All Exams > JEE > 2 Year Preparation Course for JEE > All Questions
All questions of Coordination Compounds for JEE Exam
If zeise’s salt has the formula [Pt(C2H4)CI3]-. In this, platinum primary and secondary valency are- a)+ 1 and 3
- b)+ 1 and 4
- c)+ 3 and 4
- d)+ 4 and 6
Correct answer is option 'B'. Can you explain this answer?
If zeise’s salt has the formula [Pt(C2H4)CI3]-. In this, platinum primary and secondary valency are
a)
+ 1 and 3
b)
+ 1 and 4
c)
+ 3 and 4
d)
+ 4 and 6
| | Krishna Iyer answered |
Let the oxidation state of pt be x.
Oxidation state of cl is -1.
So x + 0 – (1*3) must be equal to -1 since the charge of the whole compound is -1.
x + 0 – (1*3) = -1
x -3 = -1
x = -1 + 3
x = +2
So the oxidation state of platinum is +2.
Secondary is due to legend there are mono deadened legend
then 1×4= 4
Oxidation state of cl is -1.
So x + 0 – (1*3) must be equal to -1 since the charge of the whole compound is -1.
x + 0 – (1*3) = -1
x -3 = -1
x = -1 + 3
x = +2
So the oxidation state of platinum is +2.
Secondary is due to legend there are mono deadened legend
then 1×4= 4
Type of bonding in K4 [Fe(CN)6] is/a- a)ionic
- b)covalent
- c)metallic
- d)coordinate covalent
Correct answer is option 'A,B,D'. Can you explain this answer?
Type of bonding in K4 [Fe(CN)6] is/a
a)
ionic
b)
covalent
c)
metallic
d)
coordinate covalent
| | Shubham Jain answered |
The complex K4[Fe(CN)6] whose formula can be written like that of double salt. Fe(CN)2 . 4KCN, dissociates to give K+ and [Fe(CN)6]4- ions in the aqueous solution.
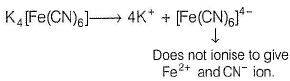
In the complex PtCl4 . 5NH3 if coordination number of platinum is 6, number of chloride ions precipitated by adding AgNO3 are
Correct answer is '3'. Can you explain this answer?
In the complex PtCl4 . 5NH3 if coordination number of platinum is 6, number of chloride ions precipitated by adding AgNO3 are
| | Poulomi Desai answered |
Coordination number of platinum is 6.
∴ Number of chloride ions precipitated by adding AgCI are 3.
∴ Number of chloride ions precipitated by adding AgCI are 3.
The magnetic moment of [Ru(H2O)6]2+ corresponds to the presence of ...... unpaired electrons.
Correct answer is '4'. Can you explain this answer?
The magnetic moment of [Ru(H2O)6]2+ corresponds to the presence of ...... unpaired electrons.
 | Anuj Iyer answered |
Ru2+ =[Kr] 4d6. This forms outer complex. Hence, unpaired electrons are 4.
The spin only magnetic moment value (in Bohr magneton units) of Cr(CO)6 is- a)0
- b)2.84
- c)4.90
- d)5.92
Correct answer is option 'A'. Can you explain this answer?
The spin only magnetic moment value (in Bohr magneton units) of Cr(CO)6 is
a)
0
b)
2.84
c)
4.90
d)
5.92
| | Rahul Bansal answered |
The electron configuration is [Ar]3d^5 4s^1.We have to accomodate the 6 Ligands and the fact that CO is a strong ligand.
This results in d^2sp^3 hybridization. Therefore, there are no unpaired electrons in Cr(CO)6. Hence n=0
And the spin only magnetic moment is also 0.
The effective atomic number of Fe in Fe(CO)5 is- a)36
- b)24
- c)34
- d)26
Correct answer is option 'A'. Can you explain this answer?
The effective atomic number of Fe in Fe(CO)5 is
a)
36
b)
24
c)
34
d)
26
 | Anupama Nair answered |
EAN= atomic no of Fe - oxidation state + no of e donated by ligand... Oxidation state of Fe is 0 since CO is neutral ligand... Two donor atoms hence no of e = 2×5=10.... EAN= 26-0+10=36
Tollen’s reagent contains- a)AgOH
- b)AgNO3
- c)[Ag(NO3)2]+
- d)[Ag(NH3)2]+
Correct answer is option 'B'. Can you explain this answer?
Tollen’s reagent contains
a)
AgOH
b)
AgNO3
c)
[Ag(NO3)2]+
d)
[Ag(NH3)2]+
 | Akash Shah answered |
Toilen’s reagent is ammoniacal silver nitrate solution. It is used to distinguish aldehydes and ketones, reducing and non-reducing sugars.
Which of the following statements is/are correct?- a)[Co(en)2NO2Cl] Br is cationic complex
- b)[Co(en)3]CI3 produces 3 ions in solution
- c)[Fe(CO)5] is neutral complex
- d)[Cu(NH3)4]SO4 is deep blue colour
Correct answer is option 'A,C,D'. Can you explain this answer?
Which of the following statements is/are correct?
a)
[Co(en)2NO2Cl] Br is cationic complex
b)
[Co(en)3]CI3 produces 3 ions in solution
c)
[Fe(CO)5] is neutral complex
d)
[Cu(NH3)4]SO4 is deep blue colour
| | Nikita Singh answered |
[Co(en)3]CI3 produces 4 ions in solution as follows :
[Co(en)3] CI3→ [Co(en)3]3+ + 3CI-
[Co(en)3] CI3→ [Co(en)3]3+ + 3CI-
- is correct as charge on the complex ion will be +1
- is incorrect as the complex will form 4 ions in solution
- is correct as there is no charge on the complex
- is also correct as cu+2 has blue color in solution
Hence A, C and D are correct.
Complex compounds are addition compounds formed by the stoichiometric combination of two or more simple salts but do not decompose into constituent ions completely. The first such complex prepared by Tassaert is hexamine cobalt (III) chloride. Later many such compounds were prepared and their properties were studied. The chloramines complexes of cobalt (III) chromium (III) not only exhibit a spectrum of colours but also differ in the reactivity of their chlorides. Moreover, greater the number of ions produced by a complex in solution, greater is the electrical conductivity. This type of information was obtained for several series of complexes.Q. The number of ions per mole of the complex CoCI3 . 5NH3 in aqueous solution will be- a)3
- b)9
- c)2
- d)4
Correct answer is option 'A'. Can you explain this answer?
Complex compounds are addition compounds formed by the stoichiometric combination of two or more simple salts but do not decompose into constituent ions completely. The first such complex prepared by Tassaert is hexamine cobalt (III) chloride. Later many such compounds were prepared and their properties were studied. The chloramines complexes of cobalt (III) chromium (III) not only exhibit a spectrum of colours but also differ in the reactivity of their chlorides. Moreover, greater the number of ions produced by a complex in solution, greater is the electrical conductivity. This type of information was obtained for several series of complexes.
Q.
The number of ions per mole of the complex CoCI3 . 5NH3 in aqueous solution will be
a)
3
b)
9
c)
2
d)
4
| | Suresh Iyer answered |
[Co(NH3)5CI]CI2→[Co(NH3)5CI]2++ 2Cl-
Hence, 3 ions are present for one mole of the complex in the solution.
Hence, 3 ions are present for one mole of the complex in the solution.
A freshly prepared aqueous solution of Pd(NH3)2CI2 does not conduct electricity, it suggests that- a)the structure of the compound involves covalent bonding only
- b)the chlorine atoms must be in coordination sphere
- c)the van't Hoff factor of the compound would be unity
- d)on adding excess aqueous AgNO3 to 0.1 L of 0.1 M solution of the compound, 0.02 mole of AgCI would be obtained
Correct answer is option 'B,C'. Can you explain this answer?
A freshly prepared aqueous solution of Pd(NH3)2CI2 does not conduct electricity, it suggests that
a)
the structure of the compound involves covalent bonding only
b)
the chlorine atoms must be in coordination sphere
c)
the van't Hoff factor of the compound would be unity
d)
on adding excess aqueous AgNO3 to 0.1 L of 0.1 M solution of the compound, 0.02 mole of AgCI would be obtained
 | Sankar Chakraborty answered |
In the aqueous solution of Pd(NH3)2CI2, the atoms of chlorine I are in coordination sphere and the van't Hoff factor of the ] compound are unite.
In the estimation of hardness of water, the reagent used is- a)hypo solution
- b)KMnO4
- c)EDTA solution
- d)K2Cr2O7
Correct answer is option 'C'. Can you explain this answer?
In the estimation of hardness of water, the reagent used is
a)
hypo solution
b)
KMnO4
c)
EDTA solution
d)
K2Cr2O7
| | Preeti Iyer answered |
The EDTA solution can then be used to determine the hardness of an unknown water sample. Since both EDTA and Ca2+ are colorless, it is necessary to use a special indicator to detect the end point of the titration.
When AgNO3 solution is added in excess to 1 M solution of CoCI3 . xNH3, one mole of AgCI is formed. The value of x is
Correct answer is '4'. Can you explain this answer?
When AgNO3 solution is added in excess to 1 M solution of CoCI3 . xNH3, one mole of AgCI is formed. The value of x is
| | Nikita Singh answered |
AgNO3 solution is added in excess of 1 M solution of CoCI3 . xNH3.
CoCl3.xNH3+AgNO3→AgCl (1mole)
CoCl3.xNH3+AgNO3→AgCl (1mole)
This precipitation of 1 mol of AgCl by this reaction shows that there is only one Cl outside the coordination sphere, which is not as a ligand ( as ligands are not ionisable).
Hence, the compound must be as follows:(showing the coordination sphere) [Co(NH3)4Cl2]Cl, as this is the octahedral complex, where it is clear that there are only 2 Cl as ligand and other ligands are NH3.
So, 6−2 = 4 NH3 ligands.
A cobaltamine has the formula CoCI3 . xNH3. This when reacted with AgNO3 solution, one third of the chloride is precipitated. It can have the structure- a)[Co(NH3)6]CI3
- b)[Co(NH3)5]CI2
- c)[Co(NH3)4CI2]CI
- d)[Co(NH3)5H2O]CI3
Correct answer is option 'C'. Can you explain this answer?
A cobaltamine has the formula CoCI3 . xNH3. This when reacted with AgNO3 solution, one third of the chloride is precipitated. It can have the structure
a)
[Co(NH3)6]CI3
b)
[Co(NH3)5]CI2
c)
[Co(NH3)4CI2]CI
d)
[Co(NH3)5H2O]CI3
| | Om Desai answered |
Total chlorine molecules are 3 and one third means one chlorine molecule precipitates. So, one chlorine has to be outside the coordination sphere and that is in option C.
One or More than One Options Correct TypeDirection (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.Q. An example of double salt is/a- a)hypo
- b)bleaching powder
- c)common alum
- d)carnallite
Correct answer is option 'C,D'. Can you explain this answer?
One or More than One Options Correct Type
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
An example of double salt is/a
a)
hypo
b)
bleaching powder
c)
common alum
d)
carnallite
 | Aravind Mehra answered |
Common alum is K2SO4.AI2(SO4)3.24H2O and carnallite is KCl.MgCl2.6H2O.
An example for bidentate and negatively charged ligand is- a)acetylacetonate (acac)
- b)propylene diamine (pn)
- c)ethylene diamine (en)
- d)bipyridyl (bip
Correct answer is option 'A'. Can you explain this answer?
An example for bidentate and negatively charged ligand is
a)
acetylacetonate (acac)
b)
propylene diamine (pn)
c)
ethylene diamine (en)
d)
bipyridyl (bip
 | Ishani Yadav answered |
CH3COCH2COCH3
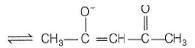
It can ligate through two oxygen atoms.
Only One Option Correct TypeDirection (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.Q. Primary and secondary valency of Pt in [Pt(en)2CI2] are- a)+ 4 and - 4
- b)+ 4 and 6
- c)+ 6 and 4
- d)+ 2 and 6
Correct answer is option 'D'. Can you explain this answer?
Only One Option Correct Type
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
Primary and secondary valency of Pt in [Pt(en)2CI2] are
a)
+ 4 and - 4
b)
+ 4 and 6
c)
+ 6 and 4
d)
+ 2 and 6
| | Rahul Bansal answered |
The primary valence is its oxidation number, which is +2 in this case (en is uncharged, and the Cl ligands carry a -1 charge each, so Pt must be +2 to balance).
Secondary valence is coordination number. The en ligand is bidentate, and Cl is monodentate, so you have 2(2) + 2(1) = 6, so the Pt has a coordination number of 6.
Can you explain the answer of this question below:sp3-hybridisation is found in
- A:
[ZnCI4]2-
- B:
[Cu(NH3)4]2+
- C:
[CuCI4]2-
- D:
[Ni(CO)4]
The answer is A,C,D.
sp3-hybridisation is found in
[ZnCI4]2-
[Cu(NH3)4]2+
[CuCI4]2-
[Ni(CO)4]
| | Dipika Rane answered |
Explanation:
SP3 hybridization is a type of hybridization where one s orbital and three p orbitals of the same shell of an atom mix to form four sp3 hybrid orbitals. These hybrid orbitals are arranged in a tetrahedral shape around the central atom.
The given options are:
a) [ZnCl4]2-
b) [Cu(NH3)4]2
c) [CuCl4]2-
d) [Ni(CO)4]
a) [ZnCl4]2-: In this complex ion, the central zinc atom is sp3 hybridized. The zinc ion has an electronic configuration of [Ar]3d104s0. The hybridization of the zinc ion takes place by mixing one 4s orbital and three 4p orbitals to form four sp3 hybrid orbitals.
b) [Cu(NH3)4]2: In this complex ion, the central copper atom is dsp2 hybridized. The copper ion has an electronic configuration of [Ar]3d104s1. The hybridization of the copper ion takes place by mixing one 4s orbital, two 4p orbitals, and one 3d orbital to form five dsp2 hybrid orbitals.
c) [CuCl4]2-: In this complex ion, the central copper atom is sp3 hybridized. The copper ion has an electronic configuration of [Ar]3d104s1. The hybridization of the copper ion takes place by mixing one 4s orbital and three 4p orbitals to form four sp3 hybrid orbitals.
d) [Ni(CO)4]: In this complex ion, the central nickel atom is sp3 hybridized. The nickel ion has an electronic configuration of [Ar]3d84s2. The hybridization of the nickel ion takes place by mixing one 4s orbital and three 4p orbitals to form four sp3 hybrid orbitals.
Conclusion:
Thus, the correct options are A, C, and D, as all these complex ions have a central atom that is sp3 hybridized. The complex ion in option B has a central copper atom that is dsp2 hybridized.
SP3 hybridization is a type of hybridization where one s orbital and three p orbitals of the same shell of an atom mix to form four sp3 hybrid orbitals. These hybrid orbitals are arranged in a tetrahedral shape around the central atom.
The given options are:
a) [ZnCl4]2-
b) [Cu(NH3)4]2
c) [CuCl4]2-
d) [Ni(CO)4]
a) [ZnCl4]2-: In this complex ion, the central zinc atom is sp3 hybridized. The zinc ion has an electronic configuration of [Ar]3d104s0. The hybridization of the zinc ion takes place by mixing one 4s orbital and three 4p orbitals to form four sp3 hybrid orbitals.
b) [Cu(NH3)4]2: In this complex ion, the central copper atom is dsp2 hybridized. The copper ion has an electronic configuration of [Ar]3d104s1. The hybridization of the copper ion takes place by mixing one 4s orbital, two 4p orbitals, and one 3d orbital to form five dsp2 hybrid orbitals.
c) [CuCl4]2-: In this complex ion, the central copper atom is sp3 hybridized. The copper ion has an electronic configuration of [Ar]3d104s1. The hybridization of the copper ion takes place by mixing one 4s orbital and three 4p orbitals to form four sp3 hybrid orbitals.
d) [Ni(CO)4]: In this complex ion, the central nickel atom is sp3 hybridized. The nickel ion has an electronic configuration of [Ar]3d84s2. The hybridization of the nickel ion takes place by mixing one 4s orbital and three 4p orbitals to form four sp3 hybrid orbitals.
Conclusion:
Thus, the correct options are A, C, and D, as all these complex ions have a central atom that is sp3 hybridized. The complex ion in option B has a central copper atom that is dsp2 hybridized.
Which is the diamagnetic?- a)[CoF6]3-
- b)[Ni(CN)4]2-
- c)[NiCI3]2-
- d)[Fe(CN)6]3-
Correct answer is option 'B'. Can you explain this answer?
Which is the diamagnetic?
a)
[CoF6]3-
b)
[Ni(CN)4]2-
c)
[NiCI3]2-
d)
[Fe(CN)6]3-
| | Niti Mishra answered |
Explanation:
Diamagnetic substances are those which do not have any unpaired electrons and are not attracted by a magnetic field. On the other hand, paramagnetic substances have unpaired electrons and are attracted by a magnetic field.
Let's examine the given options to determine which one is diamagnetic.
[CoF6]3-
Cobalt has 27 electrons. In this complex, cobalt is in the +3 oxidation state. The six fluoride ions act as ligands and each donate one pair of electrons to the cobalt ion. The complex has a total of 33 electrons, and there are three unpaired electrons in the d-orbitals of the cobalt ion. Therefore, [CoF6]3- is a paramagnetic complex.
[Ni(CN)4]2-
Nickel has 28 electrons. In this complex, nickel is in the +2 oxidation state. The four cyanide ions act as ligands and each donate one pair of electrons to the nickel ion. The complex has a total of 34 electrons, and all the electrons are paired. Therefore, [Ni(CN)4]2- is a diamagnetic complex.
[NiCl4]2-
Similar to the above complex, nickel has 28 electrons and is in the +2 oxidation state. The four chloride ions act as ligands and each donate one pair of electrons to the nickel ion. The complex has a total of 32 electrons, and all the electrons are paired. Therefore, [NiCl4]2- is a diamagnetic complex.
[Fe(CN)6]3-
Iron has 26 electrons. In this complex, iron is in the +3 oxidation state. The six cyanide ions act as ligands and each donate one pair of electrons to the iron ion. The complex has a total of 32 electrons, and there are five unpaired electrons in the d-orbitals of the iron ion. Therefore, [Fe(CN)6]3- is a paramagnetic complex.
Therefore, the diamagnetic complex among the given options is [Ni(CN)4]2-.
Diamagnetic substances are those which do not have any unpaired electrons and are not attracted by a magnetic field. On the other hand, paramagnetic substances have unpaired electrons and are attracted by a magnetic field.
Let's examine the given options to determine which one is diamagnetic.
[CoF6]3-
Cobalt has 27 electrons. In this complex, cobalt is in the +3 oxidation state. The six fluoride ions act as ligands and each donate one pair of electrons to the cobalt ion. The complex has a total of 33 electrons, and there are three unpaired electrons in the d-orbitals of the cobalt ion. Therefore, [CoF6]3- is a paramagnetic complex.
[Ni(CN)4]2-
Nickel has 28 electrons. In this complex, nickel is in the +2 oxidation state. The four cyanide ions act as ligands and each donate one pair of electrons to the nickel ion. The complex has a total of 34 electrons, and all the electrons are paired. Therefore, [Ni(CN)4]2- is a diamagnetic complex.
[NiCl4]2-
Similar to the above complex, nickel has 28 electrons and is in the +2 oxidation state. The four chloride ions act as ligands and each donate one pair of electrons to the nickel ion. The complex has a total of 32 electrons, and all the electrons are paired. Therefore, [NiCl4]2- is a diamagnetic complex.
[Fe(CN)6]3-
Iron has 26 electrons. In this complex, iron is in the +3 oxidation state. The six cyanide ions act as ligands and each donate one pair of electrons to the iron ion. The complex has a total of 32 electrons, and there are five unpaired electrons in the d-orbitals of the iron ion. Therefore, [Fe(CN)6]3- is a paramagnetic complex.
Therefore, the diamagnetic complex among the given options is [Ni(CN)4]2-.
Statement TypeDirection (Q. Nos. 24 and 25) This section is based on Statement I and Statement II. Select the correct answer from the codes given below.Q. Statement I : Oxidation state of Fe in Fe(CO)5 is zero.
Statement II : EAN of Fe in this complex is 36.- a)Both Statement I and Statement II are correct and Statement II is the correct explanation of Statement I
- b)Both Statement I and Statement II are correct but Statement II is not the correct explanation of Statement I
- c)Statement I is correct but Statement II is incorrect
- d)Statement II is correct but Statement I is incorrect
Correct answer is option 'B'. Can you explain this answer?
Statement Type
Direction (Q. Nos. 24 and 25) This section is based on Statement I and Statement II. Select the correct answer from the codes given below.
Q.
Statement I : Oxidation state of Fe in Fe(CO)5 is zero.
Statement II : EAN of Fe in this complex is 36.
Statement II : EAN of Fe in this complex is 36.
a)
Both Statement I and Statement II are correct and Statement II is the correct explanation of Statement I
b)
Both Statement I and Statement II are correct but Statement II is not the correct explanation of Statement I
c)
Statement I is correct but Statement II is incorrect
d)
Statement II is correct but Statement I is incorrect
 | Shail Chakraborty answered |
Statement I is true because CO is a neutral ligand, Statement II is true because
EAN of Fe
= Atomic number of Fe + Electrons gained by coordination
= 26 + 2 x 5 = 36
But Statement II is not the correct explanation. Correct explanation is that oxidation state of Fe in Fe(CO)5 is zero because CO is a neutral ligand.
EAN of Fe
= Atomic number of Fe + Electrons gained by coordination
= 26 + 2 x 5 = 36
But Statement II is not the correct explanation. Correct explanation is that oxidation state of Fe in Fe(CO)5 is zero because CO is a neutral ligand.
The volume (in mL) of 0.1 M AgNO3 required for complex precipitation of chloride ions present in 30 mL of 0.01 M solution of [Cr(H2O)5Cl]Cl2, as silver chloride is close to- a)3
- b)4
- c)5
- d)6
Correct answer is option 'D'. Can you explain this answer?
The volume (in mL) of 0.1 M AgNO3 required for complex precipitation of chloride ions present in 30 mL of 0.01 M solution of [Cr(H2O)5Cl]Cl2, as silver chloride is close to
a)
3
b)
4
c)
5
d)
6
 | Sanchita Reddy answered |
The chemical reaction between them is...[Cr(H2O)5Cl]Cl2 + {2}AgNO3 ——> {2}AgCl(↓) + [Cr(H2O)5Cl]^2+ + {2}NO3-moles of coordinate compound given = molarity x volume(in litre)mole = 0.01 x (30/1000) = (3/10000)for one mole coordinate compound.. we need 2 mole AgNO3....i.e.,1mole complex ===> 2 mole AgNO3(3/10000) mole ==> (2x3/10000) mole AgNO3molarity = moles/volume (in litre)volume(in litre) = mole / molarityV= (6/10000)/0.1 = (0.6) Litrehence volume required = 0.6litre = 6ml
One Integer Value Correct TypeDirection (Q. Nos. 19-23) This section contains 5 questions. When worked out will result in an integer from 0 to 9 (both inclusive).Q. Formula of Mohr’s salt is FeSO4 . (NH4)2SO4 . xH2O. Here x value is
Correct answer is '6'. Can you explain this answer?
