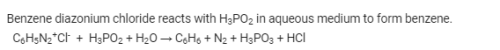All Exams > NEET > 3 Months Class 12 Board Preparation (Medical) > All Questions
All questions of Amines for NEET Exam
Hoffmann Bromamide Degradation reaction is shown by __________.- a) ArNH2
- b)ArCONH2
- c)ArNO2
- d)ArCH2NH2
Correct answer is option 'B'. Can you explain this answer?
Hoffmann Bromamide Degradation reaction is shown by __________.
a)
ArNH2
b)
ArCONH2
c)
ArNO2
d)
ArCH2NH2
| | Hansa Sharma answered |
The Correct answer is option B
Hoffman bromamide degradation reaction is shown by ArCONH2.
Where the aryl amide is converted to aryl amine in the presence of Br2 and NaOH
Hoffman bromamide degradation reaction is shown by ArCONH2.
Where the aryl amide is converted to aryl amine in the presence of Br2 and NaOH

Which of the following has highest boiling point?- a)(C2H5)2NH
- b)C2H5N(CH3)2
- c)C2H5NH(CH3)2
- d)n-C4H9NH2
Correct answer is option 'D'. Can you explain this answer?
Which of the following has highest boiling point?
a)
(C2H5)2NH
b)
C2H5N(CH3)2
c)
C2H5NH(CH3)2
d)
n-C4H9NH2
| | Krishna Iyer answered |
The correct answer is Option D.
Because C4H9NH2 is primary amine and can form hydrogen bonding more than secondary amine (C2H5)2NH and tertiary amine C2H5N(CH3)2 . More hydrogen bonding leads to strong bonding between the molecules, hence increases the boiling point.
Nitro compounds are reduced to amines. The catalyst that is preferred is:- a)Sn + HCl
- b)Fe + HCl
- c)Ethanol
- d)Mg + HCl
Correct answer is option 'B'. Can you explain this answer?
Nitro compounds are reduced to amines. The catalyst that is preferred is:
a)
Sn + HCl
b)
Fe + HCl
c)
Ethanol
d)
Mg + HCl
 | Top Rankers answered |
Fe + HCl is preferred due to the following reasons:
- Scrap iron i.e Fe is cheap and commercially easily available
- FeCl2 formed will get hydrolysed and release HCl, so, HCl which is required in the reaction will be produced by itself
- Hence only small amount of HCl is required to initiate the reaction.
- Scrap iron i.e Fe is cheap and commercially easily available
- FeCl2 formed will get hydrolysed and release HCl, so, HCl which is required in the reaction will be produced by itself
- Hence only small amount of HCl is required to initiate the reaction.
Which of the following amine liberates nitrogen gas on reaction with HNO2 ?- a)(CH3)2NH
- b)(CH3)3 N
- c)C6H5 NH2
- d)CH3NH2
Correct answer is option 'D'. Can you explain this answer?
Which of the following amine liberates nitrogen gas on reaction with HNO2 ?
a)
(CH3)2NH
b)
(CH3)3 N
c)
C6H5 NH2
d)
CH3NH2
| | Krishna Iyer answered |
The correct answer is Option D.
Only primary amines liberate nitrogen gas on reaction with HNO2.
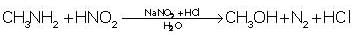
Only primary amines liberate nitrogen gas on reaction with HNO2.
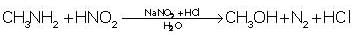
When hypophosphorous acid is treated with diazonium salts, it is reduced to:- a)Arenes
- b)Methane
- c)Ethyl alcohol
- d)Amines
Correct answer is option 'A'. Can you explain this answer?
When hypophosphorous acid is treated with diazonium salts, it is reduced to:
a)
Arenes
b)
Methane
c)
Ethyl alcohol
d)
Amines
 | Mohit Rajpoot answered |
The correct answer is option A
Hypophosphorous acid when treated with diazonium salts is reduced to arenes.
ArN2Cl + H3PO2 + H2O ⟶ ArH + N2 + H3PO3 +HCl
ArN2Cl + H3PO2 + H2O ⟶ ArH + N2 + H3PO3 +HCl
Which reaction can be used for the direct conversion of amides into 10 amine ?- a)Reduction with LiAlH4
- b)Perkin’s reaction
- c)Claissen reaction
- d)Hoffmann ammonolysis
Correct answer is option 'A'. Can you explain this answer?
Which reaction can be used for the direct conversion of amides into 10 amine ?
a)
Reduction with LiAlH4
b)
Perkin’s reaction
c)
Claissen reaction
d)
Hoffmann ammonolysis
 | Gunjan Lakhani answered |
The correct answer is option A
Reduction with LiAlH4
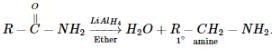
Reduction with LiAlH4
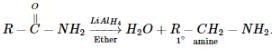
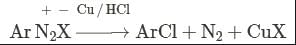
- a)Sandmeyers reaction
- b)Gattermanns reaction
- c)Dehydrogenation reaction
- d)Esterification reaction
Correct answer is option 'B'. Can you explain this answer?
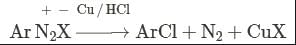
a)
Sandmeyers reaction
b)
Gattermanns reaction
c)
Dehydrogenation reaction
d)
Esterification reaction
 | Mohit Rajpoot answered |
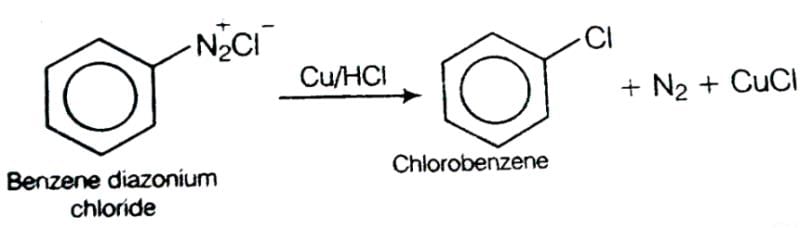 This reaction is called Gattermann reaction. In this reaction, Cl, Br and CN can be introduced into the benzene ring by simply treating diazonium salts with HCl, HBr, KCN. Respectively in presnce of copper powder instead of using Cu(I) salts.
This reaction is called Gattermann reaction. In this reaction, Cl, Br and CN can be introduced into the benzene ring by simply treating diazonium salts with HCl, HBr, KCN. Respectively in presnce of copper powder instead of using Cu(I) salts. When Primary amide is treated with an aqueous solution of KOH and bromine, it gives a primary amine. The name of the reaction is:- a)Gabriel-phthalamine reaction
- b)Reimer-Tiemann reaction
- c)Ammonolysis
- d)Hoffmann Bromamide reaction
Correct answer is option 'D'. Can you explain this answer?
When Primary amide is treated with an aqueous solution of KOH and bromine, it gives a primary amine. The name of the reaction is:
a)
Gabriel-phthalamine reaction
b)
Reimer-Tiemann reaction
c)
Ammonolysis
d)
Hoffmann Bromamide reaction
 | Gunjan Lakhani answered |
Hoffmann Bromamide reaction is Treatment of alkylamide with KOH and Bromine gas which converts it into alkylamine with one less carbon atom, this reaction is generally helpful in converting Carboxylic acids to amines with one less carbon atom . For reaction you may refer to internet but i can give you a hint .
Acetamide + KOH + Bromine gas —-> Alkylamine+ KBr + K2CO3 + H20 ( not sure about the products)
Which of the following amine gives diazonium salt on reaction with HNO2?- a)(CH3)2NH
- b)CH3NH2
- c)C6H5NH2
- d)(CH3)3N
Correct answer is option 'C'. Can you explain this answer?
Which of the following amine gives diazonium salt on reaction with HNO2?
a)
(CH3)2NH
b)
CH3NH2
c)
C6H5NH2
d)
(CH3)3N
 | Mohit Rajpoot answered |
C6H5NH2 reacts with HNO2 to forms diazonium salts, the reaction are as follows,
C6H5NH2 + HNO2 ------> C6H5OH + H2O + N2
C6H5NH2 + HNO2 ------> C6H5OH + H2O + N2
Which of the following amine will form stable diazonium salt at 273-283 K ?- a)C6H5NH2
- b)C6H5N(CH3)2
- c)C2H5NH2
- d)C6H5CH2NH2
Correct answer is option 'A'. Can you explain this answer?
Which of the following amine will form stable diazonium salt at 273-283 K ?
a)
C6H5NH2
b)
C6H5N(CH3)2
c)
C2H5NH2
d)
C6H5CH2NH2
 | Sushil Kumar answered |
The correct answer is Option A.
Aromatic Primary amine will form the most stable diazonium salt because it releases water when it reacts with nitronium ions. If aliphatic primary amine reacts with nitrosonium ion it Also releases water but in this case water reacts with alkyl diazonium salt and it forms alcohol while benzene diazonium salt does not react with water at this temperature.
Aromatic Primary amine will form the most stable diazonium salt because it releases water when it reacts with nitronium ions. If aliphatic primary amine reacts with nitrosonium ion it Also releases water but in this case water reacts with alkyl diazonium salt and it forms alcohol while benzene diazonium salt does not react with water at this temperature.
The following reaction takes place in the presence of 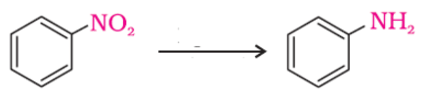
- a)NaOH/Pd
- b)H2/Pd
- c)HCl/Pd
- d)None of these
Correct answer is option 'B'. Can you explain this answer?
The following reaction takes place in the presence of
a)
NaOH/Pd
b)
H2/Pd
c)
HCl/Pd
d)
None of these
| | Anaya Patel answered |
−NO2 group is reduced to –NH2 using H2/Pd.
The stability of benzene diazonium salts is because of- a)Inductive effect
- b)Mesomeric effect
- c)Hyperconjugation
- d)Resonance
Correct answer is option 'D'. Can you explain this answer?
The stability of benzene diazonium salts is because of
a)
Inductive effect
b)
Mesomeric effect
c)
Hyperconjugation
d)
Resonance
| | Geetika Shah answered |
None of the errors that are pointed are correct.
Their stability is because of resonance. D is the correct answer.
We can obtain ethylamine by Hoffmann bromamide reaction. The amide used in this reaction is:- a)Methanamide
- b)Acetamide
- c)Propanamide
- d)Butanamide
Correct answer is option 'C'. Can you explain this answer?
We can obtain ethylamine by Hoffmann bromamide reaction. The amide used in this reaction is:
a)
Methanamide
b)
Acetamide
c)
Propanamide
d)
Butanamide
 | Gunjan Lakhani answered |
The correct answer is option C
CH3CH2CONH2 (A)⟶ CH3 − CH2 − NH2 (B)⟶ CH3 − CH2 − OH
In the above sequence A & B respectively are Br2/KOH and HNO2
The first step is Hoffmann bromamide degradation reaction in which an amide (propanamide) is converted to an amine (ethylamine) containing one carbon atom less. The reagent A is bromine in presence of KOH. In the second step, aliphatic primary amine (ethyl amine) reacts with nitrous acid (reagent B) to form aliphatic primary alcohol (ethyl alcohol).
CH3CH2CONH2 (A)⟶ CH3 − CH2 − NH2 (B)⟶ CH3 − CH2 − OH
In the above sequence A & B respectively are Br2/KOH and HNO2
The first step is Hoffmann bromamide degradation reaction in which an amide (propanamide) is converted to an amine (ethylamine) containing one carbon atom less. The reagent A is bromine in presence of KOH. In the second step, aliphatic primary amine (ethyl amine) reacts with nitrous acid (reagent B) to form aliphatic primary alcohol (ethyl alcohol).
The molecular formula of ethyl acetate is- a)C4H8O
- b)C4H8O2
- c)C5H10O2
- d)C5H8O2
Correct answer is option 'B'. Can you explain this answer?
The molecular formula of ethyl acetate is
a)
C4H8O
b)
C4H8O2
c)
C5H10O2
d)
C5H8O2
 | Tejas Singh answered |
Its molecular formula is C4H8O2
. When diazonium salt solution is treated with KI, it forms:- a)Bromobenzene
- b)Iodobenzene
- c)Phenol
- d)Acid
Correct answer is option 'B'. Can you explain this answer?
. When diazonium salt solution is treated with KI, it forms:
a)
Bromobenzene
b)
Iodobenzene
c)
Phenol
d)
Acid
 | Kshitij Pandey answered |
This reaction is unique this is only reaction in which idobenzene formed by diaazonium salt , to make idobenzene first we make diaazonium salt by aniline then we treated diaazonium salt with ki
C6H5N2Cl(diaazonium salt)+KI=C6H5I
C6H5N2Cl(diaazonium salt)+KI=C6H5I
Replacement of diazo group by other groups is helpful in preparing those substituted aromatic compounds which cannot be prepared by- a)Direct elimination
- b)Addition reaction
- c)Direct substitution
- d)Replacement reaction
Correct answer is option 'C'. Can you explain this answer?
Replacement of diazo group by other groups is helpful in preparing those substituted aromatic compounds which cannot be prepared by
a)
Direct elimination
b)
Addition reaction
c)
Direct substitution
d)
Replacement reaction
| | Vikas Kumar answered |
Replacement of diazo group by other groups is helpful in preparing those substituted aromatic compounds which cannot be prepared by direct substitution. ... The cyano group usually cannot be introduced by nucleophilic substitution of haloarenes, but such compounds can be easily prepared from diazonium salts.
Which one of the following is a tertiary amine?- a)Ethylamine
- b)Triethylamine
- c)Aniline
- d)Diethylamine
Correct answer is option 'B'. Can you explain this answer?
Which one of the following is a tertiary amine?
a)
Ethylamine
b)
Triethylamine
c)
Aniline
d)
Diethylamine
| | Suresh Iyer answered |
The correct answer is Option B.
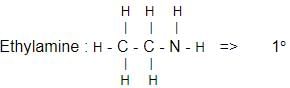
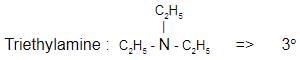
Aniline :
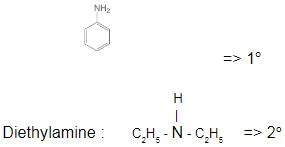
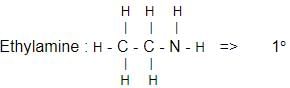
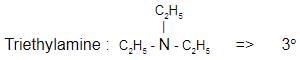
Aniline :

The main product formed by treating an alkyl or benzyl halide with excess ammonia- a)Mixed
- b)Tertiary
- c)Secondary
- d)Primary
Correct answer is option 'D'. Can you explain this answer?
The main product formed by treating an alkyl or benzyl halide with excess ammonia
a)
Mixed
b)
Tertiary
c)
Secondary
d)
Primary
| | Amita Das answered |
The N in ammonia functions as the nucleophile and attacks the electrophilic C of the alkyl halide displacing the bromide and creating the new C-N bond.
Step 2: An acid/base reaction. The base (excess ammonia) deprotonates the positive N (ammonium) center creating the alkylation product, the primary amine.
To convert methyl cyanide to ethylamine we use:- a)Br2
- b)HNO3
- c)KOH
- d)LiAlH4
Correct answer is option 'D'. Can you explain this answer?
To convert methyl cyanide to ethylamine we use:
a)
Br2
b)
HNO3
c)
KOH
d)
LiAlH4
| | Riya Banerjee answered |
LiAlH4 reduces all oxiginated functional groups to alcohol and all nitrogenated functional groups to amine thus cyanide is a nitrogenated functional group and to convert cyanide to amine LiAlH4 can be used.
When diazonium salt solution is treated with water at a temperature of 283 K it forms?- a)Ester
- b)Phenol
- c)Amines
- d)Alcohol
Correct answer is option 'B'. Can you explain this answer?
When diazonium salt solution is treated with water at a temperature of 283 K it forms?
a)
Ester
b)
Phenol
c)
Amines
d)
Alcohol
 | Vatturi Anjani answered |
Yes as when the temperature of the diazonium salt is allowed to rise upto 283K then the salt is reduced to phenol releasing N2 and hydrochloric acid as the by-products.
Benzene diazonium chloride on reaction with phenol in weakly basic medium gives:- a)p-Hydroxyazobenzene
- b)Benzene
- c)Diphenyl ether
- d)Chlorobenzene
Correct answer is option 'A'. Can you explain this answer?
Benzene diazonium chloride on reaction with phenol in weakly basic medium gives:
a)
p-Hydroxyazobenzene
b)
Benzene
c)
Diphenyl ether
d)
Chlorobenzene
| | Arka Das answered |
**Benzene diazonium chloride**
Benzene diazonium chloride (C6H5N2Cl) is an organic compound that is commonly used in diazotization reactions. It is formed by the reaction of aniline (C6H5NH2) with nitrous acid (HNO2) in the presence of hydrochloric acid (HCl).
**Reaction with Phenol**
When benzene diazonium chloride reacts with phenol (C6H5OH) in a weakly basic medium, it undergoes a substitution reaction known as the Sandmeyer reaction. This reaction involves the replacement of the diazonium group (-N2Cl) with the phenolic group (-OH) to form a new compound.
**Formation of p-Hydroxyazobenzene**
The reaction between benzene diazonium chloride and phenol results in the formation of p-hydroxyazobenzene (C12H10N2O), which is the correct answer (option A).
The reaction proceeds as follows:
1. The weakly basic medium provides the necessary conditions for the reaction to occur. It helps in the deprotonation of phenol to form the phenoxide ion (C6H5O-).
2. The diazonium group of benzene diazonium chloride is highly reactive and undergoes nucleophilic substitution. The nitrogen atom of the diazonium group attacks the phenoxide ion, leading to the formation of a new carbon-nitrogen bond.
3. The chlorine atom attached to the nitrogen atom is replaced by the phenoxide group, resulting in the formation of p-hydroxyazobenzene.
The reaction can be represented by the following equation:
C6H5N2Cl + C6H5O- → C12H10N2O + Cl-
**Explanation of Other Options**
Option B: Benzenedoes not participate in the reaction. It remains unchanged.
Option C: Diphenyl ether is not formed in this reaction. It involves the formation of a carbon-oxygen bond between two phenyl groups, which is not observed in the given reaction.
Option D: Chlorobenzene is not formed in this reaction. The chlorine atom from the diazonium chloride is replaced by the phenoxide group, not by another chlorine atom.
Thus, the correct answer is option A: p-Hydroxyazobenzene.
Benzene diazonium chloride (C6H5N2Cl) is an organic compound that is commonly used in diazotization reactions. It is formed by the reaction of aniline (C6H5NH2) with nitrous acid (HNO2) in the presence of hydrochloric acid (HCl).
**Reaction with Phenol**
When benzene diazonium chloride reacts with phenol (C6H5OH) in a weakly basic medium, it undergoes a substitution reaction known as the Sandmeyer reaction. This reaction involves the replacement of the diazonium group (-N2Cl) with the phenolic group (-OH) to form a new compound.
**Formation of p-Hydroxyazobenzene**
The reaction between benzene diazonium chloride and phenol results in the formation of p-hydroxyazobenzene (C12H10N2O), which is the correct answer (option A).
The reaction proceeds as follows:
1. The weakly basic medium provides the necessary conditions for the reaction to occur. It helps in the deprotonation of phenol to form the phenoxide ion (C6H5O-).
2. The diazonium group of benzene diazonium chloride is highly reactive and undergoes nucleophilic substitution. The nitrogen atom of the diazonium group attacks the phenoxide ion, leading to the formation of a new carbon-nitrogen bond.
3. The chlorine atom attached to the nitrogen atom is replaced by the phenoxide group, resulting in the formation of p-hydroxyazobenzene.
The reaction can be represented by the following equation:
C6H5N2Cl + C6H5O- → C12H10N2O + Cl-
**Explanation of Other Options**
Option B: Benzenedoes not participate in the reaction. It remains unchanged.
Option C: Diphenyl ether is not formed in this reaction. It involves the formation of a carbon-oxygen bond between two phenyl groups, which is not observed in the given reaction.
Option D: Chlorobenzene is not formed in this reaction. The chlorine atom from the diazonium chloride is replaced by the phenoxide group, not by another chlorine atom.
Thus, the correct answer is option A: p-Hydroxyazobenzene.
By treating diazonium salts with cuprous cyanide or KCN and copper powder it forms:- a)Citric acid
- b)Benzoic acid
- c)Aryl nitrile
- d)Oxalic acid
Correct answer is option 'C'. Can you explain this answer?
By treating diazonium salts with cuprous cyanide or KCN and copper powder it forms:
a)
Citric acid
b)
Benzoic acid
c)
Aryl nitrile
d)
Oxalic acid
| | Rishika Patel answered |
Formation of Aryl Nitrile from Diazonium Salts
Diazonium salts are compounds containing a positively charged nitrogen atom that is linked to an aromatic ring. These salts are often used in organic synthesis as a source of the aryl group. When treated with cuprous cyanide or KCN and copper powder, diazonium salts undergo a reaction known as Sandmeyer reaction, which forms aryl nitriles.
Reaction Mechanism
The reaction mechanism involves several steps:
1. Formation of Copper(I) Salt
First, cuprous cyanide or KCN is added to the diazonium salt solution to form a copper(I) salt. The copper(I) salt plays a crucial role in the reaction by catalyzing the formation of the aryl nitrile.
2. Formation of Aryl Copper(I) Intermediate
Next, copper powder is added to the reaction mixture, which reduces the copper(I) salt to copper metal. The copper metal then reacts with the diazonium salt to form an aryl copper(I) intermediate.
3. Formation of Aryl Cyanide
The aryl copper(I) intermediate then reacts with the cyanide ion from the cuprous cyanide or KCN to form the aryl cyanide. The reaction releases copper metal, which can then react with more diazonium salt to form more aryl copper(I) intermediate.
Overall Reaction
The overall reaction can be represented as follows:
ArN2+X- + CuCN/KCN → [CuX] + N2 + ArCu + HX
ArCu + CN- → ArCN + Cu
Where Ar represents the aryl group, X represents the anion of the diazonium salt, and HX represents the acid formed in the reaction.
Conclusion
In conclusion, the treatment of diazonium salts with cuprous cyanide or KCN and copper powder forms aryl nitriles through the Sandmeyer reaction. This reaction is an important method for the synthesis of aryl nitriles, which have a variety of applications in organic synthesis and industry.
Diazonium salts are compounds containing a positively charged nitrogen atom that is linked to an aromatic ring. These salts are often used in organic synthesis as a source of the aryl group. When treated with cuprous cyanide or KCN and copper powder, diazonium salts undergo a reaction known as Sandmeyer reaction, which forms aryl nitriles.
Reaction Mechanism
The reaction mechanism involves several steps:
1. Formation of Copper(I) Salt
First, cuprous cyanide or KCN is added to the diazonium salt solution to form a copper(I) salt. The copper(I) salt plays a crucial role in the reaction by catalyzing the formation of the aryl nitrile.
2. Formation of Aryl Copper(I) Intermediate
Next, copper powder is added to the reaction mixture, which reduces the copper(I) salt to copper metal. The copper metal then reacts with the diazonium salt to form an aryl copper(I) intermediate.
3. Formation of Aryl Cyanide
The aryl copper(I) intermediate then reacts with the cyanide ion from the cuprous cyanide or KCN to form the aryl cyanide. The reaction releases copper metal, which can then react with more diazonium salt to form more aryl copper(I) intermediate.
Overall Reaction
The overall reaction can be represented as follows:
ArN2+X- + CuCN/KCN → [CuX] + N2 + ArCu + HX
ArCu + CN- → ArCN + Cu
Where Ar represents the aryl group, X represents the anion of the diazonium salt, and HX represents the acid formed in the reaction.
Conclusion
In conclusion, the treatment of diazonium salts with cuprous cyanide or KCN and copper powder forms aryl nitriles through the Sandmeyer reaction. This reaction is an important method for the synthesis of aryl nitriles, which have a variety of applications in organic synthesis and industry.
Amines are basic in nature as they have:- a)Replaceable H atom
- b)A lone pair of electrons on nitrogen
- c)Replaceable N-H group
- d)A hydroxyl group in the molecule
Correct answer is option 'B'. Can you explain this answer?
Amines are basic in nature as they have:
a)
Replaceable H atom
b)
A lone pair of electrons on nitrogen
c)
Replaceable N-H group
d)
A hydroxyl group in the molecule
 | Rishita Rai answered |
Correct option is b because as there is lone pair on nitrogen they possess a lone pair that can be donated that's why amines are bases
The Hofmann elimination proceeds via a(n) __________ pathway.- a)SN2
- b)E2
- c)E1
- d)SN1
Correct answer is option 'B'. Can you explain this answer?
The Hofmann elimination proceeds via a(n) __________ pathway.
a)
SN2
b)
E2
c)
E1
d)
SN1
| | Rajat Kapoor answered |
The Hofmann Elimination is an elimination reaction that forms C-C double (pi) bonds that specifically occurs when the leaving group is NR3 [note] It proceeds through an E2 mechanism. Although the key concepts are no different than one learns in the chapter on elimination from way back in Org 1, it is often included in Org 2 as part of the grab-bag chapter on amines. Because, well… nitrogen.
p-amino azo benzene is obtained by treating diazoniumchloride with:- a)Phenol
- b)Aniline
- c)Alcohol
- d)Benzoic acid
Correct answer is option 'B'. Can you explain this answer?
p-amino azo benzene is obtained by treating diazoniumchloride with:
a)
Phenol
b)
Aniline
c)
Alcohol
d)
Benzoic acid
| | Lavanya Menon answered |
p-amino azo benzene is obtained by treating diazonium chloride with aniline. The reactions are specifically acid catalyzed and involve pre‐equilibrium formation of amine and diazonium salt followed by rate‐limiting attack of the diazonium ion at a C‐atom (C‐coupling) to give the corresponding amino azo compounds.
Hinsberg’s reagent reacts with primary and secondary amines to form sulphonamides. This reagent is also known as- a)N Methylbenzamide
- b)p – toluenesulphonyl chloride
- c)None of these
- d)Benzenesulphonyl chloride
Correct answer is option 'D'. Can you explain this answer?
Hinsberg’s reagent reacts with primary and secondary amines to form sulphonamides. This reagent is also known as
a)
N Methylbenzamide
b)
p – toluenesulphonyl chloride
c)
None of these
d)
Benzenesulphonyl chloride
 | Mohit Patel answered |
C6H5SO2Cl this is benzenesulphonyl chloride or hinsberg reagent.
Diazonium salts are used in the preparation of- a)Hormones
- b)Vitamins
- c)Dyes
- d)Proteins
Correct answer is option 'C'. Can you explain this answer?
Diazonium salts are used in the preparation of
a)
Hormones
b)
Vitamins
c)
Dyes
d)
Proteins
| | Vijay Bansal answered |
A diazonium salt is an organic compound that contains a nitrogen-nitrogen triple bond and some other generic side group that could be either alkyl (an alkane derivative) or aryl (benzene ring). The 'salt' portion of the name comes from the fact that the diazo (meaning 'di-nitrogen') portion of the compound is present as its ionic salt, with a chloride ion being a typical counter-ion for the positively charged nitrogen atom.
Which one of the following is the weakest base in gaseous phase?- a)Triethyl amine
- b)Ethyl amine
- c)Diethyl amine
- d)Ammonia
Correct answer is option 'D'. Can you explain this answer?
Which one of the following is the weakest base in gaseous phase?
a)
Triethyl amine
b)
Ethyl amine
c)
Diethyl amine
d)
Ammonia
| | Rajat Kapoor answered |
As the number of alkyl group increases, due to +I effect the basicity of amines increases.
Directions: These questions consist of two statements, each printed as Assertion and Reason. While answering these questions, you are required to choose any one of the following four responses.Assertion : Nitrating mixture used for carrying out nitration of benzene consists of conc. HNO3+ conc. H2SO4Reason : In presence of H2SO4, HNO3 acts as a base and produces NO2+ ions.- a)If both Assertion and Reason are correct and the Reason is a correct explanation of the Assertion.
- b)If both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion.
- c)If the Assertion is correct but Reason is incorrect.
- d)If both the Assertion and Reason are incorrect.
Correct answer is option 'A'. Can you explain this answer?
Directions: These questions consist of two statements, each printed as Assertion and Reason. While answering these questions, you are required to choose any one of the following four responses.
Assertion : Nitrating mixture used for carrying out nitration of benzene consists of conc. HNO3+ conc. H2SO4
Reason : In presence of H2SO4, HNO3 acts as a base and produces NO2+ ions.
a)
If both Assertion and Reason are correct and the Reason is a correct explanation of the Assertion.
b)
If both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion.
c)
If the Assertion is correct but Reason is incorrect.
d)
If both the Assertion and Reason are incorrect.
| | Kirti Kaur answered |
Assertion: Nitrating mixture used for carrying out nitration of benzene consists of concentrated HNO3 and concentrated H2SO4.
Reason: In the presence of H2SO4, HNO3 acts as a base and produces NO2 ions.
Explanation:
The nitration of benzene is a chemical reaction in which a nitro group (-NO2) is introduced into the benzene ring. This reaction is carried out using a nitrating mixture, which typically consists of concentrated nitric acid (HNO3) and concentrated sulfuric acid (H2SO4).
Reasoning:
The presence of concentrated sulfuric acid (H2SO4) in the nitrating mixture is crucial for the nitration reaction. H2SO4 acts as a catalyst and plays a vital role in the reaction. When HNO3 is mixed with H2SO4, it forms an intermediate species called nitronium ion (NO2+). This nitronium ion is highly reactive and serves as an electrophile in the nitration reaction.
Assertion Explanation:
The assertion states that the nitrating mixture used for benzene nitration consists of concentrated HNO3 and concentrated H2SO4. This is true because both acids are essential for the successful nitration of benzene. Concentrated HNO3 provides the nitro group, and concentrated H2SO4 acts as a catalyst and helps in the formation of the electrophilic nitronium ion.
Reason Explanation:
The reason given is that in the presence of H2SO4, HNO3 acts as a base and produces NO2 ions. This statement is partially correct. HNO3 is not acting as a base in this reaction but rather as a reactant. H2SO4, on the other hand, acts as a catalyst and facilitates the formation of the nitronium ion (NO2+), which is responsible for the nitration of benzene.
Conclusion:
Both the assertion and the reason are correct. However, the reason is not a correct explanation of the assertion. The reason incorrectly states that HNO3 acts as a base in the presence of H2SO4, while in reality, it acts as a reactant to form the nitronium ion. Therefore, the correct answer is option 'b'.
Reason: In the presence of H2SO4, HNO3 acts as a base and produces NO2 ions.
Explanation:
The nitration of benzene is a chemical reaction in which a nitro group (-NO2) is introduced into the benzene ring. This reaction is carried out using a nitrating mixture, which typically consists of concentrated nitric acid (HNO3) and concentrated sulfuric acid (H2SO4).
Reasoning:
The presence of concentrated sulfuric acid (H2SO4) in the nitrating mixture is crucial for the nitration reaction. H2SO4 acts as a catalyst and plays a vital role in the reaction. When HNO3 is mixed with H2SO4, it forms an intermediate species called nitronium ion (NO2+). This nitronium ion is highly reactive and serves as an electrophile in the nitration reaction.
Assertion Explanation:
The assertion states that the nitrating mixture used for benzene nitration consists of concentrated HNO3 and concentrated H2SO4. This is true because both acids are essential for the successful nitration of benzene. Concentrated HNO3 provides the nitro group, and concentrated H2SO4 acts as a catalyst and helps in the formation of the electrophilic nitronium ion.
Reason Explanation:
The reason given is that in the presence of H2SO4, HNO3 acts as a base and produces NO2 ions. This statement is partially correct. HNO3 is not acting as a base in this reaction but rather as a reactant. H2SO4, on the other hand, acts as a catalyst and facilitates the formation of the nitronium ion (NO2+), which is responsible for the nitration of benzene.
Conclusion:
Both the assertion and the reason are correct. However, the reason is not a correct explanation of the assertion. The reason incorrectly states that HNO3 acts as a base in the presence of H2SO4, while in reality, it acts as a reactant to form the nitronium ion. Therefore, the correct answer is option 'b'.
Which of the following amines is most soluble in water?- a)(CH3CH2CH2)2NH
- b)ethylamine
- c)pyrrolidine
- d)(CH3CH3)3N
Correct answer is option 'B'. Can you explain this answer?
Which of the following amines is most soluble in water?
a)
(CH3CH2CH2)2NH
b)
ethylamine
c)
pyrrolidine
d)
(CH3CH3)3N
 | Sankar Gupta answered |
Because to maximum hydrogen bonding.
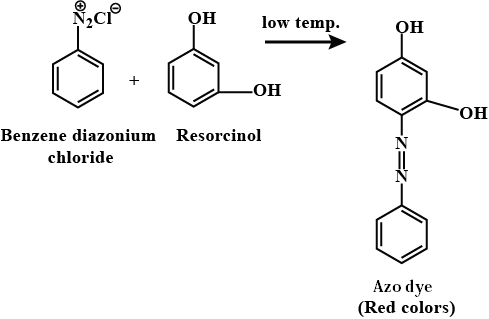
An azo compound is formed when benzene diazonium chloride reacts with:- a)2,4,6-Trimethylphenol
- b)Both (A) and (B)
- c)Phenol
- d)Aniline
Correct answer is option 'B'. Can you explain this answer?
An azo compound is formed when benzene diazonium chloride reacts with:
a)
2,4,6-Trimethylphenol
b)
Both (A) and (B)
c)
Phenol
d)
Aniline
| | Manoj Chauhan answered |
Formation of Azo Compound
An azo compound is formed when benzene diazonium chloride reacts with 2,4,6-trimethylphenol and phenol. The reaction involves the substitution of diazonium group (-N2+) with the hydroxyl group (-OH) of the phenol. The reaction is called diazotization.
Diazotization Reaction
Diazotization is a chemical reaction that involves the conversion of an aromatic primary amine to a diazonium salt by the action of nitrous acid. It is a two-step process that involves the formation of a diazonium intermediate, which then reacts with a nucleophile to form the final product.
Step 1: Diazotization of Aromatic Primary Amine
In the first step of diazotization, an aromatic primary amine is treated with nitrous acid (HNO2) to form a diazonium salt. The reaction can be represented as:
R-NH2 + HNO2 → R-N2+Cl- + H2O
where R represents the aromatic group.
Step 2: Reaction with Nucleophile
In the second step, the diazonium salt reacts with a nucleophile (such as phenol or 2,4,6-trimethylphenol) to form the final product, which is an azo compound. The reaction can be represented as:
R-N2+Cl- + Ar-OH → R-N=N-Ar + HCl + H2O
where Ar represents the aromatic ring.
Formation of Azo Compound from Benzene Diazonium Chloride
When benzene diazonium chloride reacts with 2,4,6-trimethylphenol or phenol, an azo compound is formed. The reaction can be represented as:
C6H5-N2+Cl- + Ar-OH → C6H5-N=N-Ar + HCl + H2O
where Ar represents the aromatic ring of 2,4,6-trimethylphenol or phenol.
Therefore, option B (Both A and B) is the correct answer as both 2,4,6-trimethylphenol and phenol can react with benzene diazonium chloride to form azo compounds.
An azo compound is formed when benzene diazonium chloride reacts with 2,4,6-trimethylphenol and phenol. The reaction involves the substitution of diazonium group (-N2+) with the hydroxyl group (-OH) of the phenol. The reaction is called diazotization.
Diazotization Reaction
Diazotization is a chemical reaction that involves the conversion of an aromatic primary amine to a diazonium salt by the action of nitrous acid. It is a two-step process that involves the formation of a diazonium intermediate, which then reacts with a nucleophile to form the final product.
Step 1: Diazotization of Aromatic Primary Amine
In the first step of diazotization, an aromatic primary amine is treated with nitrous acid (HNO2) to form a diazonium salt. The reaction can be represented as:
R-NH2 + HNO2 → R-N2+Cl- + H2O
where R represents the aromatic group.
Step 2: Reaction with Nucleophile
In the second step, the diazonium salt reacts with a nucleophile (such as phenol or 2,4,6-trimethylphenol) to form the final product, which is an azo compound. The reaction can be represented as:
R-N2+Cl- + Ar-OH → R-N=N-Ar + HCl + H2O
where Ar represents the aromatic ring.
Formation of Azo Compound from Benzene Diazonium Chloride
When benzene diazonium chloride reacts with 2,4,6-trimethylphenol or phenol, an azo compound is formed. The reaction can be represented as:
C6H5-N2+Cl- + Ar-OH → C6H5-N=N-Ar + HCl + H2O
where Ar represents the aromatic ring of 2,4,6-trimethylphenol or phenol.
Therefore, option B (Both A and B) is the correct answer as both 2,4,6-trimethylphenol and phenol can react with benzene diazonium chloride to form azo compounds.
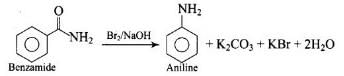
For producing amines, the reaction of nitro compounds with iron scrap is preferred because- a)HCl is very cheap
- b)HCl is formed on hydrolysis of FeCl2
- c)Not too much HCl is formed in the reaction
- d)Nitro compounds are easily available
Correct answer is option 'B'. Can you explain this answer?
For producing amines, the reaction of nitro compounds with iron scrap is preferred because
a)
HCl is very cheap
b)
HCl is formed on hydrolysis of FeCl2
c)
Not too much HCl is formed in the reaction
d)
Nitro compounds are easily available
 | Kavya Das answered |
This reaction is preferred because on reduction of nitro compouns to amine , Fe will get oxidize to Fe(II) a which on hydrolysis will produce HCl which is required in the reaction
Directions: These questions consist of two statements, each printed as Assertion and Reason. While answering these questions, you are required to choose any one of the following four responses.Assertion : Acetanilide is less basic than aniline.Reason : Acetylation of aniline results in decrease of electron density on nitrogen.- a)If both the Assertion and Reason are incorrect.
- b)If both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion.
- c)If the Assertion is correct but Reason is incorrect.
- d)If both Assertion and Reason are correct and the Reason is a correct explanation of the Assertion.
Correct answer is option 'D'. Can you explain this answer?
Directions: These questions consist of two statements, each printed as Assertion and Reason. While answering these questions, you are required to choose any one of the following four responses.
Assertion : Acetanilide is less basic than aniline.
Reason : Acetylation of aniline results in decrease of electron density on nitrogen.
a)
If both the Assertion and Reason are incorrect.
b)
If both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion.
c)
If the Assertion is correct but Reason is incorrect.
d)
If both Assertion and Reason are correct and the Reason is a correct explanation of the Assertion.
| | Shalini Patel answered |
Acetylation of aniline results in decrease of electron density on nitrogen hence acetanilide is less basic than aniline.
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.Assertion (A): N, N-Diethylbenzene sulphonamide is insoluble in alkali.Reason (R): Sulphonyl group attached to nitrogen atom is strong electron withdrawing group.- a)Both A and R are true but R is NOT the correct explanation of A
- b)Both A and R are true and R is the correct explanation of A
- c)A is true but R is false
- d)A is false and R is True
Correct answer is option 'A'. Can you explain this answer?
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): N, N-Diethylbenzene sulphonamide is insoluble in alkali.
Reason (R): Sulphonyl group attached to nitrogen atom is strong electron withdrawing group.
a)
Both A and R are true but R is NOT the correct explanation of A
b)
Both A and R are true and R is the correct explanation of A
c)
A is true but R is false
d)
A is false and R is True
 | Lead Academy answered |
Assertion (A) states that N,N-diethylbenzene sulphonamide is insoluble in alkali.
N,N-diethylbenzene sulphonamide has a benzene ring attached to a sulphonamide (SO₂NH) with two ethyl groups on nitrogen. Solubility in alkali typically requires acidic hydrogens to react with OH⁻ ions. However, N,N-diethylbenzene sulphonamide lacks these acidic hydrogens, making it insoluble in alkali.
Hence, Assertion is correct.
Reason (R) states that the sulphonyl group is a strong electron-withdrawing group.
Reason (R) states that the sulphonyl group is a strong electron-withdrawing group.
The SO₂ group does withdraw electrons, reducing electron density on nitrogen. However, this electron withdrawal doesn’t directly cause the insolubility; the lack of acidic hydrogen is the main reason.
Hence, Reason is correct but does not explain the assertion.
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.Assertion (A): Aromatic 1° amines can be prepared by Gabriel Phthalimide synthesis.Reason (R): Aryl halides do not undergo nucleophilic substitution with anion formed by phthalimide.- a)Both A and R are true and R is the correct explanation of A
- b)Both A and R are true but R is NOT the correct explanation of A
- c)A is true but R is false
- d)Both A and R are False
Correct answer is option 'D'. Can you explain this answer?
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): Aromatic 1° amines can be prepared by Gabriel Phthalimide synthesis.
Reason (R): Aryl halides do not undergo nucleophilic substitution with anion formed by phthalimide.
a)
Both A and R are true and R is the correct explanation of A
b)
Both A and R are true but R is NOT the correct explanation of A
c)
A is true but R is false
d)
Both A and R are False
 | Lead Academy answered |
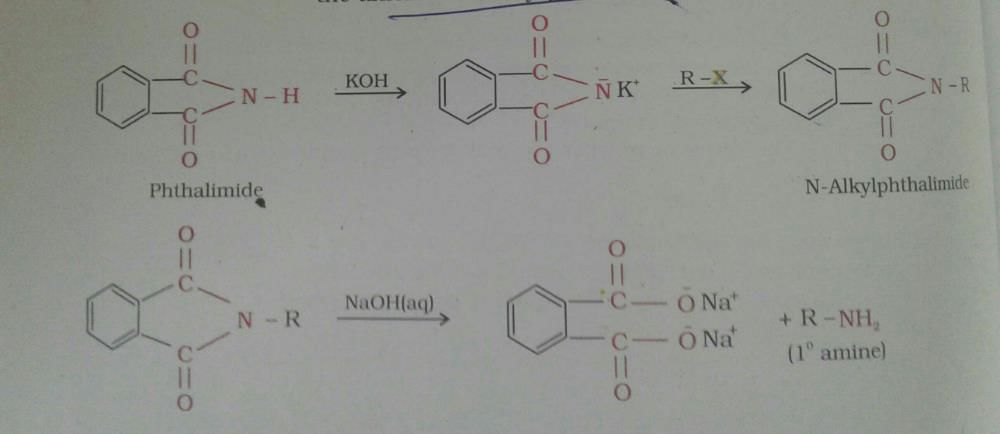
Aromatic primary amines cannot be prepared by Gabriel phthalimide synthesis as it is used for the preparation of aliphatic primary amines and not aromatic primary amines.
When phthalimide is treated with aqueous or ethanolic potassium hydroxide, it forms its potassium salt. This salt, when heated with an alkyl halide and then subjected to alkaline hydrolysis, yields the corresponding primary amine.
When phthalimide is treated with aqueous or ethanolic potassium hydroxide, it forms its potassium salt. This salt, when heated with an alkyl halide and then subjected to alkaline hydrolysis, yields the corresponding primary amine.
Aromatic primary amines cannot be prepared by this method because aryl halides do not undergo nucleophilic substitution with the anion formed by phthalimide.
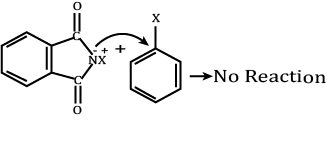
So, the assertion is wrong.
Reason:
Aryl halides do not undergo nucleophilic substitution with anion formed by phthalimide.
So, the reason is wrong.
Hence, both assertion and reason are wrong.
Aniline can be prepared by:- a)Reduction of nitrobenzene
- b)Nitration of nitrobenzene
- c)Oxidation of nitrobenzene
- d)Nitration of nitrobenzene followed by reduction
Correct answer is option 'A'. Can you explain this answer?
Aniline can be prepared by:
a)
Reduction of nitrobenzene
b)
Nitration of nitrobenzene
c)
Oxidation of nitrobenzene
d)
Nitration of nitrobenzene followed by reduction
| | Sanaya Kumar answered |
Aniline is prepared commercially by the catalytic hydrogenation of nitrobenzene or by the action of ammonia on chlorobenzene. The reduction of nitrobenzene can also be carried out with iron borings in aqueous acid. A primary aromatic amine, aniline is a weak base and forms salts with mineral acids.
Aniline does not undergo Friedel – Crafts reaction- a)Anilium ion deactivates any further reaction.
- b)aluminium chloride, the catalyst reacts with NH2 group
- c)nitrogen of aniline acquires positive charge
- d)all of these
Correct answer is option 'D'. Can you explain this answer?
Aniline does not undergo Friedel – Crafts reaction
a)
Anilium ion deactivates any further reaction.
b)
aluminium chloride, the catalyst reacts with NH2 group
c)
nitrogen of aniline acquires positive charge
d)
all of these
 | Gauri Sharma answered |
NH2 has lp of electron which reacts with AlCl3 or other catalyst that we add to form anilium ion.
Chapter doubts & questions for Amines - 3 Months Class 12 Board Preparation (Medical) 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Amines - 3 Months Class 12 Board Preparation (Medical) in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
3 Months Class 12 Board Preparation (Medical)159 videos|895 docs|246 tests |
Top Courses NEET
Related NEET Content
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup

 A
A  B
B