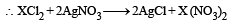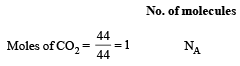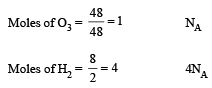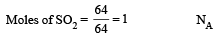All Exams > NEET > 30-Day Revision Course for NEET > All Questions
All questions of Some Basic Concepts of Chemistry for NEET Exam
Chapter doubts & questions for Some Basic Concepts of Chemistry - 30-Day Revision Course for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Some Basic Concepts of Chemistry - 30-Day Revision Course for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
30-Day Revision Course for NEET85 videos|301 docs|171 tests |