)The presence of dark matter.
- c)The interaction of dark energy with matter.
- d)The formation of galaxies.
Correct answer is option 'A'. Can you explain this answer?
What is the inflationary model's proposed explanation for the early Universe's homogeneity problem?
a)
The rapid expansion phase shortly after the Big Bang.
b)
The presence of dark matter.
c)
The interaction of dark energy with matter.
d)
The formation of galaxies.
| | Kavita Shah answered |
The inflationary model proposes a short period of rapid expansion shortly after the Big Bang, which helps explain the early Universe's homogeneity.
How did the Universe change from an opaque state to a transparent one?- a)Through the formation of dark matter.
- b)Through the cooling down and recombination of atoms.
- c)Through the rapid expansion of galaxies.
- d)Through the influence of dark energy.
Correct answer is option 'B'. Can you explain this answer?
How did the Universe change from an opaque state to a transparent one?
a)
Through the formation of dark matter.
b)
Through the cooling down and recombination of atoms.
c)
Through the rapid expansion of galaxies.
d)
Through the influence of dark energy.
| | Kavita Shah answered |
The Universe changed from an opaque state to a transparent one through the cooling down and recombination of atoms.
When 1 mol of a gas is heated at constant volume, temperature is raised from 298 to 308 K. Heat supplied to gas is 500 J. Then which statement is correct?- a)q = W = 500 J ; ∆U = 0
- b)q = ∆U = 500 ; W = 0
- c)q = W = 500 J ; ∆U = 500
- d)∆U = 0,q = W = -500 J
Correct answer is option 'B'. Can you explain this answer?
When 1 mol of a gas is heated at constant volume, temperature is raised from 298 to 308 K. Heat supplied to gas is 500 J. Then which statement is correct?
a)
q = W = 500 J ; ∆U = 0
b)
q = ∆U = 500 ; W = 0
c)
q = W = 500 J ; ∆U = 500
d)
∆U = 0,q = W = -500 J
| | Christy Paul answered |
Constant volume hence w= 0
H=U=Q=500
H=U=Q=500
Chapter doubts & questions for Physics - 6 Months Preparation Course for UPSC EPFO 2026 is part of UPSC CSE exam preparation. The chapters have been prepared according to the UPSC CSE exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for UPSC CSE 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Physics - 6 Months Preparation Course for UPSC EPFO in English & Hindi are available as part of UPSC CSE exam. Download more important topics, notes, lectures and mock test series for UPSC CSE Exam by signing up for free.
6 Months Preparation Course for UPSC EPFO603 videos|2061 docs|720 tests |
Top Courses UPSC CSE
Related UPSC CSE Content
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup



