All Exams > JEE > 2 Year Preparation Course for JEE > All Questions
All questions of Atoms for JEE Exam
Number of alpha particles N scattered at an angle θ during Rutherford’s alpha scattering experiment is :- a)
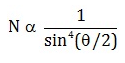
- b)
- c)
- d)
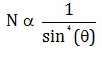
Correct answer is option 'A'. Can you explain this answer?
Number of alpha particles N scattered at an angle θ during Rutherford’s alpha scattering experiment is :
a)
b)
c)
d)
 | Sushil Kumar answered |
Answer :- a
Solution :- For a single scatterer, such as a single gold nucleus within a thin gold foil layer, the differential scattering cross section is defined as follows [2]:
dσ(θ, φ)/dΩ = flux scattered into element dΩ at angles (θ, φ)/incident flux per unit area
dσ/dΩ = (Iθ × A)/ (dΩ × I0 × NAvo × ρ × x(foil))........... (1)
where NAvo is Avogadro’s number, xfoil is the thickness of the target foil, A is the atomic mass of the material in the target foil, dΩ is the solid-angle of the detector, I0 is the unattenuated intensity of the alpha particle beam.
dσ/dΩ = [(ZZ0 e^2/4E )^2]/(1/sin^4(θ/2 ))
dσ/dΩ is directly proportional to 1/sin^4(θ/2 )
Select an incorrect alternative:
i. the radius of the nth orbit is proprtional to n2
ii. the total energy of the electron in the nth orbit is inversely proportional to n
iii. the angular momentum of the electron in nth orbit is an integral multiple of h/2π
iv. the magnitude of potential energy of the electron in any orbit is greater than its kinetic energy- a)Statement i
- b)Statement iv
- c)Statement ii
- d)Statement iii
Correct answer is option 'C'. Can you explain this answer?
Select an incorrect alternative:
i. the radius of the nth orbit is proprtional to n2
ii. the total energy of the electron in the nth orbit is inversely proportional to n
iii. the angular momentum of the electron in nth orbit is an integral multiple of h/2π
iv. the magnitude of potential energy of the electron in any orbit is greater than its kinetic energy
i. the radius of the nth orbit is proprtional to n2
ii. the total energy of the electron in the nth orbit is inversely proportional to n
iii. the angular momentum of the electron in nth orbit is an integral multiple of h/2π
iv. the magnitude of potential energy of the electron in any orbit is greater than its kinetic energy
a)
Statement i
b)
Statement iv
c)
Statement ii
d)
Statement iii
| | Hansa Sharma answered |
Statement i. Radius of Bohr's orbit of hydrogen atom is given by
r= n2h2/4π2mKze2
or, r=(0.59A˚)(n2/z)
So, from expression we found r∝n2
Hence the 1st statement is correct.
Statement ii.
We know that
En=-13.6 x z2/n2
So, En ∝1/n2
Hence the 2nd statement is wrong.
Statement iii.Bohr defined these stable orbits in his second postulate. According to this postulate:
r= n2h2/4π2mKze2
or, r=(0.59A˚)(n2/z)
So, from expression we found r∝n2
Hence the 1st statement is correct.
Statement ii.
We know that
En=-13.6 x z2/n2
So, En ∝1/n2
Hence the 2nd statement is wrong.
Statement iii.Bohr defined these stable orbits in his second postulate. According to this postulate:
- An electron revolves around the nucleus in orbits
- The angular momentum of revolution is an integral multiple of h/2π – where Planck’s constant [h = 6.6 x 10-34 J-s].
- Hence, the angular momentum (L) of the orbiting electron is: L = nh/2 π
Hence the 3rd statement is correct.
Statement iv.According to Bohr's theory
Angular momentum of electron in an orbit will be Integral multiple of (h/2π)
Magnitude of potential energy is twice of kinetic energy of electron in an orbit
∣P.E∣=2∣K.E∣
K.E=(13.6ev)( z2/n2)
Hence, The 4th statement is correct.
Statement iv.According to Bohr's theory
Angular momentum of electron in an orbit will be Integral multiple of (h/2π)
Magnitude of potential energy is twice of kinetic energy of electron in an orbit
∣P.E∣=2∣K.E∣
K.E=(13.6ev)( z2/n2)
Hence, The 4th statement is correct.
Rutherford’s experiments on scattering of alpha particles proved that:- a)atoms contain electrons
- b)number of positive charges is equal to the number of negative charges
- c)atom is mostly empty
- d)positive charge is uniformly distributed in the atom
Correct answer is option 'C'. Can you explain this answer?
Rutherford’s experiments on scattering of alpha particles proved that:
a)
atoms contain electrons
b)
number of positive charges is equal to the number of negative charges
c)
atom is mostly empty
d)
positive charge is uniformly distributed in the atom
 | Divey Sethi answered |
Most of the α-particle passed through the foil straight without suffering any change in their direction. This shows that most of the space inside the atom is empty or hollow.
A small fraction of α-particles was deflected through small angles and a few through larger angles. For this to happen α- particles (positively charged) must approach a heavy positively charged core inside the atom (like charges repel each other). This heavy positively charged core inside the atom was named as the nucleus.
A small fraction of α-particles was deflected through small angles and a few through larger angles. For this to happen α- particles (positively charged) must approach a heavy positively charged core inside the atom (like charges repel each other). This heavy positively charged core inside the atom was named as the nucleus.
In Rutherford’s experiment, a thin gold foil was bombarded with alpha particles. According to Thomson’s “plum-pudding” model of the atom, what should have happened?- a)All the alpha particles would have been deflected by the foil.
- b)All the alpha particles should have bounced straight back from the foil.
- c)Alpha particles should have passed through the foil with little or no deflection.
- d)Alpha particles should have become embedded in the foil.
Correct answer is option 'C'. Can you explain this answer?
In Rutherford’s experiment, a thin gold foil was bombarded with alpha particles. According to Thomson’s “plum-pudding” model of the atom, what should have happened?
a)
All the alpha particles would have been deflected by the foil.
b)
All the alpha particles should have bounced straight back from the foil.
c)
Alpha particles should have passed through the foil with little or no deflection.
d)
Alpha particles should have become embedded in the foil.
 | Snehal Gosavi answered |
Correct Option C ===>ΔΔΔ ΔAlfa particles are massive particles and they have speed when they bombarded.... ΔAccording plum pudding model if atom given by Thomson protons and electrons are equally distributed.... Δthat's why massive alpha particles will pass through the foil with little deflection (due to protons)...... ΔBcoz here protons are like spreaded cloud and not in nucleus ( massive part or atom) .... Δand alpha particle is more massive than single proton....so they will not deflect due to protons and will pass through foil....
In hydrogen atom the kinetic energy of electron in an orbit of radius r is given by- a)
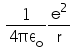
- b)
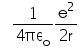
- c)
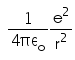
- d)
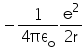
Correct answer is option 'B'. Can you explain this answer?
In hydrogen atom the kinetic energy of electron in an orbit of radius r is given by
a)
b)
c)
d)
| | Swati Verma answered |
K.E. of nth orbit
=> (1/k) Ze2/2r
For H atom,
K.E.=(1/4πε) x (e2/2r)
=> (1/k) Ze2/2r
For H atom,
K.E.=(1/4πε) x (e2/2r)
We know that the Rutherford model of the atom is superior to the Thompson model because when alpha particles are scattered from atoms:- a)the deflected angle is always large
- b)they are usually observed with kinetic energy between 5 and 10 MeV
- c)some alpha particles are deflected to large angles
- d)the deflected angle is usually small
Correct answer is option 'C'. Can you explain this answer?
We know that the Rutherford model of the atom is superior to the Thompson model because when alpha particles are scattered from atoms:
a)
the deflected angle is always large
b)
they are usually observed with kinetic energy between 5 and 10 MeV
c)
some alpha particles are deflected to large angles
d)
the deflected angle is usually small
| | Tanuja Kapoor answered |
In ruther ford experiment he suggest that all the positive charge and mass are concentrated at the centre when he bombarded the alpha partical which is dipositive in nature and when it is more close to centre it get deflect to a large angle and with increase of closenes to centre its deflection angle increase and some alpha partical deflect to 180 degree so it prove that all the positive charge and mass are concentrated at the centre where as acccording to thomson atom is hard solid sphere in which its total +ve charge and mass uniformalyy distributed on the surface and electrone reside as seed in watermelon ( plum pudding model)
Which of these is true?- a)The alpha particles used in Rutherford’s experiment are positively charged particles
- b)Dalton assumed that atoms are made up of electrons, protons, and neutrons
- c)In Rutherford’s alpha scattering experiment, all of the alpha particles passed through the gold foil.
- d)JJ Thomson determined the charge and mass of electrons
Correct answer is 'A'. Can you explain this answer?
Which of these is true?
a)
The alpha particles used in Rutherford’s experiment are positively charged particles
b)
Dalton assumed that atoms are made up of electrons, protons, and neutrons
c)
In Rutherford’s alpha scattering experiment, all of the alpha particles passed through the gold foil.
d)
JJ Thomson determined the charge and mass of electrons
| | Shalini Basu answered |
**Explanation:**
The correct answer is **a) The alpha particles used in Rutherford's experiment are positively charged particles**.
Rutherford's experiment, also known as the gold foil experiment, was conducted in 1909 by Ernest Rutherford and his colleagues Hans Geiger and Ernest Marsden. The experiment aimed to understand the structure of the atom and investigate the distribution of positive charge within it.
In this experiment, Rutherford and his team bombarded a thin gold foil with a beam of alpha particles. Alpha particles are positively charged particles that consist of two protons and two neutrons, which are the same as helium nuclei. They are emitted from a radioactive source, such as radium or polonium.
Rutherford observed that while most of the alpha particles passed straight through the gold foil, some of them were deflected at different angles, and a very small fraction bounced back in the direction opposite to the source. This unexpected result led to the discovery of the atomic nucleus and the concept of a mostly empty space within the atom.
Based on the observations from the experiment, Rutherford proposed a new atomic model known as the nuclear model. According to this model, atoms have a dense, positively charged nucleus at the center, which contains most of the atom's mass. The electrons, which are negatively charged particles, orbit around the nucleus in specific energy levels.
Therefore, the correct answer is a) The alpha particles used in Rutherford's experiment are positively charged particles.
The correct answer is **a) The alpha particles used in Rutherford's experiment are positively charged particles**.
Rutherford's experiment, also known as the gold foil experiment, was conducted in 1909 by Ernest Rutherford and his colleagues Hans Geiger and Ernest Marsden. The experiment aimed to understand the structure of the atom and investigate the distribution of positive charge within it.
In this experiment, Rutherford and his team bombarded a thin gold foil with a beam of alpha particles. Alpha particles are positively charged particles that consist of two protons and two neutrons, which are the same as helium nuclei. They are emitted from a radioactive source, such as radium or polonium.
Rutherford observed that while most of the alpha particles passed straight through the gold foil, some of them were deflected at different angles, and a very small fraction bounced back in the direction opposite to the source. This unexpected result led to the discovery of the atomic nucleus and the concept of a mostly empty space within the atom.
Based on the observations from the experiment, Rutherford proposed a new atomic model known as the nuclear model. According to this model, atoms have a dense, positively charged nucleus at the center, which contains most of the atom's mass. The electrons, which are negatively charged particles, orbit around the nucleus in specific energy levels.
Therefore, the correct answer is a) The alpha particles used in Rutherford's experiment are positively charged particles.
Emission line spectra of different elements- a)is not different
- b)is the same if both elements are at the same temperature
- c)is the same if both elements are in liquid form
- d)is different
Correct answer is option 'D'. Can you explain this answer?
Emission line spectra of different elements
a)
is not different
b)
is the same if both elements are at the same temperature
c)
is the same if both elements are in liquid form
d)
is different
 | Adithya Shasan answered |
Option D : Emission line spectra of different elements is different..
In hydrogen atom the angular momentum of the electron in the lowest energy state is- a)2h
- b)h/2π
- c)2π/h
- d)h/π
Correct answer is option 'B'. Can you explain this answer?
In hydrogen atom the angular momentum of the electron in the lowest energy state is
a)
2h
b)
h/2π
c)
2π/h
d)
h/π
| | Kiran Khanna answered |
C)h/π
d)h
The correct answer is d) h.
The angular momentum of an electron in the hydrogen atom is given by the formula L = nħ, where n is the principal quantum number and ħ is the reduced Planck constant.
In the lowest energy state, the electron is in the ground state with n = 1. Therefore, the angular momentum is L = 1ħ = h.
d)h
The correct answer is d) h.
The angular momentum of an electron in the hydrogen atom is given by the formula L = nħ, where n is the principal quantum number and ħ is the reduced Planck constant.
In the lowest energy state, the electron is in the ground state with n = 1. Therefore, the angular momentum is L = 1ħ = h.
The distance of closest approach when a 15.0 MeV proton approaches gold nucleus (Z = 79) is- a)758 fm
- b)7.58 fm
- c)75.8 fm
- d)0.758 fm
Correct answer is option 'B'. Can you explain this answer?
The distance of closest approach when a 15.0 MeV proton approaches gold nucleus (Z = 79) is
a)
758 fm
b)
7.58 fm
c)
75.8 fm
d)
0.758 fm
| | Riya Banerjee answered |
Correct Answer :- b
Explanation : E = 15.0MeV
= 15 * 106 eV
= 15 * 106 * 1.6 * 10-19 J
= 15 * 1.6 * 10-13 J
E = (1/4πεo)*(ze2/r02)
r0 = (1/4πεo)*(ze2/E)
r0 = (9*109*79*(1.6*10-19)2)/(15*1.6*10-13)
= 75.84 * 10-16 m
= 7.58 fm
In Geiger-Marsden experiment very small deflection of the beam was expected because- a)there are no electrical forces at work
- b)positive charge and the negative electrons are distributed through the whole atom reducing electric field inside the atom
- c)particles are collimated by lead screens
- d)most particles pass through
Correct answer is option 'B'. Can you explain this answer?
In Geiger-Marsden experiment very small deflection of the beam was expected because
a)
there are no electrical forces at work
b)
positive charge and the negative electrons are distributed through the whole atom reducing electric field inside the atom
c)
particles are collimated by lead screens
d)
most particles pass through
| | Shraddha Choudhury answered |
Explanation:
The Geiger-Marsden experiment was conducted to study the structure of an atom. In this experiment, a beam of alpha particles was directed towards a thin gold foil. The alpha particles were expected to pass through the gold foil with little or no deflection, as it was believed that the positive charge and the negative electrons in an atom are distributed uniformly, reducing the electric field inside the atom. However, the results of the experiment were surprising, as some of the alpha particles were deflected at large angles, and some even bounced back.
Reasons for very small deflection of the beam:
- Electrical forces: According to Coulomb's law, any two charged particles exert a force on each other. In an atom, the positively charged nucleus and the negatively charged electrons are attracted to each other by electrical forces. However, the electrons are in constant motion, creating a cloud of negative charge around the nucleus. This cloud of negative charge reduces the electric field inside the atom, making it difficult for the alpha particles to be deflected.
- Distribution of charge: The positive charge in an atom is concentrated in the nucleus, while the negative charge is distributed throughout the atom. This distribution of charge makes the electric field inside the atom more uniform, reducing the chances of the alpha particles being deflected.
- Collimation of particles: The alpha particles were collimated by lead screens before they were directed towards the gold foil. This was done to ensure that the particles were traveling in a straight line and were not scattered by other particles or objects in the environment.
- Most particles pass through: Despite the above factors, it was still expected that some of the alpha particles would be deflected at small angles due to the random nature of the collisions between the particles and the atoms in the gold foil. However, it was not expected that some of the particles would be deflected at large angles or bounce back, as this implied that the positive charge in an atom was not uniformly distributed.
Conclusion:
In conclusion, the very small deflection of the beam was expected in the Geiger-Marsden experiment due to the distribution of charge in an atom and the reduction of electric field inside the atom. However, the unexpected results of the experiment led to the discovery of the nucleus and the development of the modern atomic model.
The Geiger-Marsden experiment was conducted to study the structure of an atom. In this experiment, a beam of alpha particles was directed towards a thin gold foil. The alpha particles were expected to pass through the gold foil with little or no deflection, as it was believed that the positive charge and the negative electrons in an atom are distributed uniformly, reducing the electric field inside the atom. However, the results of the experiment were surprising, as some of the alpha particles were deflected at large angles, and some even bounced back.
Reasons for very small deflection of the beam:
- Electrical forces: According to Coulomb's law, any two charged particles exert a force on each other. In an atom, the positively charged nucleus and the negatively charged electrons are attracted to each other by electrical forces. However, the electrons are in constant motion, creating a cloud of negative charge around the nucleus. This cloud of negative charge reduces the electric field inside the atom, making it difficult for the alpha particles to be deflected.
- Distribution of charge: The positive charge in an atom is concentrated in the nucleus, while the negative charge is distributed throughout the atom. This distribution of charge makes the electric field inside the atom more uniform, reducing the chances of the alpha particles being deflected.
- Collimation of particles: The alpha particles were collimated by lead screens before they were directed towards the gold foil. This was done to ensure that the particles were traveling in a straight line and were not scattered by other particles or objects in the environment.
- Most particles pass through: Despite the above factors, it was still expected that some of the alpha particles would be deflected at small angles due to the random nature of the collisions between the particles and the atoms in the gold foil. However, it was not expected that some of the particles would be deflected at large angles or bounce back, as this implied that the positive charge in an atom was not uniformly distributed.
Conclusion:
In conclusion, the very small deflection of the beam was expected in the Geiger-Marsden experiment due to the distribution of charge in an atom and the reduction of electric field inside the atom. However, the unexpected results of the experiment led to the discovery of the nucleus and the development of the modern atomic model.
The ratio of volume of atom to volume of nucleus is- a)1/1000
- b)10
- c)1015
- d)1010
Correct answer is option 'C'. Can you explain this answer?
The ratio of volume of atom to volume of nucleus is
a)
1/1000
b)
10
c)
1015
d)
1010
| | Anaya Patel answered |
The ratio of the volume of the atom and the volume of the nucleus is 1015
The radius of an atomic nucleus is of the order of 10−13cm or 10−15m or one Fermi unit.
On the other hand, the radius of an atom is of the order of 10−8cm or 10−10m or one angstrom unit.
Note:
The radius of nucleus is much smaller than atomic radius.
The ratio of atomic radius to radius of nucleus is 10−10m /10−15m =105
Volume is proportional to cube of radius.
The ratio of atomic radius to radius of nucleus is (105)3=1015
The radius of an atomic nucleus is of the order of 10−13cm or 10−15m or one Fermi unit.
On the other hand, the radius of an atom is of the order of 10−8cm or 10−10m or one angstrom unit.
Note:
The radius of nucleus is much smaller than atomic radius.
The ratio of atomic radius to radius of nucleus is 10−10m /10−15m =105
Volume is proportional to cube of radius.
The ratio of atomic radius to radius of nucleus is (105)3=1015
Absorption line spectrum is obtained- a)If we pass off-white (discrete-spectrum) light through a cool gas
- b)If we pass white (continuous-spectrum) light through a hot gas
- c)If we pass off-white (discrete-spectrum) light through a hot gas
- d)If we pass white (continuous-spectrum) light through a cool gas
Correct answer is option 'D'. Can you explain this answer?
Absorption line spectrum is obtained
a)
If we pass off-white (discrete-spectrum) light through a cool gas
b)
If we pass white (continuous-spectrum) light through a hot gas
c)
If we pass off-white (discrete-spectrum) light through a hot gas
d)
If we pass white (continuous-spectrum) light through a cool gas
| | Nishant Sharma answered |
Answer is D
In Bohr model of hydrogen atom, radiation is emitted when the electron- a)jumps from higher orbit into the lower orbit
- b)the electron escapes from the orbit
- c)jumps from lower orbit into the higher orbit
- d)revolves in an orbit
Correct answer is option 'A'. Can you explain this answer?
In Bohr model of hydrogen atom, radiation is emitted when the electron
a)
jumps from higher orbit into the lower orbit
b)
the electron escapes from the orbit
c)
jumps from lower orbit into the higher orbit
d)
revolves in an orbit
| | Yamuna Mani answered |
Answer is A
E2-E1=hv
E2-E1=hv
The targets used in the alpha particle atomic experiments in the early 1900’s was:- a)metal foils
- b)alpha particles
- c)radioactive sources
- d)gold foil
Correct answer is option 'D'. Can you explain this answer?
The targets used in the alpha particle atomic experiments in the early 1900’s was:
a)
metal foils
b)
alpha particles
c)
radioactive sources
d)
gold foil
| | Shreya Singh answered |
It's Rutherford alpha Ray scattering experiment.....in this experiment gold foil was used...
Which of these statements about Bohr model hypothesis is correct?- a)velocity of electron is quantized
- b)mass of electron is quantized
- c)radius of electron is quantized
- d)angular momentum of electron is quantized
Correct answer is option 'D'. Can you explain this answer?
Which of these statements about Bohr model hypothesis is correct?
a)
velocity of electron is quantized
b)
mass of electron is quantized
c)
radius of electron is quantized
d)
angular momentum of electron is quantized
| | Krishna Iyer answered |
Bohr never assumed stable electron orbits with the electronic angular momentum quantized as l=mvr=(nh/2π) Quantization of angular momentum means that the radius of the orbit and the energy will be quantized as well. Bohr assumed that the discrete lines seen in the spectrum of the hydrogen atom were due to transitions of an electron from one allowed orbit/energy to another.
In Geiger-Marsden experiment, at the point of closest approach- a)the kinetic energy is not zero and the electrical potential is less than the initial kinetic energy supplied
- b)the kinetic energy is not zero and the electrical potential equals the initial kinetic energy supplied
- c)the kinetic energy is zero and the electrical potential equals the initial kinetic energy supplied
- d)the kinetic energy is not zero and the electrical potential is greater than the initial kinetic energy supplied
Correct answer is option 'C'. Can you explain this answer?
In Geiger-Marsden experiment, at the point of closest approach
a)
the kinetic energy is not zero and the electrical potential is less than the initial kinetic energy supplied
b)
the kinetic energy is not zero and the electrical potential equals the initial kinetic energy supplied
c)
the kinetic energy is zero and the electrical potential equals the initial kinetic energy supplied
d)
the kinetic energy is not zero and the electrical potential is greater than the initial kinetic energy supplied
| | Dino James answered |
The kinetic energy is zero and the electrical potential equals the initial kinetic energy supplied
Which of these statements about Bohr model applied to hydrogen atom correct?- a)hydrogen atom in its ground level has no magnetic moment due to orbital motion
- b)hydrogen atom in its ground level has magnetic moment due to orbital motion
- c)hydrogen atom in some orbits radiates electromagnetic waves
- d)hydrogen atom in quantized orbits radiates electromagnetic waves
Correct answer is option 'B'. Can you explain this answer?
Which of these statements about Bohr model applied to hydrogen atom correct?
a)
hydrogen atom in its ground level has no magnetic moment due to orbital motion
b)
hydrogen atom in its ground level has magnetic moment due to orbital motion
c)
hydrogen atom in some orbits radiates electromagnetic waves
d)
hydrogen atom in quantized orbits radiates electromagnetic waves
| | Swati Verma answered |
A hydrogen atom has magnetic properties because the motion of the electron acts as a current loop. The energy levels of a hydrogen atom associated with orbital angular momentum are split by an external magnetic field because the orbital angular magnetic moment interacts with the field.
Find the longest wavelength present in the Balmer series of hydrogen, corresponding to the H- line.- a)656 nm
- b)676 nm
- c)666 nm
- d)686 nm
Correct answer is option 'A'. Can you explain this answer?
Find the longest wavelength present in the Balmer series of hydrogen, corresponding to the H- line.
a)
656 nm
b)
676 nm
c)
666 nm
d)
686 nm
| | Arun Khanna answered |
If that is so grant us some suggestion! Or Balmer sequence (2nd sequence from electrons laying off to n = 2 from bigger than 2) longest wavelength [velocityconstant = frequency x wavelength] is smallest frequency [power = hconstant x frequency] is least enegy is from transition n = 3 to n = 2 or Google it 656.3 nm
An electron collides with a hydrogen atom in its ground state and excites it to a state of n = 3. How much energy was given to the hydrogen atom in this inelastic collision?- a)
15.1 eV - b)13.1 eV
- c)12.1 eV
- d)14.1 eV
Correct answer is option 'C'. Can you explain this answer?
An electron collides with a hydrogen atom in its ground state and excites it to a state of n = 3. How much energy was given to the hydrogen atom in this inelastic collision?
a)
15.1 eV
b)
13.1 eV
c)
12.1 eV
d)
14.1 eV
| | Ritu Singh answered |
Energy at ground state E1=−13.6 eV
Energy at n=3: E3=−13.6/9=1.5 eV
To excite it to n=3 energy given to electron is E3−E1=12.1 eV
Energy at n=3: E3=−13.6/9=1.5 eV
To excite it to n=3 energy given to electron is E3−E1=12.1 eV
A triply ionized beryllium ion Be3+, (a beryllium atom with three electrons removed), behaves very much like a hydrogen atom except that the nuclear charge is four times as great. For the hydrogen atom, the wavelength of the photon emitted in the n =2 to n=1 to transition is 122 nm. What is the wavelength of the photon emitted when a Be3+ ion undergoes this transition?- a)7.60 nm
- b)10.60 nm
- c)9.60 nm
- d)8.60 nm
Correct answer is option 'A'. Can you explain this answer?
A triply ionized beryllium ion Be3+, (a beryllium atom with three electrons removed), behaves very much like a hydrogen atom except that the nuclear charge is four times as great. For the hydrogen atom, the wavelength of the photon emitted in the n =2 to n=1 to transition is 122 nm. What is the wavelength of the photon emitted when a Be3+ ion undergoes this transition?
a)
7.60 nm
b)
10.60 nm
c)
9.60 nm
d)
8.60 nm
| | Suresh Iyer answered |
1/ λ =Z2. (both have same transition so same value of n)
λ /122=1/16
λ =122/16
=7.62nm
λ /122=1/16
λ =122/16
=7.62nm
According to ‘plum pudding model’ atoms on the whole are electrically neutral because- a)positive charge is concentrated at one place and negative charge is elsewhere.
- b)the negative charge of the atom is uniformly distributed throughout the volume of the atom and the positively charged electrons are embedded in it
- c)the positive charge of the atom is uniformly distributed throughout the volume of the atom and the negatively charged electrons are embedded in it
- d)the positive charge of the atom is uniformly distributed throughout the volume of the electron and the negative charge of electrons balances positive parts
Correct answer is option 'C'. Can you explain this answer?
According to ‘plum pudding model’ atoms on the whole are electrically neutral because
a)
positive charge is concentrated at one place and negative charge is elsewhere.
b)
the negative charge of the atom is uniformly distributed throughout the volume of the atom and the positively charged electrons are embedded in it
c)
the positive charge of the atom is uniformly distributed throughout the volume of the atom and the negatively charged electrons are embedded in it
d)
the positive charge of the atom is uniformly distributed throughout the volume of the electron and the negative charge of electrons balances positive parts
| | Manoj Chauhan answered |
The plum pudding model is one of several scientific models of the atom. According to J.J. Thomson atomic models the positive particles in the atom form something like the "batter" in a plum pudding, while the negative electrons are scattered through this "batter".
Reason why there are many lines in an atomic spectrum is because- a)All atoms are in the same excited state and make transition to same state
- b)There are many atoms in different states of excitation making transition to the same state
- c)There are many atoms in different states of excitation making transition to different states
- d)All atoms are in the same excited state and make transition to different states
Correct answer is option 'C'. Can you explain this answer?
Reason why there are many lines in an atomic spectrum is because
a)
All atoms are in the same excited state and make transition to same state
b)
There are many atoms in different states of excitation making transition to the same state
c)
There are many atoms in different states of excitation making transition to different states
d)
All atoms are in the same excited state and make transition to different states
| | Nishtha Dasgupta answered |
Lines in the spectrum were due to transitions in which an electron moved from a higher-energy orbit with a larger radius to a lower-energy orbit with smaller radius. The orbit closest to the nucleus represented the ground state of the atom and was most stable; orbits farther away were higher-energy excited states.
In a Geiger -Marsden experiment, what is the distance of closest approach d to the nucleus of a 7.7 MeV α−particle before it comes momentarily to rest and reverses its direction?- a)35 fm
- b)40 fm
- c)30 fm
- d)45 fm
Correct answer is option 'C'. Can you explain this answer?
In a Geiger -Marsden experiment, what is the distance of closest approach d to the nucleus of a 7.7 MeV α−particle before it comes momentarily to rest and reverses its direction?
a)
35 fm
b)
40 fm
c)
30 fm
d)
45 fm
 | Lead Academy answered |
The distance of closest approach is given as
r0= (1/4πε0)(2Ze2/E)
Here,
Z= 75
e = 1.6x10-19 C
E = 7.7 MeV = 7.7 × 106X 1.6 X 10-9 J = 1.2×10−12J
1/4πε0 = 9x109 Nm2C-1
so,
r0 = [9x109 x 2x79x(1.6x10-19)2] / [1.2 X 10-12] m
r0=30x10−15 m
r0=30
r0= (1/4πε0)(2Ze2/E)
Here,
Z= 75
e = 1.6x10-19 C
E = 7.7 MeV = 7.7 × 106X 1.6 X 10-9 J = 1.2×10−12J
1/4πε0 = 9x109 Nm2C-1
so,
r0 = [9x109 x 2x79x(1.6x10-19)2] / [1.2 X 10-12] m
r0=30x10−15 m
r0=30
Average angle of deflection of α-particles by a thin gold foil predicted by Thomson’s model is- a)about the same as predicted by Rutherford’s model
- b)incomparable to Rutherford’s model
- c)about the more than predicted by Rutherford’s model
- d)about the less than predicted by Rutherford’s model
Correct answer is option 'A'. Can you explain this answer?
Average angle of deflection of α-particles by a thin gold foil predicted by Thomson’s model is
a)
about the same as predicted by Rutherford’s model
b)
incomparable to Rutherford’s model
c)
about the more than predicted by Rutherford’s model
d)
about the less than predicted by Rutherford’s model
| | Priya Patel answered |
About the same
The average angle of deflection of α-particles by a thin gold foil predicted by Thomson’s model is about the same size as predicted by Rutherford’s model. This is because the average angle was taken in both models.
Which of these statements correctly describe the atomic model according to classical electromagnetic theory ?- a)The electrons would spiral inwards and fall into the nucleus
- b)The waves emitted by electrons are discrete
- c)Electrons do not radiate electromagnetic waves
- d)The positive charge and negative electron cancel and no waves are radiated
Correct answer is option 'A'. Can you explain this answer?
Which of these statements correctly describe the atomic model according to classical electromagnetic theory ?
a)
The electrons would spiral inwards and fall into the nucleus
b)
The waves emitted by electrons are discrete
c)
Electrons do not radiate electromagnetic waves
d)
The positive charge and negative electron cancel and no waves are radiated
| | Avantika Dasgupta answered |
In classical electromagnetic theory, atoms and molecules are considered to contain electrical charges (i.e. electrons, ions) which are regarded as oscillating about positions of equilibrium, each with its appropriate natural frequency, v0 . When placed in a radiation field of frequency v , each oscillator in the atom or molecule is set into forced vibration with the same frequency as that of the radiation. The amplitude of the forced vibration is small, but as v approaches v0 , the amplitude of the forced vibration increases rapidly. To account for the absorption of energy from the radiation field, it is necessary to assume that the oscillator in the atom or molecule must overcome some frictional force proportional to its velocity during its forced motion. For small amplitudes of forced oscillation, the frictional force, and therefore the absorption of energy, is negligible. Near resonance , the amplitude of oscillation becomes large, with a correspondingly large absorption of energy to overcome the frictional force. Therefore, the radiation of frequencies near the natural frequency of the oscillator corresponds to an absorption band.
Which of these statements about Bohr model hypothesis is correct?- a)angular momentum is not quantized
- b)electron in a stable orbit does not radiate electromagnetic waves
- c)velocity of electron is quantized
- d)electron in a stable orbit emit quanta of light
Correct answer is option 'B'. Can you explain this answer?
Which of these statements about Bohr model hypothesis is correct?
a)
angular momentum is not quantized
b)
electron in a stable orbit does not radiate electromagnetic waves
c)
velocity of electron is quantized
d)
electron in a stable orbit emit quanta of light
| | Shraddha Choudhury answered |
Bohr Model Hypothesis and Stable Electron Orbit
Bohr model hypothesis is a model of the atom proposed by Niels Bohr in 1913. It was one of the earliest attempts to explain the structure of atoms and their behavior. The model was based on the assumption that electrons orbit the nucleus in circular paths.
The correct statement about Bohr model hypothesis is:
- Electron in a stable orbit does not radiate electromagnetic waves.
Explanation:
- Electrons in atoms can exist only in certain discrete energy levels. According to Bohr's model, electrons in atoms move around the nucleus in stable orbits, each with a specific energy level.
- Electrons in stable orbits do not emit electromagnetic waves because they are in a stable state and have a fixed amount of energy.
- However, when an electron transitions from a higher energy level to a lower energy level, it emits a photon of light.
- This is because the energy lost by the electron is emitted as a photon of light. The energy of the photon is equal to the difference in energy between the two energy levels.
- Therefore, the Bohr model hypothesis proposed that the electron does not continuously lose energy as it moves in a circular orbit around the nucleus, but instead only loses energy when it transitions between energy levels.
Conclusion:
Bohr model hypothesis is an important model that helps explain the behavior of atoms. The model is based on the assumption that electrons move around the nucleus in stable orbits and only emit electromagnetic waves when they transition between energy levels. The correct statement about the model is that electrons in a stable orbit do not radiate electromagnetic waves.
Bohr model hypothesis is a model of the atom proposed by Niels Bohr in 1913. It was one of the earliest attempts to explain the structure of atoms and their behavior. The model was based on the assumption that electrons orbit the nucleus in circular paths.
The correct statement about Bohr model hypothesis is:
- Electron in a stable orbit does not radiate electromagnetic waves.
Explanation:
- Electrons in atoms can exist only in certain discrete energy levels. According to Bohr's model, electrons in atoms move around the nucleus in stable orbits, each with a specific energy level.
- Electrons in stable orbits do not emit electromagnetic waves because they are in a stable state and have a fixed amount of energy.
- However, when an electron transitions from a higher energy level to a lower energy level, it emits a photon of light.
- This is because the energy lost by the electron is emitted as a photon of light. The energy of the photon is equal to the difference in energy between the two energy levels.
- Therefore, the Bohr model hypothesis proposed that the electron does not continuously lose energy as it moves in a circular orbit around the nucleus, but instead only loses energy when it transitions between energy levels.
Conclusion:
Bohr model hypothesis is an important model that helps explain the behavior of atoms. The model is based on the assumption that electrons move around the nucleus in stable orbits and only emit electromagnetic waves when they transition between energy levels. The correct statement about the model is that electrons in a stable orbit do not radiate electromagnetic waves.
Suppose you are given a chance to repeat the alpha-particle scattering experiment using a thin sheet of solid hydrogen in place of the gold foil. (Hydrogen is a solid at temperatures below 14 K.) What results do you expect?- a)there would be scattering at all angles
- b)there would be no scattering
- c)there would be scattering at 90∘
- d)there would be no large-angle scattering
Correct answer is option 'D'. Can you explain this answer?
Suppose you are given a chance to repeat the alpha-particle scattering experiment using a thin sheet of solid hydrogen in place of the gold foil. (Hydrogen is a solid at temperatures below 14 K.) What results do you expect?
a)
there would be scattering at all angles
b)
there would be no scattering
c)
there would be scattering at 90∘
d)
there would be no large-angle scattering
 | Yash Kumar answered |
In the alpha-particle scattering experiment, if a thin sheet of solid hydrogen is used in place of a gold foil, then the scattering angle would not be large enough. This is because the mass of hydrogen (1.67 x 10 −^27 kg) is less than the mass of incident a−particles (6.64 x 10 ^−27 kg). Thus, the mass of the scattering particle is more than the target nucleus (hydrogen).
As a result, the α−particles would not bounce back if solid hydrogen is used in the α-particle scattering experiment.
As a result, the α−particles would not bounce back if solid hydrogen is used in the α-particle scattering experiment.
The alpha particle scattering experiment was carried out by:- a)J. Chadwick
- b)Rutherford
- c)Goldstein
- d)J.J. Thomson
Correct answer is option 'B'. Can you explain this answer?
The alpha particle scattering experiment was carried out by:
a)
J. Chadwick
b)
Rutherford
c)
Goldstein
d)
J.J. Thomson
| | Arun Khanna answered |
Rutherford Alpha Particle Scattering Experiment.Rutherford directed beams of alpha particles (which are the nuclei of helium atoms and hence positively charged) at thin gold foil to test this model and noted how the alpha particles scattered from the foil.
Which of the following is/are deduced from the Rutherford’s scattering experiment?
(1) There are neutrons inside the nucleus.
(2) The sign of the charge of the nuclei is the same as the sign of alpha particles.
(3) Electrons are embedded in the nucleus.- a)(1) only
- b)(2) only
- c)(3) only
- d)(1) (2) and (3)
Correct answer is option 'B'. Can you explain this answer?
Which of the following is/are deduced from the Rutherford’s scattering experiment?
(1) There are neutrons inside the nucleus.
(2) The sign of the charge of the nuclei is the same as the sign of alpha particles.
(3) Electrons are embedded in the nucleus.
(1) There are neutrons inside the nucleus.
(2) The sign of the charge of the nuclei is the same as the sign of alpha particles.
(3) Electrons are embedded in the nucleus.
a)
(1) only
b)
(2) only
c)
(3) only
d)
(1) (2) and (3)
| | Himaja Ammu answered |
When he sent alpha particles during his gold foil experiment he observed that most of the rays moved away from the nucleus.As we know that like charges always repel ,he concluded that the charge the nuclei is same as the charge of alpha particles
Which of these statements about Bohr model is correct?- a)Bohr model postulates wavy paths around the nucleus
- b)Bohr model combines classical and early quantum concepts
- c)Bohr model is based classical electromagnetic theory
- d)Bohr model is pure quantum mechanical theory
Correct answer is option 'B'. Can you explain this answer?
Which of these statements about Bohr model is correct?
a)
Bohr model postulates wavy paths around the nucleus
b)
Bohr model combines classical and early quantum concepts
c)
Bohr model is based classical electromagnetic theory
d)
Bohr model is pure quantum mechanical theory
| | Isha Rane answered |
Bohr model of the hydrogen atom attempts to plug in certain gaps as suggested by Rutherford’s model by including ideas from the newly developing Quantum hypothesis. According to Rutherford’s model, an atom has a central nucleus and electron/s revolve around it like the sun-planet system.
However, the fundamental difference between the two is that, while the planetary system is held in place by the gravitational force, the nucleus-electron system interacts by Coulomb’s Law of Force. This is because the nucleus and electrons are charged particles. Also, an object moving in a circle undergoes constant acceleration due to the centripetal force.
Further, electromagnetic theory teaches us that an accelerating charged particle emits radiation in the form of electromagnetic waves. Therefore, the energy of such an electron should constantly decrease and the electron should collapse into the nucleus. This would make the atom unstable.
The classical electromagnetic theory also states that the frequency of the electromagnetic waves emitted by an accelerating electron is equal to the frequency of revolution. This would mean that, as the electron spirals inwards, it would emit electromagnetic waves of changing frequencies. In other words, it would emit a continuous spectrum. However, actual observation tells us that the electron emits a line spectrum.
However, the fundamental difference between the two is that, while the planetary system is held in place by the gravitational force, the nucleus-electron system interacts by Coulomb’s Law of Force. This is because the nucleus and electrons are charged particles. Also, an object moving in a circle undergoes constant acceleration due to the centripetal force.
Further, electromagnetic theory teaches us that an accelerating charged particle emits radiation in the form of electromagnetic waves. Therefore, the energy of such an electron should constantly decrease and the electron should collapse into the nucleus. This would make the atom unstable.
The classical electromagnetic theory also states that the frequency of the electromagnetic waves emitted by an accelerating electron is equal to the frequency of revolution. This would mean that, as the electron spirals inwards, it would emit electromagnetic waves of changing frequencies. In other words, it would emit a continuous spectrum. However, actual observation tells us that the electron emits a line spectrum.
Probability of backward scattering (i.e., scattering of α -particles at angles greater than 90∘) predicted by Thomson’s model is- a)much less than predicted by Rutherford’s model
- b)much more than predicted by Rutherford’s model
- c)same as predicted by Rutherford’s model
- d)more than predicted by Rutherford’s model
Correct answer is option 'A'. Can you explain this answer?
Probability of backward scattering (i.e., scattering of α -particles at angles greater than 90∘) predicted by Thomson’s model is
a)
much less than predicted by Rutherford’s model
b)
much more than predicted by Rutherford’s model
c)
same as predicted by Rutherford’s model
d)
more than predicted by Rutherford’s model
| | Sounak Mukherjee answered |
In Rutherford's model we have a large massive core called the nucleus whereas, in Thomson's model we do not have. Thus the probability of backward scattering by Thomson's model is much less than that predicted by Rutherford model.
In which of the models An atom has a nearly continuous mass distribution?- a)Thomson’s model
- b)Bohr model
- c)Rutherford’s model
- d)No model
Correct answer is option 'A'. Can you explain this answer?
In which of the models An atom has a nearly continuous mass distribution?
a)
Thomson’s model
b)
Bohr model
c)
Rutherford’s model
d)
No model
| | Naina Datta answered |
An atom has a nearly continuous mass distribution in Thomson’s model, but has a highly non-uniform mass distribution in Rutherford’s model.
According to Bohr model radiation takes place when- a)there is transition from one of the stable orbits of definite energy to another of higher energy
- b)there is transition from one of the unstable orbits of definite energy to another of higher energy
- c)there is transition from one of the unstable orbits of definite energy to another of same energy
- d)there is transition from one of the stable orbits of definite energy to another of lower energy. hν = Ef−Ei
Correct answer is option 'D'. Can you explain this answer?
According to Bohr model radiation takes place when
a)
there is transition from one of the stable orbits of definite energy to another of higher energy
b)
there is transition from one of the unstable orbits of definite energy to another of higher energy
c)
there is transition from one of the unstable orbits of definite energy to another of same energy
d)
there is transition from one of the stable orbits of definite energy to another of lower energy. hν = Ef−Ei
| | Kalyan Chavan answered |
In 1913 Bohr proposed his quantized shell model of the atom to explain how electrons can have stable orbits around the nucleus. The motion of the electrons in the Rutherford model was unstable because, according to classical mechanics and electromagnetic theory, any charged particle moving on a curved path emits electromagnetic radiation; thus, the electrons would lose energy and spiral into the nucleus. To remedy the stability problem, Bohr modified the Rutherford model by requiring that the electrons move in orbits of fixed size and energy. The energy of an electron depends on the size of the orbit and is lower for smaller orbits. Radiation can occur only when the electron jumps from one orbit to another. The atom will be completely stable in the state with the smallest orbit, since there is no orbit of lower energy into which the electron can jump.
hν = Ef−Ei
hν = Ef−Ei
In the ground state of which model electrons are in stable equilibrium with zero net force?- a)Bohr model
- b)No model
- c)Rutherford’s model
- d)Thomson’s model
Correct answer is option 'D'. Can you explain this answer?
In the ground state of which model electrons are in stable equilibrium with zero net force?
a)
Bohr model
b)
No model
c)
Rutherford’s model
d)
Thomson’s model
| | Jyoti Kapoor answered |
In Thomson's model, the atom is composed of electrons surrounded by a soup of positive charge to balance the electrons' negative charges, like negatively charged “plums” surrounded by positively charged “pudding”. The 1904 Thomson model was disproved by Hans Geiger's and Ernest Marsden's 1909 gold foil experiment.
To explain his theory Bohr used:- a)conservation of quantum frequency
- b)conservation of energy
- c)conservation of linear momentum
- d)conservation of angular momentum
Correct answer is option 'D'. Can you explain this answer?
To explain his theory Bohr used:
a)
conservation of quantum frequency
b)
conservation of energy
c)
conservation of linear momentum
d)
conservation of angular momentum
| | Mansi Nair answered |
Bohr used conservation of angular momentum.
For stationary orbits, Angular momentum Iω=nh2π
where n=1,2,3,...etc
For stationary orbits, Angular momentum Iω=nh2π
where n=1,2,3,...etc
In scattering, the impact parameter b is defined as the:- a)maximum kinetic energy of the scattering projectile
- b)distance of closest approach between projectile and target
- c)closest distance between projectile and target if there were no deflection
- d)distance between projectile and target for which there is maximal deflection
Correct answer is option 'C'. Can you explain this answer?
In scattering, the impact parameter b is defined as the:
a)
maximum kinetic energy of the scattering projectile
b)
distance of closest approach between projectile and target
c)
closest distance between projectile and target if there were no deflection
d)
distance between projectile and target for which there is maximal deflection
| | Rohit Shah answered |
The impact parameter is defined as the perpendicular distance between the path of a projectile and the center of a potential field created by an object that the projectile is approaching. It is often referred to in nuclear physics (see Rutherford scattering) and in classical mechanics.
To produce an emission spectrum of hydrogen- a)It needs to be in glowing gaseous form
- b)It needs to be cold and white light shining through
- c)It needs to be cool liquid form
- d)It needs to be hot and white light shining through
Correct answer is option 'A'. Can you explain this answer?
To produce an emission spectrum of hydrogen
a)
It needs to be in glowing gaseous form
b)
It needs to be cold and white light shining through
c)
It needs to be cool liquid form
d)
It needs to be hot and white light shining through
 | Ishita Reddy answered |
Answer:
To produce an emission spectrum of hydrogen, the hydrogen gas needs to be in a glowing gaseous form. This is because the emission spectrum of an element is produced when the electrons in the atoms of that element are excited to higher energy levels and then fall back to lower energy levels, emitting photons of specific wavelengths in the process.
Glowing Gaseous Form
When hydrogen gas is in a glowing gaseous form, the atoms are excited by an external energy source such as an electric discharge or a flame. This excitation causes the electrons in the hydrogen atoms to move to higher energy levels. As the excited electrons return to their original energy levels, they release energy in the form of photons. The photons emitted have specific wavelengths corresponding to the energy difference between the excited and ground states of the hydrogen atom.
Emission Spectrum
The emitted photons create a spectrum of discrete lines, known as an emission spectrum. These lines are unique to each element and can be used to identify the presence of that element. In the case of hydrogen, the emission spectrum consists of several series of lines, with each series corresponding to a different energy transition within the hydrogen atom.
Other Options
The other options mentioned in the question, such as cold and white light shining through, cool liquid form, and hot and white light shining through, are not suitable for producing an emission spectrum of hydrogen.
- Cold and white light shining through: White light is a combination of all visible wavelengths, and shining it through a cold hydrogen gas would not result in the emission of specific wavelengths characteristic of hydrogen.
- Cool liquid form: When hydrogen is in a cool liquid form, the atoms are not excited, and therefore, no emission spectrum is produced.
- Hot and white light shining through: Similar to the cold and white light scenario, shining white light through hot hydrogen gas would not produce specific wavelengths characteristic of hydrogen.
Conclusion
In conclusion, to produce an emission spectrum of hydrogen, the hydrogen gas must be in a glowing gaseous form. This allows for the excitation of the hydrogen atoms and the subsequent emission of photons with specific wavelengths, creating the characteristic emission spectrum of hydrogen.
Introduction
To produce an emission spectrum of hydrogen, the hydrogen gas needs to be in a glowing gaseous form. This is because the emission spectrum of an element is produced when the electrons in the atoms of that element are excited to higher energy levels and then fall back to lower energy levels, emitting photons of specific wavelengths in the process.
Explanation
Glowing Gaseous Form
When hydrogen gas is in a glowing gaseous form, the atoms are excited by an external energy source such as an electric discharge or a flame. This excitation causes the electrons in the hydrogen atoms to move to higher energy levels. As the excited electrons return to their original energy levels, they release energy in the form of photons. The photons emitted have specific wavelengths corresponding to the energy difference between the excited and ground states of the hydrogen atom.
Emission Spectrum
The emitted photons create a spectrum of discrete lines, known as an emission spectrum. These lines are unique to each element and can be used to identify the presence of that element. In the case of hydrogen, the emission spectrum consists of several series of lines, with each series corresponding to a different energy transition within the hydrogen atom.
Other Options
The other options mentioned in the question, such as cold and white light shining through, cool liquid form, and hot and white light shining through, are not suitable for producing an emission spectrum of hydrogen.
- Cold and white light shining through: White light is a combination of all visible wavelengths, and shining it through a cold hydrogen gas would not result in the emission of specific wavelengths characteristic of hydrogen.
- Cool liquid form: When hydrogen is in a cool liquid form, the atoms are not excited, and therefore, no emission spectrum is produced.
- Hot and white light shining through: Similar to the cold and white light scenario, shining white light through hot hydrogen gas would not produce specific wavelengths characteristic of hydrogen.
Conclusion
In conclusion, to produce an emission spectrum of hydrogen, the hydrogen gas must be in a glowing gaseous form. This allows for the excitation of the hydrogen atoms and the subsequent emission of photons with specific wavelengths, creating the characteristic emission spectrum of hydrogen.
It is found experimentally that for small thickness t, the number of α-particles scattered at moderate angles is proportional to t. What clue does this linear dependence on t provide?- a)scattering is predominantly due to a single collision
- b)scattering is predominantly due to multiple collisions
- c)scattering is predominantly due to deflecting fields being proportional to t
- d)scattering is predominantly due to no collison
Correct answer is option 'A'. Can you explain this answer?
It is found experimentally that for small thickness t, the number of α-particles scattered at moderate angles is proportional to t. What clue does this linear dependence on t provide?
a)
scattering is predominantly due to a single collision
b)
scattering is predominantly due to multiple collisions
c)
scattering is predominantly due to deflecting fields being proportional to t
d)
scattering is predominantly due to no collison
| | Nisha Pillai answered |
Linear Dependence on Thickness t in Particle Scattering
Particle scattering refers to the process of a particle being deflected or redirected from its original trajectory after interacting with another particle or a field. In experiments involving particle scattering, the number of particles scattered at moderate angles is found to be proportional to the thickness t of the scattering material for small t values. This linear dependence on t provides a clue about the nature of the scattering process.
Predominance of Single Collision
The linear dependence of particle scattering on small thickness t suggests that the scattering is predominantly due to a single collision between the incident particle and the scattering material. This means that the incident particle interacts with only one scattering center or atom in the material and is deflected by a small angle. The probability of the incident particle interacting with multiple scattering centers and undergoing multiple collisions is low and does not significantly contribute to the overall scattering.
Explanation of Option A
Option A states that scattering is predominantly due to a single collision. This is consistent with the linear dependence of scattering on small thickness t, as explained above. Therefore, option A is the correct answer.
Conclusion
The linear dependence of particle scattering on small thickness t provides a clue about the nature of the scattering process. In particular, it suggests that scattering is predominantly due to a single collision between the incident particle and the scattering material. This information can be useful in designing experiments and understanding the behavior of particles in various materials.
Particle scattering refers to the process of a particle being deflected or redirected from its original trajectory after interacting with another particle or a field. In experiments involving particle scattering, the number of particles scattered at moderate angles is found to be proportional to the thickness t of the scattering material for small t values. This linear dependence on t provides a clue about the nature of the scattering process.
Predominance of Single Collision
The linear dependence of particle scattering on small thickness t suggests that the scattering is predominantly due to a single collision between the incident particle and the scattering material. This means that the incident particle interacts with only one scattering center or atom in the material and is deflected by a small angle. The probability of the incident particle interacting with multiple scattering centers and undergoing multiple collisions is low and does not significantly contribute to the overall scattering.
Explanation of Option A
Option A states that scattering is predominantly due to a single collision. This is consistent with the linear dependence of scattering on small thickness t, as explained above. Therefore, option A is the correct answer.
Conclusion
The linear dependence of particle scattering on small thickness t provides a clue about the nature of the scattering process. In particular, it suggests that scattering is predominantly due to a single collision between the incident particle and the scattering material. This information can be useful in designing experiments and understanding the behavior of particles in various materials.
If the electron in H atom jumps from the third orbit to second orbit, the wavelength of the emitted radiation is given by- a)
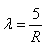
- b)
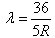
- c)
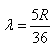
- d)
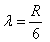
Correct answer is option 'B'. Can you explain this answer?
If the electron in H atom jumps from the third orbit to second orbit, the wavelength of the emitted radiation is given by
a)
b)
c)
d)
 | Ayush Joshi answered |
Wavelength of emitted radiation during transition is given by 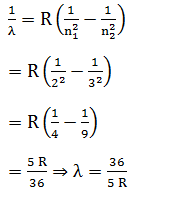
Chapter doubts & questions for Atoms - 2 Year Preparation Course for JEE 2026 is part of JEE exam preparation. The chapters have been prepared according to the JEE exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for JEE 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Atoms - 2 Year Preparation Course for JEE in English & Hindi are available as part of JEE exam. Download more important topics, notes, lectures and mock test series for JEE Exam by signing up for free.
;
Signup on EduRev and stay on top of your study goals
10M+ students crushing their study goals daily