One Integer Value Correct Type
Direction (Q. Nos. 19-23) This section contains 5 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
Q.
Formula of Mohr’s salt is FeSO4 . (NH4)2SO4 . xH2O. Here x value is
| | Keerthana Iyer answered |
Calculation of x value in Mohr's Salt Formula
Heading: Introduction to Mohr's Salt Formula
Mohr's salt is a chemical compound with the formula FeSO4.(NH4)2SO4.xH2O. It is a hydrated double salt of ferrous sulfate and ammonium sulfate.
Heading: Calculation of x value
The x value in the Mohr's salt formula represents the number of water molecules present in the crystal structure of the compound. To calculate the value of x, we need to use the following formula:
Molecular weight of Mohr's salt = Molecular weight of FeSO4 + 2 × Molecular weight of (NH4)2SO4 + x × Molecular weight of H2O
The molecular weight of FeSO4 is 151.91 g/mol.
The molecular weight of (NH4)2SO4 is 132.14 g/mol.
The molecular weight of H2O is 18.02 g/mol.
Substituting these values in the formula, we get:
Molecular weight of Mohr's salt = 151.91 + 2 × 132.14 + x × 18.02
Molecular weight of Mohr's salt = 416.19 + 18.02x
To find the value of x, we need to know the molecular weight of Mohr's salt. It is 392.14 g/mol.
Substituting this value in the above equation, we get:
392.14 = 416.19 + 18.02x
18.02x = -24.05
x = -1.335
Heading: Interpretation of x value
The calculated value of x is -1.335, which is not possible as the number of water molecules cannot be negative. Therefore, we need to take the absolute value of x, which gives us 1.335. However, this is still not the correct answer as the x value should be an integer.
To convert 1.335 into an integer, we need to round it off to the nearest integer. As 1.335 is closer to 1 than 2, we round it off to 1.
Heading: Conclusion
Therefore, the value of x in the Mohr's salt formula is 1, which represents the presence of one water molecule in the crystal structure of the compound.
Heading: Introduction to Mohr's Salt Formula
Mohr's salt is a chemical compound with the formula FeSO4.(NH4)2SO4.xH2O. It is a hydrated double salt of ferrous sulfate and ammonium sulfate.
Heading: Calculation of x value
The x value in the Mohr's salt formula represents the number of water molecules present in the crystal structure of the compound. To calculate the value of x, we need to use the following formula:
Molecular weight of Mohr's salt = Molecular weight of FeSO4 + 2 × Molecular weight of (NH4)2SO4 + x × Molecular weight of H2O
The molecular weight of FeSO4 is 151.91 g/mol.
The molecular weight of (NH4)2SO4 is 132.14 g/mol.
The molecular weight of H2O is 18.02 g/mol.
Substituting these values in the formula, we get:
Molecular weight of Mohr's salt = 151.91 + 2 × 132.14 + x × 18.02
Molecular weight of Mohr's salt = 416.19 + 18.02x
To find the value of x, we need to know the molecular weight of Mohr's salt. It is 392.14 g/mol.
Substituting this value in the above equation, we get:
392.14 = 416.19 + 18.02x
18.02x = -24.05
x = -1.335
Heading: Interpretation of x value
The calculated value of x is -1.335, which is not possible as the number of water molecules cannot be negative. Therefore, we need to take the absolute value of x, which gives us 1.335. However, this is still not the correct answer as the x value should be an integer.
To convert 1.335 into an integer, we need to round it off to the nearest integer. As 1.335 is closer to 1 than 2, we round it off to 1.
Heading: Conclusion
Therefore, the value of x in the Mohr's salt formula is 1, which represents the presence of one water molecule in the crystal structure of the compound.
In which of the following porphyrin acts as ligand?- a)Haemoglobin
- b)Chlorophyll
- c)Vitamin B-12
- d)Insulin
Correct answer is option 'A,B'. Can you explain this answer?
In which of the following porphyrin acts as ligand?
a)
Haemoglobin
b)
Chlorophyll
c)
Vitamin B-12
d)
Insulin
| | Rohan Singh answered |
The Heme Porphyrin. Although the hemoglobin and myoglobin molecules are very large, complex proteins, the active site is actually a non-protein group called heme. The heme consists of a flat organic ring surrounding an iron atom.
Chlorophylls are numerous in types, but all are defined by the presence of a fifth ring beyond the four pyrrole-like rings. Most chlorophylls are classified as chlorins, which are reduced relatives to porphyrins (found in hemoglobin). They share a common biosynthetic pathway as porphyrins, including the precursor uroporphyrinogen III. Unlike hemes, which feature iron at the center of the tetrapyrrole ring, chlorophylls bind magnesium. For the structures depicted in this article, some of the ligands attached to the Mg2+ center are omitted for clarity. The chlorin ring can have various side chains, usually including a long phytol chain. The most widely distributed form in terrestrial plants is chlorophyll a.
The pair of compounds having the same hybridisation for the central atom is- a)[Cu(NH3)4]2+ and [Ni(NH3)4]2+
- b)[NiCI4]2- and [PtCI4]2-
- c)(Cu(NH3)4]2+ and [Zn(NH3)4]2+
- d)[Co(NH3)6]3+ and [Co(H2O)6]3+
Correct answer is option 'A'. Can you explain this answer?
The pair of compounds having the same hybridisation for the central atom is
a)
[Cu(NH3)4]2+ and [Ni(NH3)4]2+
b)
[NiCI4]2- and [PtCI4]2-
c)
(Cu(NH3)4]2+ and [Zn(NH3)4]2+
d)
[Co(NH3)6]3+ and [Co(H2O)6]3+
 | Vaishnavi Dasgupta answered |
[Cu(NH3)4]2+ and [Ni(NH3)4]2+ the central atoms have dsp2 hybridisation.
Which are π-bonded organometallic compounds?- a)[Cr(η6 — (C6H6)]
- b)[Fe(CO)5]
- c)K[PtCI3η2 - C2H4]
- d)[Fe(η5 - C5H5)2]
Correct answer is option 'A,C,D'. Can you explain this answer?
Which are π-bonded organometallic compounds?
a)
[Cr(η6 — (C6H6)]
b)
[Fe(CO)5]
c)
K[PtCI3η2 - C2H4]
d)
[Fe(η5 - C5H5)2]
 | Kunal Pillai answered |
η6 -arene is 6-electron ligand, η2 — C2H4 is 2-electron ligand η6-cyclopentadienyl is 5-electron donor. All these 3 ligands creates π-bonded complex. CO as a ligand is sigma donor and pi-acceptor ligand.
The tetrachloro complexes of Ni(ll) and Pd(ll) respectively are (atomic number of Ni and Pd are 28 and 46 respectively).- a)diamagnetic and diamagnetic
- b)diamagnetic and paramagnetic
- c)paramagnetic and diamagnetic
- d)paramagnetic and paramagnetic
Correct answer is option 'C'. Can you explain this answer?
The tetrachloro complexes of Ni(ll) and Pd(ll) respectively are (atomic number of Ni and Pd are 28 and 46 respectively).
a)
diamagnetic and diamagnetic
b)
diamagnetic and paramagnetic
c)
paramagnetic and diamagnetic
d)
paramagnetic and paramagnetic
| | Madhurima Mishra answered |
The tetrachloro complexes of Ni(ll) and Pd(ll) have Ni and Pd ions in the +2 oxidation state, which means they have two fewer electrons than their neutral atoms. To determine the magnetic properties of these complexes, we need to consider the electronic configuration and the presence of unpaired electrons.
Electronic configuration of Ni(ll): [Ar] 3d8 4s2
Electronic configuration of Pd(ll): [Kr] 4d8 5s2
Diamagnetic vs. Paramagnetic:
- Diamagnetic substances have all their electrons paired up and do not have any net magnetic moment.
- Paramagnetic substances have unpaired electrons, which result in a net magnetic moment.
Analyzing the electronic configurations:
- Both Ni(ll) and Pd(ll) have partially filled d-orbitals (d8 configuration), which means they can exhibit paramagnetic behavior due to the presence of unpaired electrons.
- However, in the case of tetrachloro complexes, the chloride ions (Cl-) act as strong ligands, which cause a significant amount of pairing of electrons in the d-orbitals. This effect is known as the "ligand field stabilization energy" (LFSE).
- The LFSE is greater for Ni(ll) compared to Pd(ll) because Ni has a smaller atomic size and higher charge density, leading to stronger interaction between the metal ion and the ligands.
- As a result, the tetrachloro complex of Ni(ll) has a greater tendency to pair up its electrons and becomes diamagnetic.
- On the other hand, the tetrachloro complex of Pd(ll) has a weaker LFSE and retains some unpaired electrons, making it paramagnetic.
Summary:
- The tetrachloro complex of Ni(ll) is diamagnetic because of the strong ligand field stabilization energy, which causes complete pairing of electrons.
- The tetrachloro complex of Pd(ll) is paramagnetic because it has a weaker ligand field stabilization energy and retains some unpaired electrons.
Electronic configuration of Ni(ll): [Ar] 3d8 4s2
Electronic configuration of Pd(ll): [Kr] 4d8 5s2
Diamagnetic vs. Paramagnetic:
- Diamagnetic substances have all their electrons paired up and do not have any net magnetic moment.
- Paramagnetic substances have unpaired electrons, which result in a net magnetic moment.
Analyzing the electronic configurations:
- Both Ni(ll) and Pd(ll) have partially filled d-orbitals (d8 configuration), which means they can exhibit paramagnetic behavior due to the presence of unpaired electrons.
- However, in the case of tetrachloro complexes, the chloride ions (Cl-) act as strong ligands, which cause a significant amount of pairing of electrons in the d-orbitals. This effect is known as the "ligand field stabilization energy" (LFSE).
- The LFSE is greater for Ni(ll) compared to Pd(ll) because Ni has a smaller atomic size and higher charge density, leading to stronger interaction between the metal ion and the ligands.
- As a result, the tetrachloro complex of Ni(ll) has a greater tendency to pair up its electrons and becomes diamagnetic.
- On the other hand, the tetrachloro complex of Pd(ll) has a weaker LFSE and retains some unpaired electrons, making it paramagnetic.
Summary:
- The tetrachloro complex of Ni(ll) is diamagnetic because of the strong ligand field stabilization energy, which causes complete pairing of electrons.
- The tetrachloro complex of Pd(ll) is paramagnetic because it has a weaker ligand field stabilization energy and retains some unpaired electrons.
Only One Option Correct TypeDirection (Q. Nos. 1- 10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.Q. Which one of the following is wrongly matched?- a)[Ni(CO)4] = neutral complex
- b)[Cu(NH3)4]2+= square planar
- c)[Co(en)3]3+ = follow EAN rule
- d)[Fe(CN)6]3+ = sp3d2
Correct answer is option 'D'. Can you explain this answer?
Only One Option Correct Type
Direction (Q. Nos. 1- 10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
Which one of the following is wrongly matched?
a)
[Ni(CO)4] = neutral complex
b)
[Cu(NH3)4]2+= square planar
c)
[Co(en)3]3+ = follow EAN rule
d)
[Fe(CN)6]3+ = sp3d2
 | Anuj Iyer answered |
Due to strong ligand, this is inner orbital complex and the hybridisation of Fe is d2sp3.
Comprehension TypeDirection (Q. Nos. 16 and 17) This section contains a paragraph, describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).PassageComplex compounds are addition compounds formed by the stoichiometric combination of two or more simple salts but do not decompose into constituent ions completely. The first such complex prepared by Tassaert is hexamine cobalt (III) chloride. Later many such compounds were prepared and their properties were studied. The chloramines complexes of cobalt (III) chromium (III) not only exhibit a spectrum of colours but also differ in the reactivity of their chlorides. Moreover, greater the number of ions produced by a complex in solution, greater is the electrical conductivity. This type of information was obtained for several series of complexes.Q. Coordination number of Co in CoCl3 . 5H2O is six. The volume of 0.1 N AgNO3 needed to precipitate the chlorine in 200 mL of 0.01 M solution of complex is- a)140 mL
- b)40 mL
- c)80 mL
- d)20 mL
Correct answer is option 'B'. Can you explain this answer?
Comprehension Type
Direction (Q. Nos. 16 and 17) This section contains a paragraph, describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).
Passage
Complex compounds are addition compounds formed by the stoichiometric combination of two or more simple salts but do not decompose into constituent ions completely. The first such complex prepared by Tassaert is hexamine cobalt (III) chloride. Later many such compounds were prepared and their properties were studied. The chloramines complexes of cobalt (III) chromium (III) not only exhibit a spectrum of colours but also differ in the reactivity of their chlorides. Moreover, greater the number of ions produced by a complex in solution, greater is the electrical conductivity. This type of information was obtained for several series of complexes.
Q.
Coordination number of Co in CoCl3 . 5H2O is six. The volume of 0.1 N AgNO3 needed to precipitate the chlorine in 200 mL of 0.01 M solution of complex is
a)
140 mL
b)
40 mL
c)
80 mL
d)
20 mL
 | Gauri Khanna answered |
Since, the oxidation number of Co is + 3 hence, the complex compound would be [CoCI . 5H2O]CI2.
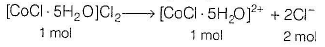
Hence, 1 mole of [CoCI . 5H2O]CI2 gives 2 moles of Cl- ion.
Given, moles of [CoCI . 5H2O]CI2 = 200 mL x 0.01 M = 2 millimol
2 millimol of [CoCI . 5H2O]CI2 will give 4 millimol of Cl-.
Now, to neutralise 4 millimol of Cl-, 4 millimol of AgNO3 is required.
Let the volume o! AgNO3 = VmL
Millimoles of AgNO3 = 0.1 x V
0.1 x V = 4 millimol
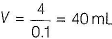
Hence, 1 mole of [CoCI . 5H2O]CI2 gives 2 moles of Cl- ion.
Given, moles of [CoCI . 5H2O]CI2 = 200 mL x 0.01 M = 2 millimol
2 millimol of [CoCI . 5H2O]CI2 will give 4 millimol of Cl-.
Now, to neutralise 4 millimol of Cl-, 4 millimol of AgNO3 is required.
Let the volume o! AgNO3 = VmL
Millimoles of AgNO3 = 0.1 x V
0.1 x V = 4 millimol
Oxidation state of cobalt in Co(CO)6 is
Correct answer is '0'. Can you explain this answer?
Oxidation state of cobalt in Co(CO)6 is
 | Mehul Choudhary answered |
ln Co(CO)6
Let the oxidation state of Co is x.
x + 6 x 0 = 0
[Carbonyl (CO) is a neutral ligand hence, zero charg]
x = 0
Hence, Co has zero oxidation state.
Let the oxidation state of Co is x.
x + 6 x 0 = 0
[Carbonyl (CO) is a neutral ligand hence, zero charg]
x = 0
Hence, Co has zero oxidation state.
In Fe2(CO)9, the number of CO present between iron atoms ......
Correct answer is '3'. Can you explain this answer?
In Fe2(CO)9, the number of CO present between iron atoms ......
 | Rahul Desai answered |
Fe2(CO)9 has three bridging CO groups.
Statement TypeDirection (Q. No. 25) This section is based on Statement I and Statement II. Select the correct answer from the codes given below.Q. Statement I : Both [Ni(CN)4]2- and [NiCI4]2- have same shape and same magnetic behaviour.Statement II : Both are square planar and diamagnetic- a)Both Statement I and Statement II are correct and Statement II is the correct explanation of Statement I
- b)Both Statement I and Statement II are correct but Statement II is not the correct explanation of Statement I
- c)Statement I is correct but Statement II is incorrect
- d)Statement II is correct but Statement I is incorrect
Correct answer is option 'B'. Can you explain this answer?
Statement Type
Direction (Q. No. 25) This section is based on Statement I and Statement II. Select the correct answer from the codes given below.
Q.
Statement I : Both [Ni(CN)4]2- and [NiCI4]2- have same shape and same magnetic behaviour.
Statement II : Both are square planar and diamagnetic
a)
Both Statement I and Statement II are correct and Statement II is the correct explanation of Statement I
b)
Both Statement I and Statement II are correct but Statement II is not the correct explanation of Statement I
c)
Statement I is correct but Statement II is incorrect
d)
Statement II is correct but Statement I is incorrect
| | Shubham Jain answered |
Explanation:
Statement I: Both [Ni(CN)4]2- and [NiCl4]2- have the same shape and same magnetic behaviour.
Statement II: Both are square planar and diamagnetic.
The given statements are related to the coordination compounds of nickel. Let us understand the concepts related to the given statements.
Coordination Compounds:
A coordination compound is a compound in which a central metal ion is surrounded by a group of ligands. A ligand is an atom, ion, or molecule that donates a pair of electrons to the metal ion. The coordination number of a central metal ion is the number of ligands that are attached to it.
Square Planar Geometry:
A coordination compound having a coordination number of 4 with the ligands in the same plane is said to have a square planar geometry. The square planar geometry is exhibited by complexes of metals with d8 electron configuration.
Magnetic Behaviour of Coordination Compounds:
The magnetic behaviour of coordination compounds is dependent on the presence or absence of unpaired electrons in the metal ion. If there are unpaired electrons in the metal ion, the compound is said to be paramagnetic and if there are no unpaired electrons, the compound is said to be diamagnetic.
Now, let us analyze the given statements.
Statement I states that both [Ni(CN)4]2- and [NiCl4]2- have the same shape and same magnetic behaviour. This statement is correct.
Statement II states that both [Ni(CN)4]2- and [NiCl4]2- are square planar and diamagnetic. This statement is also correct.
Therefore, both Statement I and Statement II are correct. However, Statement II does not explain Statement I. Hence, option (b) is the correct answer.
Conclusion:
Both [Ni(CN)4]2- and [NiCl4]2- have the same shape and same magnetic behaviour. They are square planar and diamagnetic. However, Statement II does not explain Statement I.
Statement I: Both [Ni(CN)4]2- and [NiCl4]2- have the same shape and same magnetic behaviour.
Statement II: Both are square planar and diamagnetic.
The given statements are related to the coordination compounds of nickel. Let us understand the concepts related to the given statements.
Coordination Compounds:
A coordination compound is a compound in which a central metal ion is surrounded by a group of ligands. A ligand is an atom, ion, or molecule that donates a pair of electrons to the metal ion. The coordination number of a central metal ion is the number of ligands that are attached to it.
Square Planar Geometry:
A coordination compound having a coordination number of 4 with the ligands in the same plane is said to have a square planar geometry. The square planar geometry is exhibited by complexes of metals with d8 electron configuration.
Magnetic Behaviour of Coordination Compounds:
The magnetic behaviour of coordination compounds is dependent on the presence or absence of unpaired electrons in the metal ion. If there are unpaired electrons in the metal ion, the compound is said to be paramagnetic and if there are no unpaired electrons, the compound is said to be diamagnetic.
Now, let us analyze the given statements.
Statement I states that both [Ni(CN)4]2- and [NiCl4]2- have the same shape and same magnetic behaviour. This statement is correct.
Statement II states that both [Ni(CN)4]2- and [NiCl4]2- are square planar and diamagnetic. This statement is also correct.
Therefore, both Statement I and Statement II are correct. However, Statement II does not explain Statement I. Hence, option (b) is the correct answer.
Conclusion:
Both [Ni(CN)4]2- and [NiCl4]2- have the same shape and same magnetic behaviour. They are square planar and diamagnetic. However, Statement II does not explain Statement I.
Oxidation states of iron in the complexes [Fe(H2O)5NO]2+ are- a)+ 1
- b)- 3
- c)+ 2
- d)+ 4
Correct answer is option 'A'. Can you explain this answer?
Oxidation states of iron in the complexes [Fe(H2O)5NO]2+ are
a)
+ 1
b)
- 3
c)
+ 2
d)
+ 4
 | Anu Basu answered |
Correct Answer : a
Explanation : NO in brown ring complex has +1 oxidation state.
Let oxidation state of Fe be x.
Since the overall charge on the coordination sphere, [Fe(H2O)5NO+] is +2, the sum of oxidation states of all elements in it should be equal to +2.
Therefore, x+1=+2
or, x=+1
Hence, the oxidation state of iron in the brown ring complex is +1.
Only One Option Correct TypeDirection (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.Q. A filter paper moistened with ammoniacal sodium nitroprusside solution turns violet on contact with a drop of alkaline Na2S solution .The violet colour is due to the formation of- a)[Fe(SCN)5NO]-
- b)[Fe(CN)5NOS]
- c)[Fe(CN)5NOS]4-
- d)[Fe(SCN)5NO]2-
Correct answer is option 'B'. Can you explain this answer?
Only One Option Correct Type
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
A filter paper moistened with ammoniacal sodium nitroprusside solution turns violet on contact with a drop of alkaline Na2S solution .The violet colour is due to the formation of
a)
[Fe(SCN)5NO]-
b)
[Fe(CN)5NOS]
c)
[Fe(CN)5NOS]4-
d)
[Fe(SCN)5NO]2-
| | Shail Chawla answered |
Na2S + Na2[Fe(CN)5NO] →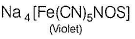
Which is the incorrect ?- a)[Co(NH3)4CI2]+ is heteroleptic complex
- b)In [Fe(C2O4)3]3+ and [Co(en)3]3+, the coordination number of both Fe and Co is 6
- c)Ferric alum is a double salt
- d)Mohr’s salt is a double salt and do not give test for Fe2+ ions
Correct answer is option 'D'. Can you explain this answer?
Which is the incorrect ?
a)
[Co(NH3)4CI2]+ is heteroleptic complex
b)
In [Fe(C2O4)3]3+ and [Co(en)3]3+, the coordination number of both Fe and Co is 6
c)
Ferric alum is a double salt
d)
Mohr’s salt is a double salt and do not give test for Fe2+ ions
 | Aarya Dasgupta answered |
Mohr's salt is a doub le salt and it will give test for Fe2+ ion. It dissociates into simple substances or ions completely when dissolved in water.
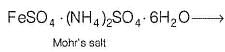
One or More than One Options Correct TypeDirection (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.Q. The formation of complex which involves d-orbitals of outer shell are called- a)low spin complex
- b)high spin complex
- c)inner orbital complex
- d)outer orbital complex
Correct answer is option 'B,D'. Can you explain this answer?
One or More than One Options Correct Type
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
The formation of complex which involves d-orbitals of outer shell are called
a)
low spin complex
b)
high spin complex
c)
inner orbital complex
d)
outer orbital complex
| | Nisha Patel answered |
Explanation:
The formation of complexes involves the interaction of metal ions with ligands. Ligands are molecules or ions that donate electrons to the metal ion to form coordinate bonds. In complexes, the metal ion typically has d-orbitals in its outer shell, which can participate in bonding with the ligands.
Low Spin Complex:
- In a low spin complex, the electrons occupy the lower energy d-orbitals before pairing up.
- This results in a smaller crystal field splitting energy and a larger energy gap between the lower and higher energy d-orbitals.
- The ligands in a low spin complex are usually strong field ligands, which cause a large crystal field splitting.
- The low spin complex is formed when the pairing energy (P) is greater than the crystal field splitting energy (Δ).
High Spin Complex:
- In a high spin complex, the electrons occupy the higher energy d-orbitals before pairing up.
- This results in a larger crystal field splitting energy and a smaller energy gap between the lower and higher energy d-orbitals.
- The ligands in a high spin complex are usually weak field ligands, which cause a small crystal field splitting.
- The high spin complex is formed when the crystal field splitting energy (Δ) is greater than the pairing energy (P).
Inner Orbital Complex:
- In an inner orbital complex, the ligands directly coordinate to the metal ion's d-orbitals.
- The d-orbitals involved in bonding are the inner d-orbitals, which are closer to the nucleus.
- The inner orbital complex is formed when the metal ion has vacant inner d-orbitals available for bonding.
Outer Orbital Complex:
- In an outer orbital complex, the ligands interact with the metal ion's outer d-orbitals.
- The outer d-orbitals are farther from the nucleus and have higher energy compared to the inner d-orbitals.
- The outer orbital complex is formed when the metal ion has vacant outer d-orbitals available for bonding.
Conclusion:
From the given options, the formation of complex involving d-orbitals of the outer shell can be referred to as both high spin complex and outer orbital complex. Therefore, the correct answers are option B (high spin complex) and option D (outer orbital complex).
The formation of complexes involves the interaction of metal ions with ligands. Ligands are molecules or ions that donate electrons to the metal ion to form coordinate bonds. In complexes, the metal ion typically has d-orbitals in its outer shell, which can participate in bonding with the ligands.
Low Spin Complex:
- In a low spin complex, the electrons occupy the lower energy d-orbitals before pairing up.
- This results in a smaller crystal field splitting energy and a larger energy gap between the lower and higher energy d-orbitals.
- The ligands in a low spin complex are usually strong field ligands, which cause a large crystal field splitting.
- The low spin complex is formed when the pairing energy (P) is greater than the crystal field splitting energy (Δ).
High Spin Complex:
- In a high spin complex, the electrons occupy the higher energy d-orbitals before pairing up.
- This results in a larger crystal field splitting energy and a smaller energy gap between the lower and higher energy d-orbitals.
- The ligands in a high spin complex are usually weak field ligands, which cause a small crystal field splitting.
- The high spin complex is formed when the crystal field splitting energy (Δ) is greater than the pairing energy (P).
Inner Orbital Complex:
- In an inner orbital complex, the ligands directly coordinate to the metal ion's d-orbitals.
- The d-orbitals involved in bonding are the inner d-orbitals, which are closer to the nucleus.
- The inner orbital complex is formed when the metal ion has vacant inner d-orbitals available for bonding.
Outer Orbital Complex:
- In an outer orbital complex, the ligands interact with the metal ion's outer d-orbitals.
- The outer d-orbitals are farther from the nucleus and have higher energy compared to the inner d-orbitals.
- The outer orbital complex is formed when the metal ion has vacant outer d-orbitals available for bonding.
Conclusion:
From the given options, the formation of complex involving d-orbitals of the outer shell can be referred to as both high spin complex and outer orbital complex. Therefore, the correct answers are option B (high spin complex) and option D (outer orbital complex).
The number of unpaired electrons calculated in [Co(NH3)6]3+ and [CoF6]3- are- a)4 and 4
- b)0 and 2
- c)2 and 4
- d)0 and 4
Correct answer is option 'D'. Can you explain this answer?
The number of unpaired electrons calculated in [Co(NH3)6]3+ and [CoF6]3- are
a)
4 and 4
b)
0 and 2
c)
2 and 4
d)
0 and 4
 | Bhargavi Banerjee answered |
In [Co(NH3)6]3+ due to pairing cobalt configuration 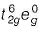 and it is diamagnetic.
and it is diamagnetic.
In [CoF6]3- , F- being weak ligand pairing does not occur and Co3+ ion has configuration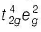 . It has 4 unpaired electrons.
. It has 4 unpaired electrons.
In [CoF6]3- , F- being weak ligand pairing does not occur and Co3+ ion has configuration
Which is the correct?- a)[Fe(NCS)6]3- = blood red
- b)[Fe(H2O)6]2+ = pale green
- c)[Cu(H2O)4]2+ = blue
- d)K2[Hgl4] = scarlet red
Correct answer is option 'A,B,C'. Can you explain this answer?
Which is the correct?
a)
[Fe(NCS)6]3- = blood red
b)
[Fe(H2O)6]2+ = pale green
c)
[Cu(H2O)4]2+ = blue
d)
K2[Hgl4] = scarlet red
 | Prashanth Banerjee answered |
[Fe(NCS)6]3-, [Fe(H2O)6]2+ ,[Cu(H2O)4]2+ are correct.
Match the Column I with Column II and mark the correct option from the codes given below.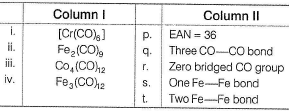

- a)a
- b)b
- c)c
- d)d
Correct answer is option 'A'. Can you explain this answer?
Match the Column I with Column II and mark the correct option from the codes given below.
a)
a
b)
b
c)
c
d)
d
 | Surbhi Sengupta answered |
(i) → (p,r), (ii) → (s), (iii) → (q), (iv) → (t)
Wilkinson’s catalyst is- a)[Rh(CO)2I2]-
- b)[(Ph3P)3RhCI]
- c)[Co2(CO)8]
- d)[(Ph3P)2Rh(CO)Cl]
Correct answer is option 'B'. Can you explain this answer?
Wilkinson’s catalyst is
a)
[Rh(CO)2I2]-
b)
[(Ph3P)3RhCI]
c)
[Co2(CO)8]
d)
[(Ph3P)2Rh(CO)Cl]
 | Devansh Joshi answered |
Wilkinson’s catalyst is a rhodium complex [(Ph3P)3RhCI] and it is used as a catalyst for hydrogenation of alkenes reactions.
In Mn2(CO)10, the number of CO molecules in between the metal atoms are
Correct answer is '0'. Can you explain this answer?
In Mn2(CO)10, the number of CO molecules in between the metal atoms are
 | Ujwal Patel answered |
Mn(CO)5 is radical with 17-electron configuration. Therefore, it undergoes dimerisation through the formation of M—M single bonds and complex their valence shells to avoid ligand-ligand repulsion the Mn(CO)5 units acquire staggered configuration.
In [Co2(CO)8], the number of CO molecules lying between the metal atoms are
Correct answer is '2'. Can you explain this answer?
In [Co2(CO)8], the number of CO molecules lying between the metal atoms are
 | Yash Kumar answered |
Each cobalt atom is bonded to three terminal CO group and share two CO ligands as bridging groups.
Which of the following complex has zero magnetic moment (spin only)?- a)[Ni(NH3)6]CI2
- b)Na3[FeF6]
- c)[Cr(H2O)6]SO4
- d)K4[Fe(CN)6]
Correct answer is option 'D'. Can you explain this answer?
Which of the following complex has zero magnetic moment (spin only)?
a)
[Ni(NH3)6]CI2
b)
Na3[FeF6]
c)
[Cr(H2O)6]SO4
d)
K4[Fe(CN)6]
 | Anuj Iyer answered |
The potassium ferrocyanide, K4 [Fe(CN)6] due to strong ligand pairing of electrons occur and it has no unpaired electrons.
One Integer Value Correct TypeDirection (Q. Nos. 20-24) This section contains 5 questions. When worked out will result in an integer from 0 to 9 (both inclusive).Q. The number of terminal carbonyl groups present in Fe2(CO)9 are
Correct answer is '6'. Can you explain this answer?
One Integer Value Correct Type
Direction (Q. Nos. 20-24) This section contains 5 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
Q.
The number of terminal carbonyl groups present in Fe2(CO)9 are
| | Siddharth Tiwari answered |
The number of terminal carbonyl groups present in Fe2(CO)9
The given compound is Fe2(CO)9. To determine the number of terminal carbonyl groups present in this compound, we need to understand the structure of the compound and the concept of terminal carbonyl groups.
Terminal carbonyl groups refer to carbonyl groups that are directly bonded to a metal atom in a coordination compound. In this case, Fe2(CO)9, Fe represents the metal atom iron, and CO represents the carbonyl ligand.
Explanation:
1. Structure of Fe2(CO)9:
- The compound Fe2(CO)9 consists of two iron (Fe) atoms bonded to nine carbonyl (CO) ligands.
- Each carbonyl ligand is composed of a carbon atom double bonded to an oxygen atom (C=O).
2. Formation of terminal carbonyl groups:
- In coordination compounds, carbonyl ligands can be either terminal or bridging.
- Terminal carbonyl ligands are directly bonded to the metal atom, whereas bridging carbonyl ligands are bonded to two or more metal atoms.
- In Fe2(CO)9, all the carbonyl ligands are terminal because they are directly bonded to the iron (Fe) atoms.
3. Counting the number of terminal carbonyl groups:
- Since Fe2(CO)9 has a total of nine carbonyl ligands, all of which are terminal, the number of terminal carbonyl groups is equal to the total number of carbonyl ligands.
- Therefore, there are 9 terminal carbonyl groups in Fe2(CO)9.
Answer: The number of terminal carbonyl groups present in Fe2(CO)9 is 9.
The given compound is Fe2(CO)9. To determine the number of terminal carbonyl groups present in this compound, we need to understand the structure of the compound and the concept of terminal carbonyl groups.
Terminal carbonyl groups refer to carbonyl groups that are directly bonded to a metal atom in a coordination compound. In this case, Fe2(CO)9, Fe represents the metal atom iron, and CO represents the carbonyl ligand.
Explanation:
1. Structure of Fe2(CO)9:
- The compound Fe2(CO)9 consists of two iron (Fe) atoms bonded to nine carbonyl (CO) ligands.
- Each carbonyl ligand is composed of a carbon atom double bonded to an oxygen atom (C=O).
2. Formation of terminal carbonyl groups:
- In coordination compounds, carbonyl ligands can be either terminal or bridging.
- Terminal carbonyl ligands are directly bonded to the metal atom, whereas bridging carbonyl ligands are bonded to two or more metal atoms.
- In Fe2(CO)9, all the carbonyl ligands are terminal because they are directly bonded to the iron (Fe) atoms.
3. Counting the number of terminal carbonyl groups:
- Since Fe2(CO)9 has a total of nine carbonyl ligands, all of which are terminal, the number of terminal carbonyl groups is equal to the total number of carbonyl ligands.
- Therefore, there are 9 terminal carbonyl groups in Fe2(CO)9.
Answer: The number of terminal carbonyl groups present in Fe2(CO)9 is 9.
In which of the following the central atom has sp3d2-hybridisation?- a)[CoF6]3-
- b)[Co(NH3)6]3+
- c)[Fe(CN)6]3-
- d)[Cr(NH3)6]3+
Correct answer is option 'A'. Can you explain this answer?
In which of the following the central atom has sp3d2-hybridisation?
a)
[CoF6]3-
b)
[Co(NH3)6]3+
c)
[Fe(CN)6]3-
d)
[Cr(NH3)6]3+
 | Tejas Singh answered |
Since, F- ion is a weak ligand hence, pairing of electrons does not occur and outer orbital complex is formed in [CoF6]3-.
The number of unpaired electrons in the complex [Mn(acac)3] is(Atomic number of Mn= 25)
Correct answer is '4'. Can you explain this answer?
The number of unpaired electrons in the complex [Mn(acac)3] is
(Atomic number of Mn= 25)
 | Kunal Pillai answered |
In this, Mn has +3 oxidation state. Mn3+ configuration = [Ar]3d4. This forms outer complex. Hence, unpaired electrons are 4.
Statement I : The oxidation num ber of platinum in Zeise’s salt is +4.Statement II : Zeise’s salt is ionic complex.- a)Both Statement I and Statement II are correct and Statement II is the correct explanation of Statement I
- b)Both Statement I and Statement II are correct but Statement II is not the correct explanation of Statement I
- c)Statement I is correct but Statement II is incorrect
- d)Statement II is correct but Statement I is incorrect
Correct answer is option 'D'. Can you explain this answer?
Statement I : The oxidation num ber of platinum in Zeise’s salt is +4.
Statement II : Zeise’s salt is ionic complex.
a)
Both Statement I and Statement II are correct and Statement II is the correct explanation of Statement I
b)
Both Statement I and Statement II are correct but Statement II is not the correct explanation of Statement I
c)
Statement I is correct but Statement II is incorrect
d)
Statement II is correct but Statement I is incorrect
| | Aravind Rane answered |
Compound is +4.
Statement II : The coordination number of platinum in Zeise compound is 6.
Statement I: The oxidation number of platinum in Zeise compound is +4. This is because the compound is [PtCl4(C2H4)]2-, and each chloride ion has a -1 charge, and the ethylene (C2H4) molecule has a neutral charge. Therefore, the total charge of the compound is -2. Since there are four chloride ions with a -1 charge, the oxidation number of platinum must be +4 to balance out the negative charges.
Statement II: The coordination number of platinum in Zeise compound is 6. This is because there are six ligands attached to the central platinum atom - four chloride ions and two ethylene molecules. Therefore, the coordination number is 6.
Statement II : The coordination number of platinum in Zeise compound is 6.
Statement I: The oxidation number of platinum in Zeise compound is +4. This is because the compound is [PtCl4(C2H4)]2-, and each chloride ion has a -1 charge, and the ethylene (C2H4) molecule has a neutral charge. Therefore, the total charge of the compound is -2. Since there are four chloride ions with a -1 charge, the oxidation number of platinum must be +4 to balance out the negative charges.
Statement II: The coordination number of platinum in Zeise compound is 6. This is because there are six ligands attached to the central platinum atom - four chloride ions and two ethylene molecules. Therefore, the coordination number is 6.
Which one of the following complex species does not involve inner orbital hybridisation?- a)[CoF6]3-
- b)[Co(NH3)6]3+
- c)[Fe(CN)6]3-
- d)[Cr(NH3)6]3+
Correct answer is option 'A'. Can you explain this answer?
Which one of the following complex species does not involve inner orbital hybridisation?
a)
[CoF6]3-
b)
[Co(NH3)6]3+
c)
[Fe(CN)6]3-
d)
[Cr(NH3)6]3+
 | Om Kumar answered |
F- ion is weak ligand hence, pairing of electrons does not occur and outer orbital complex is formed in [CoF6]3-.
[Fe(H2O)6]2+ and [Fe(CN)6]4- differ in- a)magnetic moment
- b)colour
- c)hybridisation
- d)geometry
Correct answer is option 'A,B,C'. Can you explain this answer?
[Fe(H2O)6]2+ and [Fe(CN)6]4- differ in
a)
magnetic moment
b)
colour
c)
hybridisation
d)
geometry
 | Bhargavi Banerjee answered |
[Fe(H2O)6]2+ and [Fe(CN)6]4- have different hybridisation, magnetic moment and colour also.
Chapter doubts & questions for Coordination Compounds - 2 Year Preparation Course for JEE 2026 is part of JEE exam preparation. The chapters have been prepared according to the JEE exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for JEE 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Coordination Compounds - 2 Year Preparation Course for JEE in English & Hindi are available as part of JEE exam. Download more important topics, notes, lectures and mock test series for JEE Exam by signing up for free.
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup





