All Exams > OPSC OCS (Odisha) > 6 Months Preparation Course for OPSC > All Questions
All questions of Class 10 NCERT Chemistry for OPSC OCS (Odisha) Exam
Which of the following does not conduct electricity?- a)Sodium hydroxide
- b)Rain water
- c)Hydrochloric acid
- d)Distilled water
Correct answer is option 'D'. Can you explain this answer?
Which of the following does not conduct electricity?
a)
Sodium hydroxide
b)
Rain water
c)
Hydrochloric acid
d)
Distilled water
| | Gaurav Kumar answered |
Distilled water do not conduct electricity. The reason is that a liquid conducts electricity is by the positively or negatively charged ions that are actually moving from one of the electrodes to the other, carrying charge (electricity) with them.
Fe2O3 + 2Al → Al2O3 + 2Fe
The above reaction is an example of a:- a)Decomposition reaction
- b)Combination reaction
- c)Displacement reaction
- d)Single displacement reaction
Correct answer is option 'C'. Can you explain this answer?
Fe2O3 + 2Al → Al2O3 + 2Fe
The above reaction is an example of a:
The above reaction is an example of a:
a)
Decomposition reaction
b)
Combination reaction
c)
Displacement reaction
d)
Single displacement reaction
 | Anushka Chopra answered |
The given equation is a displacement reaction in which Fe of Fe2O3 has been displaced by Al. Hence, (c) is the correct option.
How many elements were known when Mendeleev started his work on his periodic table?- a)63
- b)55
- c)69
- d)45
Correct answer is option 'A'. Can you explain this answer?
How many elements were known when Mendeleev started his work on his periodic table?
a)
63
b)
55
c)
69
d)
45
| | Nisha Choudhury answered |
There were 63 known elements in 1860 when Mendeleev published his Periodic Table. CIENTISTS HAD IDENTIFIED over 63 elements by Mendeleev's time.
The best conductor of electricity among the following is:- a)Hg (Mercury)
- b)Al (Aluminum)
- c)Ag (Silver)
- d)Au (Gold)
Correct answer is option 'C'. Can you explain this answer?
The best conductor of electricity among the following is:
a)
Hg (Mercury)
b)
Al (Aluminum)
c)
Ag (Silver)
d)
Au (Gold)
| | Avinash Patel answered |
Metals are good conductors of electricity. silver is found in in pure form from the earth. so In the given options all are metals. Among them silver is the best conductor of electricity after second best conductor of electricity is gold.
Metal which is a poor conductor of heat is:- a)Zinc
- b)Lead
- c)Gold
- d)Copper
Correct answer is option 'B'. Can you explain this answer?
Metal which is a poor conductor of heat is:
a)
Zinc
b)
Lead
c)
Gold
d)
Copper
| | Anjana Khatri answered |
Although metals are supposed to be good conductors of electricity and heat,metals like mercury, lead, alloys of iron and chromium, titanium and stainless steel are poor conductors when compared to silver, copper and gold.
Among the following metals, the metal that can displace zinc from zinc sulphate solution is:- a)Magnesium
- b)Copper
- c)Lead
- d)Silver
Correct answer is option 'A'. Can you explain this answer?
Among the following metals, the metal that can displace zinc from zinc sulphate solution is:
a)
Magnesium
b)
Copper
c)
Lead
d)
Silver
| | Arun Sharma answered |
Magnesium is more reactive than zinc therefore it can displace zinc from its solution.
Which of the following is an example of displacement reaction?- a)4 Na+ O2 → 2 Na2O
- b)2 Cu + O2 → 2 CuO
- c)Mg + 2 HCl → MgCl2 + H2
- d)N2 + 3 H2 → 2 NH3
Correct answer is option 'C'. Can you explain this answer?
Which of the following is an example of displacement reaction?
a)
4 Na+ O2 → 2 Na2O
b)
2 Cu + O2 → 2 CuO
c)
Mg + 2 HCl → MgCl2 + H2
d)
N2 + 3 H2 → 2 NH3
 | Orion Classes answered |
It is a single replacement reaction.
Mg + 2HCl → MgCl2 + H2
Mg + 2HCl → MgCl2 + H2
Which of the following metals comes above zinc in reactivity series?- a)Silver
- b)Copper
- c)Aluminium
- d)Iron
Correct answer is option 'C'. Can you explain this answer?
Which of the following metals comes above zinc in reactivity series?
a)
Silver
b)
Copper
c)
Aluminium
d)
Iron
| | Karthik murthy answered |
Reactivity Series:
The reactivity series is a list of metals arranged in order of their decreasing reactivity. The most reactive metal is placed at the top and the least reactive metal is placed at the bottom of the series.
The reactivity series of metals is as follows:
Potassium > Sodium > Calcium > Magnesium > Aluminium > Zinc > Iron > Lead > Copper > Silver > Gold
Explanation:
Aluminium is more reactive than zinc and is placed above zinc in the reactivity series. This is because aluminium has a stronger tendency than zinc to lose electrons and form positive ions.
When aluminium is exposed to air, it reacts with oxygen to form a layer of aluminium oxide. This layer of oxide is very thin and is also very stable. It prevents further reaction of aluminium with oxygen and protects the metal from corrosion.
Zinc, on the other hand, reacts with oxygen in the air to form zinc oxide. However, the layer of oxide formed on zinc is not as stable as the layer of oxide formed on aluminium. Therefore, zinc is more susceptible to corrosion than aluminium.
Conclusion:
In conclusion, aluminium is more reactive than zinc and is placed above zinc in the reactivity series. This means that aluminium can displace zinc from its compounds in a chemical reaction, but zinc cannot displace aluminium from its compounds.
The reactivity series is a list of metals arranged in order of their decreasing reactivity. The most reactive metal is placed at the top and the least reactive metal is placed at the bottom of the series.
The reactivity series of metals is as follows:
Potassium > Sodium > Calcium > Magnesium > Aluminium > Zinc > Iron > Lead > Copper > Silver > Gold
Explanation:
Aluminium is more reactive than zinc and is placed above zinc in the reactivity series. This is because aluminium has a stronger tendency than zinc to lose electrons and form positive ions.
When aluminium is exposed to air, it reacts with oxygen to form a layer of aluminium oxide. This layer of oxide is very thin and is also very stable. It prevents further reaction of aluminium with oxygen and protects the metal from corrosion.
Zinc, on the other hand, reacts with oxygen in the air to form zinc oxide. However, the layer of oxide formed on zinc is not as stable as the layer of oxide formed on aluminium. Therefore, zinc is more susceptible to corrosion than aluminium.
Conclusion:
In conclusion, aluminium is more reactive than zinc and is placed above zinc in the reactivity series. This means that aluminium can displace zinc from its compounds in a chemical reaction, but zinc cannot displace aluminium from its compounds.
Identify the amphoteric oxide from the following:- a)MgO
- b)Na2O
- c)Al2O3
- d)K2O
Correct answer is option 'C'. Can you explain this answer?
Identify the amphoteric oxide from the following:
a)
MgO
b)
Na2O
c)
Al2O3
d)
K2O
| | Arun Sharma answered |
Amphoteric metal oxides react with both acids as well as bases to produce salt and water.
Aluminium oxide is an amphoteric oxide because it reacts with acids as well as bases and form salts and water.
Al2O3 + 6HCl → 2AlCl3 + 3H2O (Basic nature)
Aluminium Hydrochloric Aluminium Water
oxide acid chloride
Al2O3 + 2NaOH → 2NaAlO2 + H2O (Acidic nature)
Aluminium Sodium Sodium Water
oxide Hydroxide aluminate
Identify ‘X’ in the reaction: 2HCl + CuO → X + H2O- a)CuCl
- b)Cu(OH)2
- c)CuCl2
- d)HOCl
Correct answer is option 'C'. Can you explain this answer?
Identify ‘X’ in the reaction: 2HCl + CuO → X + H2O
a)
CuCl
b)
Cu(OH)2
c)
CuCl2
d)
HOCl
| | Vikram Kapoor answered |
When copper oxide and dilute hydrochloric acid are mixed the blue green solution is formed.
The reaction is :-
CuO + 2HCl → CuCl2 + H2O
The reaction is :-
CuO + 2HCl → CuCl2 + H2O
All metals exist as solid at room temperature except the metal ‘X’. Name the metal “X’.- a)Tungsten
- b)Mercury
- c)Gold
- d)Potassium
Correct answer is option 'B'. Can you explain this answer?
All metals exist as solid at room temperature except the metal ‘X’. Name the metal “X’.
a)
Tungsten
b)
Mercury
c)
Gold
d)
Potassium
| | Ananya Das answered |
It is the only metal that is liquid at room temperature. It has the lowest melting point and boiling point of any other metal. It has low thermal conductivity, and a quite low electrical conductivity. It is the only metal that doesn’t form diatomic molecules in the gaseous phase.
Which metal is present in Calcium Hydroxide?- a)C
- b)O
- c)Ca
- d)H
Correct answer is option 'C'. Can you explain this answer?
Which metal is present in Calcium Hydroxide?
a)
C
b)
O
c)
Ca
d)
H
| | Amit Sharma answered |
Calcium hydroxide Ca ( OH) 2 , has calcium ( Ca) which is a metal.
The colour of phenolphthalein in acids is:- a)Colourless
- b)Red
- c)Pink
- d)Blue
Correct answer is option 'A'. Can you explain this answer?
The colour of phenolphthalein in acids is:
a)
Colourless
b)
Red
c)
Pink
d)
Blue
| | Krishna Iyer answered |
Phenolphthalein is often used as an indicator in acid–base titrations. For this application, it turns colourless in acidic solutions and magenta in basic solutions.
Carbon dioxide is an example of:- a)Amphoteric oxide
- b)Acidic oxide
- c)Basic oxide
- d)Neutral oxide
Correct answer is option 'B'. Can you explain this answer?
Carbon dioxide is an example of:
a)
Amphoteric oxide
b)
Acidic oxide
c)
Basic oxide
d)
Neutral oxide
| | Gaurav Kumar answered |
Acid oxides is a complex chemical substance oxides, which form a salt with the chemical reactions with bases or basic oxides and do not react with acidic oxides.
Examples of acidic oxides can be:
CO2 (all known carbon dioxide), P2O5 - oxide of phosphorus (formed in air if burns white phosphorus), SO3 - oxide of sulfur (VI) is a substance used for sulfuric acid.
Examples of acidic oxides can be:
CO2 (all known carbon dioxide), P2O5 - oxide of phosphorus (formed in air if burns white phosphorus), SO3 - oxide of sulfur (VI) is a substance used for sulfuric acid.
The colour of anhydrous copper sulphate is:- a)White
- b)Blue
- c)Red
- d)Yellow
Correct answer is option 'A'. Can you explain this answer?
The colour of anhydrous copper sulphate is:
a)
White
b)
Blue
c)
Red
d)
Yellow
| | Rahul Kapoor answered |
The answer is b.
The crystals of hydrated copper sulphate salt are blue in colour. When heated, the salt loses its water of crystallization and turns white.
Which of the following non-metal is good conductor of electricity?- a)Graphite
- b)Phosphorus
- c)Hydrogen
- d)Bromine
Correct answer is option 'A'. Can you explain this answer?
Which of the following non-metal is good conductor of electricity?
a)
Graphite
b)
Phosphorus
c)
Hydrogen
d)
Bromine
| | Gaurav Kumar answered |
Carbon, in the form of Graphite is a good conductor of electricity. It conducts heat and electricity like a metal or a metalloid.
The Law of Octaves was applicable only upto element ________.- a)Sodium
- b)Calcium
- c)Zinc
- d)Copper
Correct answer is option 'B'. Can you explain this answer?
The Law of Octaves was applicable only upto element ________.
a)
Sodium
b)
Calcium
c)
Zinc
d)
Copper
| | Vikram Kapoor answered |
The major limitations of Newlands' law of octaves were :
It was applicable to only lighter elements having atomic masses upto 40 u, i.e., upto calcium. After calcium, the first and the eighth element did not have similar properties. For example chromium (Cr) and yttrium (Y) are the first and the eighth element placed in the same column but they have entirely different properties.
It was applicable to only lighter elements having atomic masses upto 40 u, i.e., upto calcium. After calcium, the first and the eighth element did not have similar properties. For example chromium (Cr) and yttrium (Y) are the first and the eighth element placed in the same column but they have entirely different properties.
P, Q, R are elements of Dobereiner’s triads. If the atomic mass of P is 7 and that of Q is 23, What will be the atomic mass of R?- a)15.0
- b)40.0
- c)30.0
- d)39.0
Correct answer is option 'D'. Can you explain this answer?
P, Q, R are elements of Dobereiner’s triads. If the atomic mass of P is 7 and that of Q is 23, What will be the atomic mass of R?
a)
15.0
b)
40.0
c)
30.0
d)
39.0
| | Gaurav Kumar answered |
In a Dobereiner's traid, the atomic mass of the middle element is roughly the average of the atomic mass of the other 2 elements. Thus,
Atomic mass of Q = (at. mass of P+ at. mass of R) /2
⇒ 23 = 7+ m(R) /2
⇒ 23*2 = 7+ m(R)
⇒ 46 -7 = m(R)
⇒ atomic mass of R = 39
Atomic mass of Q = (at. mass of P+ at. mass of R) /2
⇒ 23 = 7+ m(R) /2
⇒ 23*2 = 7+ m(R)
⇒ 46 -7 = m(R)
⇒ atomic mass of R = 39
What happens when non-metals react with water?a) Hydrogen gas is formedb) Carbon dioxide gas is formedc) Non-metals do not react with waterd) None of these.Correct answer is option 'C'. Can you explain this answer?
b) Carbon dioxide gas is formed
c) Non-metals do not react with water
d) None of these.
Correct answer is option 'C'. Can you explain this answer?
| | Pooja Shah answered |
Non - metals do not react with water to evolve hydrogen gas. ( no reaction)
Identify the type of reaction: HCl + NaOH → NaCl + H2O- a)Combination reaction
- b)Double decomposition reaction
- c)Decomposition reaction
- d)Neutralisation reaction
Correct answer is option 'D'. Can you explain this answer?
Identify the type of reaction: HCl + NaOH → NaCl + H2O
a)
Combination reaction
b)
Double decomposition reaction
c)
Decomposition reaction
d)
Neutralisation reaction
| | Krishna Iyer answered |
Reaction of a strong acid with strong base is called neutralization reaction which produces salt and water,
HCl + NaOH → NaCl + H2O
This equation is already balanced.
The number of C-H bonds in ethane(C2H6) molecule are:- a)10
- b)8
- c)6
- d)4
Correct answer is option 'C'. Can you explain this answer?
The number of C-H bonds in ethane(C2H6) molecule are:
a)
10
b)
8
c)
6
d)
4
| | Naina Sharma answered |
The structure of the ethane molecule can be represented as:
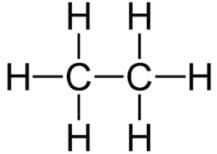
There are six C-H bonds and one C-C bond . So, Total 6 convalent bond between C-H

There are six C-H bonds and one C-C bond . So, Total 6 convalent bond between C-H
Butanone is a four-carbon compound with the functional group- a)carboxylic acid
- b)aldehyde
- c)ketone
- d)alcohol
Correct answer is option 'C'. Can you explain this answer?
Butanone is a four-carbon compound with the functional group
a)
carboxylic acid
b)
aldehyde
c)
ketone
d)
alcohol
| | Pushkar murthy answered |
The functional group is ketone (> C = 0) also known as alkanone.
Which one is more metallic element ?- a)Na
- b)Mg
- c)Al
- d)Si
Correct answer is option 'A'. Can you explain this answer?
Which one is more metallic element ?
a)
Na
b)
Mg
c)
Al
d)
Si
| | Diya Sharma answered |
Because it more fastly or quickly loss it's electron.
Structural formula of ethyne is- a)
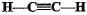
- b)
- c)
- d)
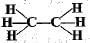
Correct answer is option 'A'. Can you explain this answer?
Structural formula of ethyne is
a)
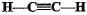
b)
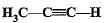
c)

d)
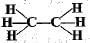
| | Puneet mukherjee answered |
'Eth' tells that there are 2 Carbon atoms and 'yne' tells that there is a triple bond.
Which gas causes the bread or cake to rise making them soft and spongy?- a)CO2
- b)O2
- c)CO
- d)HCl
Correct answer is option 'A'. Can you explain this answer?
Which gas causes the bread or cake to rise making them soft and spongy?
a)
CO2
b)
O2
c)
CO
d)
HCl
| | Vikram Kapoor answered |
Carbon dioxide is released when baking soda is added to cake and this makes the cake to rise making them soft and spongy.
Which one of the following metal reacts vigorously with oxygen and water?- a)Sodium
- b)Iron
- c)Calcium
- d)Magnesium
Correct answer is option 'A'. Can you explain this answer?
Which one of the following metal reacts vigorously with oxygen and water?
a)
Sodium
b)
Iron
c)
Calcium
d)
Magnesium
| | Ananya Das answered |
Sodium metal reacts vigorously with oxygen and water.
A lustrous non-metal is:- a)Iodine
- b)Bromine
- c)Chlorine
- d)Sulphur
Correct answer is option 'A'. Can you explain this answer?
A lustrous non-metal is:
a)
Iodine
b)
Bromine
c)
Chlorine
d)
Sulphur
| | Pooja Shah answered |
Non - metals do not have luster. They do not reflect light from their surface ( except iodine and diamond) . So, here a lustrous now Metal is iodine.
The lustre of a metal is due to:- a)Its high density
- b)Presence of free electrons
- c)Its high polishing
- d)Its chemical inertness
Correct answer is option 'B'. Can you explain this answer?
The lustre of a metal is due to:
a)
Its high density
b)
Presence of free electrons
c)
Its high polishing
d)
Its chemical inertness
 | Rohini Seth answered |
The lustre of a metal is due to presence of free electrons.
It has to do with the way light and electrons on the surface of metals interact. The outer electrons in a metal are almost not bound to any individual atom, thus are relatively free, and are concentrated on the surface. These electrons (electron density) tend to oscillate at a collective frequency.
How many elements are placed in lanthanide and actinide series?- a)57, 89
- b)14, 14
- c)89, 57
- d)14, 16
Correct answer is option 'B'. Can you explain this answer?
How many elements are placed in lanthanide and actinide series?
a)
57, 89
b)
14, 14
c)
89, 57
d)
14, 16
| | Neha Patel answered |
14 elements
The same holds for the actinide series that runs from atomic number 90 through to number 103, again 14 elements. Thus, as you move from thorium (Th) at element number 90, you begin to fill up the 5f sublevel and continue to fill up the 5f sublevel until you finish the actinide series at lawrencium (Lr).
Which of the following is not observed when aluminium is added to a solution of copper sulphate?- a)Final solution is light green.
- b)Solution is blue in the beginning.
- c)The brown mass is deposited on aluminium.
- d)Final solution is colourless.
Correct answer is option 'A'. Can you explain this answer?
Which of the following is not observed when aluminium is added to a solution of copper sulphate?
a)
Final solution is light green.
b)
Solution is blue in the beginning.
c)
The brown mass is deposited on aluminium.
d)
Final solution is colourless.
 | Ashwani Mishra answered |
The final solution becomes colorless and not light green since aluminum sulphate is colourless.
Buckminsterfullerene is an allotropic form of- a)phosphorus
- b)sulphur
- c)carbon
- d)tin
Correct answer is option 'C'. Can you explain this answer?
Buckminsterfullerene is an allotropic form of
a)
phosphorus
b)
sulphur
c)
carbon
d)
tin
| | Naina Sharma answered |
Buckminsterfullerene is an allotropic form of carbon. It resembles the geodesic dome designed by Buckminster.
Which of the following has the maximum non-metallic character ?- a)F
- b)Cl
- c)Br
- d)I
Correct answer is option 'A'. Can you explain this answer?
Which of the following has the maximum non-metallic character ?
a)
F
b)
Cl
c)
Br
d)
I
| | Ananya Das answered |
Non metallic character decreases as we move down the group. In a group, the size of an element increases because there is an addition of new shell and electron is added in that shell. Hence, fluorine has the most non metallic character.
Aqueous solution of sodium hydroxide turns blue litmus:- a)Red
- b)No change
- c)Colourless
- d)Pink
Correct answer is option 'B'. Can you explain this answer?
Aqueous solution of sodium hydroxide turns blue litmus:
a)
Red
b)
No change
c)
Colourless
d)
Pink
 | Sagar Rane answered |
Since Sodium hydroxide is a base and thus it has no effect on a blue litmus paper but it changes red litmus to blue.
Which of the following is the correct representation of electron dot structure of nitrogen?- a)
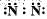
- b)
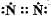
- c)
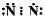
- d)
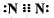
Correct answer is option 'D'. Can you explain this answer?
Which of the following is the correct representation of electron dot structure of nitrogen?
a)
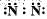
b)
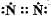
c)
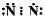
d)
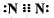
| | Raghav Bansal answered |
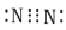
Three pairs of electrons are shared.
नम्नलिखित गद्यांशों को ध्यानपूर्वक पढ़कर नीचे दिए गए प्रश्नों के सही उत्तर विकल्पों में से चुनिए:रज्जो ने चार-पाँच आम अंजुली में लेकर मेरी ओर बढ़ा दिए। आम लेने के लिए मैंने हाथ बढ़ाया तो मेरी निगाह एक क्षण के लिए उसके हाथों पर ठिठक गई। गोरी-गोरी कलाइयों पर लाख की चूडिय़ाँ बहुत ही फब रही थीं।
बदलू ने मेरी दृष्टि देख ली और बोल पड़ा, यही आखिरी जोड़ा बनाया था ज्जमींदार साहब की बेटी के विवाह पर। दस आने पैसे मुझको दे रहे थे। मैंने जोड़ा नहीं दिया। कहा, शहर से ले आओ।प्रश्न - बदलू ने शमींदार को चूड़ियों का जोड़ा क्यों नहीं दिया? - a)सुंदर न होने के कारण
- b)अपनी बेटी को देने के कारण
- c)दाम अध्कि पाने के कारण
- d)कम दाम पाने के कारण
Correct answer is option 'D'. Can you explain this answer?
नम्नलिखित गद्यांशों को ध्यानपूर्वक पढ़कर नीचे दिए गए प्रश्नों के सही उत्तर विकल्पों में से चुनिए:
रज्जो ने चार-पाँच आम अंजुली में लेकर मेरी ओर बढ़ा दिए। आम लेने के लिए मैंने हाथ बढ़ाया तो मेरी निगाह एक क्षण के लिए उसके हाथों पर ठिठक गई। गोरी-गोरी कलाइयों पर लाख की चूडिय़ाँ बहुत ही फब रही थीं।
बदलू ने मेरी दृष्टि देख ली और बोल पड़ा, यही आखिरी जोड़ा बनाया था ज्जमींदार साहब की बेटी के विवाह पर। दस आने पैसे मुझको दे रहे थे। मैंने जोड़ा नहीं दिया। कहा, शहर से ले आओ।
बदलू ने मेरी दृष्टि देख ली और बोल पड़ा, यही आखिरी जोड़ा बनाया था ज्जमींदार साहब की बेटी के विवाह पर। दस आने पैसे मुझको दे रहे थे। मैंने जोड़ा नहीं दिया। कहा, शहर से ले आओ।
प्रश्न - बदलू ने शमींदार को चूड़ियों का जोड़ा क्यों नहीं दिया?
a)
सुंदर न होने के कारण
b)
अपनी बेटी को देने के कारण
c)
दाम अध्कि पाने के कारण
d)
कम दाम पाने के कारण
| | Geetika Shah answered |
D is the correct option.कम दाम पाने के कारण बदलू ने शमींदार को चूड़ियों का जोड़ा नहीं दिया.
Which is the most abundant non-metal on the earth?- a)Sulphur
- b)Oxygen
- c)Silicon
- d)Phosphorus
Correct answer is option 'B'. Can you explain this answer?
Which is the most abundant non-metal on the earth?
a)
Sulphur
b)
Oxygen
c)
Silicon
d)
Phosphorus
| | Jaideep mukherjee answered |
Most Abundant Non-Metal on Earth:
Explanation:
Oxygen is the most abundant non-metal on Earth. It makes up around 21% of the Earth's atmosphere, and it is also found in the Earth's crust, oceans, and other compounds like water and rocks. Oxygen is essential for life as it is a key component of the air we breathe and is necessary for the respiration of living organisms.
Other Options:
Let's take a look at the other options and why they are not the most abundant non-metal on Earth:
a) Sulphur - Sulphur is a non-metal, but it is not the most abundant on Earth. Although it is found in large quantities in the Earth's crust and oceans, it is not as abundant as oxygen.
c) Silicon - Silicon is a non-metal that is found in the Earth's crust, but it is not as abundant as oxygen. It is often found in rocks and minerals like quartz and sand, but it is not as essential to life as oxygen.
d) Phosphorus - Phosphorus is a non-metal that is found in the Earth's crust, but it is not as abundant as oxygen. It is often used in fertilizers and is an important component of DNA, but it is not as essential to life as oxygen.
Conclusion:
In conclusion, oxygen is the most abundant non-metal on Earth. It is essential for life and is found in the Earth's atmosphere, crust, oceans, and other compounds.
Explanation:
Oxygen is the most abundant non-metal on Earth. It makes up around 21% of the Earth's atmosphere, and it is also found in the Earth's crust, oceans, and other compounds like water and rocks. Oxygen is essential for life as it is a key component of the air we breathe and is necessary for the respiration of living organisms.
Other Options:
Let's take a look at the other options and why they are not the most abundant non-metal on Earth:
a) Sulphur - Sulphur is a non-metal, but it is not the most abundant on Earth. Although it is found in large quantities in the Earth's crust and oceans, it is not as abundant as oxygen.
c) Silicon - Silicon is a non-metal that is found in the Earth's crust, but it is not as abundant as oxygen. It is often found in rocks and minerals like quartz and sand, but it is not as essential to life as oxygen.
d) Phosphorus - Phosphorus is a non-metal that is found in the Earth's crust, but it is not as abundant as oxygen. It is often used in fertilizers and is an important component of DNA, but it is not as essential to life as oxygen.
Conclusion:
In conclusion, oxygen is the most abundant non-metal on Earth. It is essential for life and is found in the Earth's atmosphere, crust, oceans, and other compounds.
Which of the following salt is basic in nature?- a)Aluminium chloride
- b)Sodium acetate
- c)Sodium chloride
- d)Potassium nitrate
Correct answer is option 'B'. Can you explain this answer?
Which of the following salt is basic in nature?
a)
Aluminium chloride
b)
Sodium acetate
c)
Sodium chloride
d)
Potassium nitrate
| | Krishna Iyer answered |
Sodium acetate (CH3COONa) is a salt in solid state and can't be regarded as an acid or base in anhydrous or molten form.
Although, being an ionic compound, sodium acetate dissociates in water to produce sodium ion Na+ and acetate ion CH3COO-. These ions react with water producing NaOH and CH3COOH.
Now, as NaOH is a strong base and CH3COOH is a weak acid, the resultant solution is basic in nature.
Hence, sodium acetate is basic in aqueous medium.
Although, being an ionic compound, sodium acetate dissociates in water to produce sodium ion Na+ and acetate ion CH3COO-. These ions react with water producing NaOH and CH3COOH.
Now, as NaOH is a strong base and CH3COOH is a weak acid, the resultant solution is basic in nature.
Hence, sodium acetate is basic in aqueous medium.
Which one of the following is a poor conductor of heat?- a)Mercury
- b)Zinc
- c)Silver
- d)Copper
Correct answer is option 'A'. Can you explain this answer?
Which one of the following is a poor conductor of heat?
a)
Mercury
b)
Zinc
c)
Silver
d)
Copper
| | Anjana Khatri answered |
Silver is best conductor of heat while among metals lead is poorest conductor of heat. Cooper and aluminum are good conductor while mercury is a poor conductor. Although metals are supposed to be good conductors of electricity and heat,metals like mercury, lead, alloys of iron and chromium, titanium and stainless steel are poor conductors.
The reaction, Zn + 2HCl → ZnCl2 + H2 is an example of:- a)Displacement reaction
- b)Combination reaction
- c)Double displacement reaction
- d)Decomposition reaction
Correct answer is option 'A'. Can you explain this answer?
The reaction, Zn + 2HCl → ZnCl2 + H2 is an example of:
a)
Displacement reaction
b)
Combination reaction
c)
Double displacement reaction
d)
Decomposition reaction
| | Neha Patel answered |
This is a metal/acid reaction if carried out in aqueous solution.
It is also a displacement reaction where 1 atom of Zinc displaces 2 H+ ions
and it is a redox reaction as the Zn atom loses electrons (it is OXIDISED) and the 2 H+ ions gain electrons (they are REDUCED)
How many triads could Dobereiner identify from the... more elements known at that time?a) 3b) 5c) 4d) 2Correct answer is option 'A'. Can you explain this answer?
| | Pooja Shah answered |
In 1817 a German chemist Johann Wolfgang Döbereiner arranged the elements with similar properties into groups.
Some groups were identified having three elements each. So he called these groups ‘triads’. Only three triads could be identified from the elements discovered at that time.
Carbon forms bonds with other atoms- a)by gaining electrons
- b)by losing electr
- c)by sharing electrons
- d)none of the above
Correct answer is option 'C'. Can you explain this answer?
Carbon forms bonds with other atoms
a)
by gaining electrons
b)
by losing electr
c)
by sharing electrons
d)
none of the above
| | Gaurav Kumar answered |
Carbon forms bonds with other atoms by sharing electrons because it requires a lot of energy to remove four electrons. Adding four electrons to carbon de-stabalises it. Therefore, it completes its octet by sharing electrons.
Which of the following represents saponification reaction ?- a)
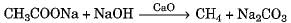
- b)
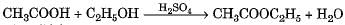
- c)
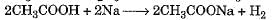
- d)
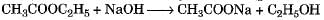
Correct answer is option 'D'. Can you explain this answer?
Which of the following represents saponification reaction ?
a)
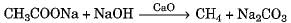
b)
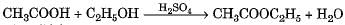
c)
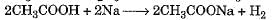
d)
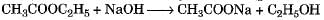
| | Anita Menon answered |
Hydrolysis of an ester with alkali is called saponification reaction.
Which of the following is an olfactory indicator?- a)Litmus
- b)Phenolphthalein
- c)Onion
- d)Methyl orange
Correct answer is option 'C'. Can you explain this answer?
Which of the following is an olfactory indicator?
a)
Litmus
b)
Phenolphthalein
c)
Onion
d)
Methyl orange
| | Amit Sharma answered |
An olfactory indicator is a material whose smell varies reliant on whether it is mixed with an acidic or basic solution. Olfactory indicators mainly used in laboratory to test whether a solution is a base or an acid. Onion is an example of olfactory indicators.
Which of the following triads is a Dobereiner’s Triad?- a)N, P, As
- b)S, O, H
- c)Na, Ra, Al
- d)Li, Na, K
Correct answer is option 'D'. Can you explain this answer?
Which of the following triads is a Dobereiner’s Triad?
a)
N, P, As
b)
S, O, H
c)
Na, Ra, Al
d)
Li, Na, K
| | Anjana Khatri answered |
DOBEREINER TRIADS RULE :
In 1817, Johann walfgang dobereiner. A German chemist, arranged the elements is group of three elements and in a manner that the atomic mass of middle element was roughly the average of the atomic masses of the other two elements of the triad.
Example:
In the triad of lithium, sodium and potassium. The atomic mass of lithium is 7 and the atomic mass of potassium is 39. The average of masses of lithium and potassium gives atomic mass of sodium 23.
Calcium sulphate hemihydrate is known as:- a)Plaster of Paris
- b)Soda ash
- c)Washing soda
- d)Gypsum
Correct answer is option 'A'. Can you explain this answer?
Calcium sulphate hemihydrate is known as:
a)
Plaster of Paris
b)
Soda ash
c)
Washing soda
d)
Gypsum
| | Rajiv Gupta answered |
The name plaster of paris (POP) is derived as the calcium sulphate hemi hydrates are found in large amount deposited in the Montmartre hill in Paris
Plaster of Paris is obtained by heating gypsum or calcium sulphate dihydrate to about 140-180 degree Celsius.
When heated to such a temperature, gypsum forms Plaster of Paris.
The name is derived from the large deposits of gypsum in the Montmartre hill in Paris.
MCQ (Multiple Choice Questions) or Practice Quiz with solutions of Chapter - "Periodic Classification of Elements" of Class 10 Science, the questions are available for practice Q. According to IUPAC recommendations, the number of groups in the long form of the periodic table is :-- a)7
- b)8
- c)16
- d)18
Correct answer is option 'D'. Can you explain this answer?
MCQ (Multiple Choice Questions) or Practice Quiz with solutions of Chapter - "Periodic Classification of Elements" of Class 10 Science, the questions are available for practice
Q. According to IUPAC recommendations, the number of groups in the long form of the periodic table is :-
a)
7
b)
8
c)
16
d)
18
| | Krishna Iyer answered |
In chemistry, a group (also known as a family) is a column of elements in the periodic table of the chemical elements. There are 18 numbered groups in the periodic table.
Which among the following are unsaturated hydrocarbons ?
(i) H3C -CH2-CH2-CH3
(ii) H3C -C = C-CH3
(iii) 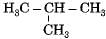
(iv) 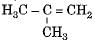
- a)(i) and (iii)
- b)(i) and (iii)
- c)(ii) and (iv)
- d)(iii) and (iv)
Correct answer is option 'C'. Can you explain this answer?
Which among the following are unsaturated hydrocarbons ?
(i) H3C -CH2-CH2-CH3
(ii) H3C -C = C-CH3
(iii)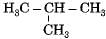
(iv)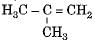
(i) H3C -CH2-CH2-CH3
(ii) H3C -C = C-CH3
(iii)
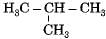
(iv)
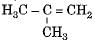
a)
(i) and (iii)
b)
(i) and (iii)
c)
(ii) and (iv)
d)
(iii) and (iv)
| | Arun Sharma answered |
Compounds of carbon and hydrogen containing double or triple bonds in the molecule are called unsaturated hydrocarbons.
Red cabbage indicator turns _______ in basic solutions.- a)Pink
- b)Green
- c)Blue
- d)Red
Correct answer is 'B'. Can you explain this answer?
Red cabbage indicator turns _______ in basic solutions.
a)
Pink
b)
Green
c)
Blue
d)
Red
 | Ruba answered |
This is a natural indicator which are found in the nature of the plants
Mostly oxides of non-metals are:- a)Alkaline
- b)Basic
- c)Amphoteric
- d)Acidic
Correct answer is option 'D'. Can you explain this answer?
Mostly oxides of non-metals are:
a)
Alkaline
b)
Basic
c)
Amphoteric
d)
Acidic
| | Srishti Gupta answered |
Acidity and Basicity of Oxides:
Oxides are binary compounds of oxygen with another element. Depending on the nature of the element, oxides can be acidic, basic, amphoteric or neutral.
Acidic Oxides:
Acidic oxides are oxides of non-metals. They react with water to form acids. For example:
1. CO2 + H2O → H2CO3 (Carbon dioxide dissolves in water to form carbonic acid)
2. SO2 + H2O → H2SO3 (Sulphur dioxide dissolves in water to form sulphurous acid)
3. NO2 + H2O → HNO3 (Nitrogen dioxide dissolves in water to form nitric acid)
4. P4O10 + 6H2O → 4H3PO4 (Phosphorus pentoxide dissolves in water to form phosphoric acid)
Basic Oxides:
Basic oxides are oxides of metals. They react with water to form bases. For example:
1. Na2O + H2O → 2NaOH (Sodium oxide dissolves in water to form sodium hydroxide)
2. MgO + H2O → Mg(OH)2 (Magnesium oxide dissolves in water to form magnesium hydroxide)
3. CaO + H2O → Ca(OH)2 (Calcium oxide dissolves in water to form calcium hydroxide)
Amphoteric Oxides:
Amphoteric oxides are oxides that can act as both acidic and basic depending on the nature of the reacting species. For example:
1. Al2O3 + 6HCl → 2AlCl3 + 3H2O (Aluminium oxide reacts with hydrochloric acid to form aluminium chloride and water)
2. ZnO + 2HCl → ZnCl2 + H2O (Zinc oxide reacts with hydrochloric acid to form zinc chloride and water)
Neutral Oxides:
Neutral oxides are oxides that do not react with water to form either an acid or a base. For example:
1. CO (Carbon monoxide)
2. N2O (Nitrous oxide)
3. NO (Nitric oxide)
Conclusion:
Mostly oxides of non-metals are acidic in nature as they react with water to form acids. Hence, the correct answer is option 'D'.
Oxides are binary compounds of oxygen with another element. Depending on the nature of the element, oxides can be acidic, basic, amphoteric or neutral.
Acidic Oxides:
Acidic oxides are oxides of non-metals. They react with water to form acids. For example:
1. CO2 + H2O → H2CO3 (Carbon dioxide dissolves in water to form carbonic acid)
2. SO2 + H2O → H2SO3 (Sulphur dioxide dissolves in water to form sulphurous acid)
3. NO2 + H2O → HNO3 (Nitrogen dioxide dissolves in water to form nitric acid)
4. P4O10 + 6H2O → 4H3PO4 (Phosphorus pentoxide dissolves in water to form phosphoric acid)
Basic Oxides:
Basic oxides are oxides of metals. They react with water to form bases. For example:
1. Na2O + H2O → 2NaOH (Sodium oxide dissolves in water to form sodium hydroxide)
2. MgO + H2O → Mg(OH)2 (Magnesium oxide dissolves in water to form magnesium hydroxide)
3. CaO + H2O → Ca(OH)2 (Calcium oxide dissolves in water to form calcium hydroxide)
Amphoteric Oxides:
Amphoteric oxides are oxides that can act as both acidic and basic depending on the nature of the reacting species. For example:
1. Al2O3 + 6HCl → 2AlCl3 + 3H2O (Aluminium oxide reacts with hydrochloric acid to form aluminium chloride and water)
2. ZnO + 2HCl → ZnCl2 + H2O (Zinc oxide reacts with hydrochloric acid to form zinc chloride and water)
Neutral Oxides:
Neutral oxides are oxides that do not react with water to form either an acid or a base. For example:
1. CO (Carbon monoxide)
2. N2O (Nitrous oxide)
3. NO (Nitric oxide)
Conclusion:
Mostly oxides of non-metals are acidic in nature as they react with water to form acids. Hence, the correct answer is option 'D'.
Chapter doubts & questions for Class 10 NCERT Chemistry - 6 Months Preparation Course for OPSC 2026 is part of OPSC OCS (Odisha) exam preparation. The chapters have been prepared according to the OPSC OCS (Odisha) exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for OPSC OCS (Odisha) 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Class 10 NCERT Chemistry - 6 Months Preparation Course for OPSC in English & Hindi are available as part of OPSC OCS (Odisha) exam. Download more important topics, notes, lectures and mock test series for OPSC OCS (Odisha) Exam by signing up for free.
6 Months Preparation Course for OPSC636 videos|2472 docs|801 tests |
Top Courses OPSC OCS (Odisha)
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup

