All Exams > NEET > 30-Day Revision Course for NEET > All Questions
All questions of Structure of Atom for NEET Exam
Electronic configuration of the element having atomic number 24.- a)
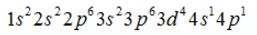
- b)
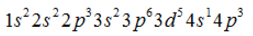
- c)
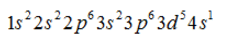
- d)
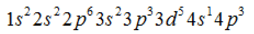
Correct answer is option 'C'. Can you explain this answer?
Electronic configuration of the element having atomic number 24.
a)
b)
c)
d)
| | Ananya Sarkar answered |
atomic no 24 is of Cr. Due to half filled orbital stability it doesnot follow Afbau rule so its configuration is
One of the orbitals is non-directional- a)s- orbital
- b)f- orbital
- c)d- orbital
- d)p- orbital
Correct answer is option 'A'. Can you explain this answer?
One of the orbitals is non-directional
a)
s- orbital
b)
f- orbital
c)
d- orbital
d)
p- orbital
| | Gaurav Kumar answered |
s orbital is spherically symmetrical hence non directional.
An cation A3+ has 18 electrons. Write the atomic number of A.- a)15
- b)24
- c)12
- d)21
Correct answer is option 'D'. Can you explain this answer?
An cation A3+ has 18 electrons. Write the atomic number of A.
a)
15
b)
24
c)
12
d)
21
| | Lavanya Menon answered |
3+ charge means no of electrons will be 3 less than no. of protons. So no. of protons will be 21. So atomic no. is 21.
Daltons atomic theory could not explain one of the following- a)Law of multiple Proportions,
- b)Law of conservation of mass
- c)Law of constant composition
- d)Discovery of sub-atomic particles
Correct answer is option 'D'. Can you explain this answer?
Daltons atomic theory could not explain one of the following
a)
Law of multiple Proportions,
b)
Law of conservation of mass
c)
Law of constant composition
d)
Discovery of sub-atomic particles
| | Om Desai answered |
Dalton’s atomic theory stated that atoms were indivisible. However, the discovery of subatomic particles (such as protons, electrons, and neutrons) disproved this postulate.
Values of e/m (charge/mass) in the categories alpha particle (α), electron (e) and protons (p) increase in the order:- a)α < e < p
- b)p < e < α
- c)e < α < p
- d)α< p < e
Correct answer is option 'D'. Can you explain this answer?
Values of e/m (charge/mass) in the categories alpha particle (α), electron (e) and protons (p) increase in the order:
a)
α < e < p
b)
p < e < α
c)
e < α < p
d)
α< p < e
| | Hansa Sharma answered |
E/m values of particle (α), electron (e) and protons (p) increase in the order α< p < e
Isobars are the atoms with- a)same atomic number but different number of neutrons
- b)same mass number but different atomic number
- c)same atomic number but different mass number
- d)same number of neutrons but different mass number
Correct answer is option 'B'. Can you explain this answer?
Isobars are the atoms with
a)
same atomic number but different number of neutrons
b)
same mass number but different atomic number
c)
same atomic number but different mass number
d)
same number of neutrons but different mass number
| | Nandini Patel answered |
Isobars are atoms (nuclides) of different chemical elements that have the same number of nucleons. Correspondingly, isobars differ in atomic number (or number of protons) but have the same mass number.
The energy associated with the first orbit in the hydrogen atom is -2.18 x 10−18 J/atom. What is the energy associated with the fifth orbit?
a) -7.72 ×10−20 J/atom
b) -5.72 ×10−20 J/atom
c) -3.72 ×10−20 J/atom
d) -8.72 ×10−20 J/atom
Correct answer is 'D'. Can you explain this answer?
| | Geetika Shah answered |
Energy for any n shell = -2.18 x 10−18 )/n'2 (') stands for raise to the power of therefore, E associated with 5 orbit = -2.18 x 10−18/52 = -2.18 × 10-18/25 = -218 × 10-20/25 = -8.72 x 10-20J/atom
As compared to 1s electron of H-atom in ground state, which of the following properties appear(s) in the radial probability density of 2s electron of H-atom in first excited state?- a)Spherical node appear
- b)Electron charge density is highest in the vicinity of the nucleus
- c)Electron density drops to zero after maximum probability is reache
- d)Electron density rises to second highest valu
Correct answer is option 'A,B,C,D'. Can you explain this answer?
As compared to 1s electron of H-atom in ground state, which of the following properties appear(s) in the radial probability density of 2s electron of H-atom in first excited state?
a)
Spherical node appear
b)
Electron charge density is highest in the vicinity of the nucleus
c)
Electron density drops to zero after maximum probability is reache
d)
Electron density rises to second highest valu
 | Jatin Dasgupta answered |
For2s-electron of H-atom (first excited state)
For 1s-electron of H-atom in ground state
At point Ar = a0 r at point Br > a0
(a) True (b) True (c) True (d) True
For 1s-electron of H-atom in ground state
At point Ar = a0 r at point Br > a0
(a) True (b) True (c) True (d) True
Photoelectric effect established that light- a)behaves like particles
- b)behaves like waves
- c)behaves like rays
- d)behaves like magnetic fields
Correct answer is option 'A'. Can you explain this answer?
Photoelectric effect established that light
a)
behaves like particles
b)
behaves like waves
c)
behaves like rays
d)
behaves like magnetic fields
| | Raghav Bansal answered |
Photoelectric effect proves the particle nature of light.
According to Pauli’s exclusion principle- a)No two electrons in an atom can have the same set of four quantum numbers
- b)Any two electrons in an atom can have the same set of four quantum numbers
- c)All electrons in an atom can have the same spin numbers
- d)Any two electrons in an atom can have the same spin numbers
Correct answer is 'A'. Can you explain this answer?
According to Pauli’s exclusion principle
a)
No two electrons in an atom can have the same set of four quantum numbers
b)
Any two electrons in an atom can have the same set of four quantum numbers
c)
All electrons in an atom can have the same spin numbers
d)
Any two electrons in an atom can have the same spin numbers
| | Gaurav Kumar answered |
Paulis exclusion principle stated that no two electron can have same set of all the four quantum nos.
The electronic configuration 1s22s22p1 belongs to- a)Beryllium
- b)lithium
- c)Boron
- d)carbon
Correct answer is option 'C'. Can you explain this answer?
The electronic configuration 1s22s22p1 belongs to
a)
Beryllium
b)
lithium
c)
Boron
d)
carbon
| | Hansa Sharma answered |
It is of boron as it has atomic number 5.
Select the correct statement(s).- a)|
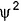 | measures the electron probability density at point in an atom
| measures the electron probability density at point in an atom - b)
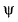 and |
and | 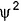 | vary as a function of the three coordinates r (radial), θ and (
| vary as a function of the three coordinates r (radial), θ and (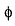 (angular part)
(angular part) - c)Angular wave function ‘θ
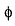 ’ depends an only l and m, and is independent of n
’ depends an only l and m, and is independent of n - d)All of the above are correct statements
Correct answer is option 'D'. Can you explain this answer?
Select the correct statement(s).
a)
| 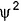 | measures the electron probability density at point in an atom
| measures the electron probability density at point in an atom
b)
c)
Angular wave function ‘θ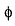 ’ depends an only l and m, and is independent of n
’ depends an only l and m, and is independent of n
d)
All of the above are correct statements
 | Telecom Tuners answered |
Thus, 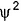 is d e pendent on r true
is d e pendent on r true
(b)
(c) Angular wave function is determined by /land m, and not by n: true
Which orbital gives an electron, a greater probability being found close to the nucleus?- a)3p
- b)3d
- c)3s
- d)equal
Correct answer is option 'C'. Can you explain this answer?
Which orbital gives an electron, a greater probability being found close to the nucleus?
a)
3p
b)
3d
c)
3s
d)
equal
 | Atharva Pillai answered |
3s is spherically symmetrical and its electron density is maximum at the nucleus. It decreases with r.
Radio frequency region of the electromagnetic spectrum is used for broadcasting. It is- a)Around 106 Hz
- b)Around 1010 Hz
- c)Around 1015 Hz
- d)Around 1013 Hz
Correct answer is 'A'. Can you explain this answer?
Radio frequency region of the electromagnetic spectrum is used for broadcasting. It is
a)
Around 106 Hz
b)
Around 1010 Hz
c)
Around 1015 Hz
d)
Around 1013 Hz
| | Hansa Sharma answered |
Radio frequency region is present around 106106 Hz.
spin quantum number with two spin states of the electron represented by two arrows, ↑ (spin up) and ↓ (spin down) was introduced to account for- a)the splitting of emission lines in a magnetic field
- b)anomalous Zeeman effect or the splitting of emission lines into more lines than given by magnetic quantum number
- c)to account for uncertainty in electron states
- d)the splitting of emission lines in an electric field
Correct answer is option 'B'. Can you explain this answer?
spin quantum number with two spin states of the electron represented by two arrows, ↑ (spin up) and ↓ (spin down) was introduced to account for
a)
the splitting of emission lines in a magnetic field
b)
anomalous Zeeman effect or the splitting of emission lines into more lines than given by magnetic quantum number
c)
to account for uncertainty in electron states
d)
the splitting of emission lines in an electric field
| | Jyoti Dey answered |
Explanation:
The spin quantum number is introduced to account for the anomalous Zeeman effect or the splitting of emission lines into more lines than given by the magnetic quantum number. This effect is observed when a magnetic field is applied to an atom, causing the energy levels to split into sub-levels. The splitting of lines is not just dependent on the magnetic quantum number, but also on the spin of the electron.
The spin of an electron is represented by two arrows: (spin up) and (spin down). This means that the electron can have two possible spin states. The spin quantum number is denoted by the symbol 's' and can have a value of either +1/2 or -1/2.
When a magnetic field is applied to an atom, the energy levels split into sub-levels. The number of sub-levels is given by 2s+1, where 's' is the spin quantum number. This means that an electron with a spin quantum number of +1/2 or -1/2 can have two sub-levels.
The anomalous Zeeman effect occurs because the magnetic field affects the energy levels differently depending on the electron's spin. Electrons with spin up are affected differently than electrons with spin down. This causes the energy levels to split into more lines than given by the magnetic quantum number alone.
Conclusion:
Therefore, the spin quantum number is introduced to account for the anomalous Zeeman effect or the splitting of emission lines into more lines than given by the magnetic quantum number.
The spin quantum number is introduced to account for the anomalous Zeeman effect or the splitting of emission lines into more lines than given by the magnetic quantum number. This effect is observed when a magnetic field is applied to an atom, causing the energy levels to split into sub-levels. The splitting of lines is not just dependent on the magnetic quantum number, but also on the spin of the electron.
The spin of an electron is represented by two arrows: (spin up) and (spin down). This means that the electron can have two possible spin states. The spin quantum number is denoted by the symbol 's' and can have a value of either +1/2 or -1/2.
When a magnetic field is applied to an atom, the energy levels split into sub-levels. The number of sub-levels is given by 2s+1, where 's' is the spin quantum number. This means that an electron with a spin quantum number of +1/2 or -1/2 can have two sub-levels.
The anomalous Zeeman effect occurs because the magnetic field affects the energy levels differently depending on the electron's spin. Electrons with spin up are affected differently than electrons with spin down. This causes the energy levels to split into more lines than given by the magnetic quantum number alone.
Conclusion:
Therefore, the spin quantum number is introduced to account for the anomalous Zeeman effect or the splitting of emission lines into more lines than given by the magnetic quantum number.
Give the name and atomic number of the inert gas atom in which the total number of d-electrons is equal to the difference between the numbers of total p and total s electrons.- a)Kr ( atomic no.=36)
- b)Br ( atomic no.=35
- c)Cl ( atomic no.=17)
- d)As ( atomic no.=33)
Correct answer is option 'A'. Can you explain this answer?
Give the name and atomic number of the inert gas atom in which the total number of d-electrons is equal to the difference between the numbers of total p and total s electrons.
a)
Kr ( atomic no.=36)
b)
Br ( atomic no.=35
c)
Cl ( atomic no.=17)
d)
As ( atomic no.=33)
| | Anjana Sharma answered |
It is Krypton .
Electronic configuration: 1s22s22p63s23p64s23d104p6
Number of electrons in s orbital are 8
Number of electrons in p orbital are 18
Number of electrons in d orbitals are 10 (18-8=10)
Electronic configuration: 1s22s22p63s23p64s23d104p6
Number of electrons in s orbital are 8
Number of electrons in p orbital are 18
Number of electrons in d orbitals are 10 (18-8=10)
Arrange the electrons present in the 4d, 3d, 4p and 3p orbitals in order of increasing energies- a)(3p) < (3d) < (4p) < (4d).
- b)(4d) < (4p) < (3p) < (3d)
- c)(4p) < (4d) < (3p) < (3d)
- d)(3d) < (3p) < (4p) < (4d)
Correct answer is option 'A'. Can you explain this answer?
Arrange the electrons present in the 4d, 3d, 4p and 3p orbitals in order of increasing energies
a)
(3p) < (3d) < (4p) < (4d).
b)
(4d) < (4p) < (3p) < (3d)
c)
(4p) < (4d) < (3p) < (3d)
d)
(3d) < (3p) < (4p) < (4d)
| | Suresh Iyer answered |
Energy of orbital is determined by n+l value. The orbital having higher n+l value will have higher energy. If n+l value is same then orbital having higher n value will have higher energy.
Around 1015 Hz corresponds to the region of the electromagnetic spectrum- a)microwave region
- b)ultraviolet region
- c)visible light
- d)infrared region
Correct answer is 'C'. Can you explain this answer?
Around 1015 Hz corresponds to the region of the electromagnetic spectrum
a)
microwave region
b)
ultraviolet region
c)
visible light
d)
infrared region
| | Rahul Bansal answered |
The electromagnetic spectrum is divided into the following regions:
Gamma-rays
[wavelength 10^-14 m - 10^-11 m, frequency 10^22 Hz – 10^19 Hz,
X-rays
[wavelength 10^-12 m - 10^-8 m, frequency 10^20 Hz –10^16 Hz,
Ultraviolet radiation
[wavelength 10^-8 m - 10^-6 m, frequency 10^17 Hz – 10^15 Hz,
Visible light
[wavelength 10^-7 m - 10^-6 m, frequency 10^15 Hz – 10^14 Hz,
Infrared radiation
[wavelength 10^-6 m -10^-3 m, frequency 10^14 Hz – 10^12 Hz,
Microwaves
[wavelength 10^-4 m – 10^-1 m, frequency 10^13 Hz – 10^9 Hz,
Radio waves
[wavelength 10 m – 10^3 m, frequency 10^8 Hz – 10^6 Hz,
Due to the presence of electrons in the inner shells, the electron in the outer shell will not experience the full positive charge of the nucleus (Ze). This is known as- a)charge stealing by inner shell electrons from the outer shell electrons
- b)shielding of the inner shell electrons from the nucleus by the outer shell electrons
- c)shielding of the outer shell electrons from the nucleus by the inner shell electrons
- d)charge stealing by outer shell electrons from the inner shell electrons
Correct answer is option 'C'. Can you explain this answer?
Due to the presence of electrons in the inner shells, the electron in the outer shell will not experience the full positive charge of the nucleus (Ze). This is known as
a)
charge stealing by inner shell electrons from the outer shell electrons
b)
shielding of the inner shell electrons from the nucleus by the outer shell electrons
c)
shielding of the outer shell electrons from the nucleus by the inner shell electrons
d)
charge stealing by outer shell electrons from the inner shell electrons
 | Muskaan Mishra answered |
This is known as shielding effect. Inner lying electrons shields the positive charge reaching the outer most electrons.
In an atom, an electron is moving with a speed of 600m/s with an accuracy of 0.005%. Certainity with which the position of the electron can be located is (h = 6.6 ×10−34 Js)- a)1.92 ×10−3m
- b)5.10 ×10−3m
- c)3.84 ×10−3m
- d)1.52 ×10−4m
Correct answer is option 'A'. Can you explain this answer?
In an atom, an electron is moving with a speed of 600m/s with an accuracy of 0.005%. Certainity with which the position of the electron can be located is (h = 6.6 ×10−34 Js)
a)
1.92 ×10−3m
b)
5.10 ×10−3m
c)
3.84 ×10−3m
d)
1.52 ×10−4m
| | Naina Sharma answered |

Emission spectrum of a material results from the material's (atom or molecules)- a)radiating an inverted spectrum
- b)radiating a continuous spectrum
- c)transition from normal to excited state
- d)transition from excited to normal state
Correct answer is option 'D'. Can you explain this answer?
Emission spectrum of a material results from the material's (atom or molecules)
a)
radiating an inverted spectrum
b)
radiating a continuous spectrum
c)
transition from normal to excited state
d)
transition from excited to normal state
| | Pooja Shah answered |
Emission spectrum is recorded when a electron de excites from higher energy level to ground state.
The Aufbau principle states : In the ground state of the atoms, the orbitals are filled in order of- a)their increasing energies
- b)their decreasing energies
- c)their decreasing radius
- d)their increasing radius
Correct answer is option 'A'. Can you explain this answer?
The Aufbau principle states : In the ground state of the atoms, the orbitals are filled in order of
a)
their increasing energies
b)
their decreasing energies
c)
their decreasing radius
d)
their increasing radius
| | Naina Sharma answered |
According to Aufbau rule electrons are filled in increasing order of energy.
Isotopes are the atoms with- a)same mass number but different atomic number
- b)same atomic number but different number of electrons
- c)same atomic number but different mass number
- d)same number of neutrons but different mass number
Correct answer is option 'C'. Can you explain this answer?
Isotopes are the atoms with
a)
same mass number but different atomic number
b)
same atomic number but different number of electrons
c)
same atomic number but different mass number
d)
same number of neutrons but different mass number
| | Sanchita Chakraborty answered |
Isotopes are the atoms with the same atomic number but different mass number.
Explanation:
Isotopes are variants of a particular chemical element, which have the same number of protons in their atomic nucleus but differ in the number of neutrons. This difference in the number of neutrons leads to variations in their mass numbers.
Atomic Number:
The atomic number of an atom is the number of protons present in its nucleus. It determines the element's identity and its position on the periodic table. For example, all carbon atoms have an atomic number of 6, indicating that they have 6 protons in their nuclei.
Mass Number:
The mass number of an atom is the sum of its protons and neutrons. Since isotopes have the same number of protons (atomic number), their mass numbers differ due to the varying number of neutrons.
Example:
Let's take the example of carbon, which has three isotopes: carbon-12, carbon-13, and carbon-14.
- Carbon-12: It has 6 protons and 6 neutrons, so its mass number is 12 (6 protons + 6 neutrons).
- Carbon-13: It has 6 protons and 7 neutrons, so its mass number is 13 (6 protons + 7 neutrons).
- Carbon-14: It has 6 protons and 8 neutrons, so its mass number is 14 (6 protons + 8 neutrons).
As we can see, all three isotopes have the same atomic number (6) because they all have 6 protons. However, their mass numbers differ due to the varying number of neutrons.
Significance of Isotopes:
Isotopes have several practical applications in various fields. Some examples include:
- Carbon dating: Carbon-14 is used to determine the age of ancient artifacts and fossils.
- Medical imaging: Isotopes such as technetium-99m are used in nuclear medicine for diagnostic purposes.
- Radiocarbon dating: Isotopes like carbon-14 are used to determine the age of organic materials.
- Nuclear power: Isotopes like uranium-235 and plutonium-239 are used as fuel in nuclear reactors.
In conclusion, isotopes are atoms with the same atomic number but different mass numbers. The difference in mass numbers is due to the varying number of neutrons in their nuclei.
Explanation:
Isotopes are variants of a particular chemical element, which have the same number of protons in their atomic nucleus but differ in the number of neutrons. This difference in the number of neutrons leads to variations in their mass numbers.
Atomic Number:
The atomic number of an atom is the number of protons present in its nucleus. It determines the element's identity and its position on the periodic table. For example, all carbon atoms have an atomic number of 6, indicating that they have 6 protons in their nuclei.
Mass Number:
The mass number of an atom is the sum of its protons and neutrons. Since isotopes have the same number of protons (atomic number), their mass numbers differ due to the varying number of neutrons.
Example:
Let's take the example of carbon, which has three isotopes: carbon-12, carbon-13, and carbon-14.
- Carbon-12: It has 6 protons and 6 neutrons, so its mass number is 12 (6 protons + 6 neutrons).
- Carbon-13: It has 6 protons and 7 neutrons, so its mass number is 13 (6 protons + 7 neutrons).
- Carbon-14: It has 6 protons and 8 neutrons, so its mass number is 14 (6 protons + 8 neutrons).
As we can see, all three isotopes have the same atomic number (6) because they all have 6 protons. However, their mass numbers differ due to the varying number of neutrons.
Significance of Isotopes:
Isotopes have several practical applications in various fields. Some examples include:
- Carbon dating: Carbon-14 is used to determine the age of ancient artifacts and fossils.
- Medical imaging: Isotopes such as technetium-99m are used in nuclear medicine for diagnostic purposes.
- Radiocarbon dating: Isotopes like carbon-14 are used to determine the age of organic materials.
- Nuclear power: Isotopes like uranium-235 and plutonium-239 are used as fuel in nuclear reactors.
In conclusion, isotopes are atoms with the same atomic number but different mass numbers. The difference in mass numbers is due to the varying number of neutrons in their nuclei.
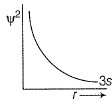
For 2s-orbital electron, radial probability density R2 as function of r (distance) is given by- a)
- b)

- c)

- d)all of these
Correct answer is option 'B'. Can you explain this answer?
For 2s-orbital electron, radial probability density R2 as function of r (distance) is given by
a)
b)
c)
d)
all of these
 | Top Rankers answered |
Correct Answer : b
Explanation : (a) It represents R2 vs r for 1s
(b) It represents R2 vs r for2s
(c) It represents R2 vs r for 2p
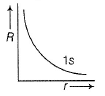
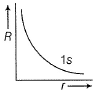
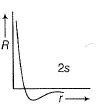
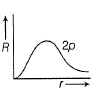
Direction (Q. Nos. 1-12) This section contains 12 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.Q. Radial wave functio ns (R) of different orbitals are plotted. Which is/are correct graphs?- a)
- b)
- c)
- d)All of these
Correct answer is option 'D'. Can you explain this answer?
Direction (Q. Nos. 1-12) This section contains 12 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q. Radial wave functio ns (R) of different orbitals are plotted. Which is/are correct graphs?
a)
b)
c)
d)
All of these
 | Subhankar Mukherjee answered |
For 1s-orbital radial wave function (Ft) is maximum at r - 0, and falls rapidly as r increases thus, (a) correct.
For2s-orbital, radial wave function (R) is maximum at (r = 0), falls to zero and further decreases with r. There appears radial nodes. Thus (b) correct.
For2p-orbital, radial wave function is zero at r = 0, reaches maximum value (at r = a0) and then falls thus (c) is correct.
For2s-orbital, radial wave function (R) is maximum at (r = 0), falls to zero and further decreases with r. There appears radial nodes. Thus (b) correct.
For2p-orbital, radial wave function is zero at r = 0, reaches maximum value (at r = a0) and then falls thus (c) is correct.
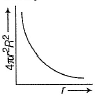
For an electron in 2p-orbital, radial probability function 4πr2R2 as a function of r is given by- a)
- b)
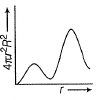
- c)
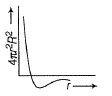
- d)
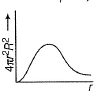
Correct answer is option 'D'. Can you explain this answer?
For an electron in 2p-orbital, radial probability function 4πr2R2 as a function of r is given by
a)
b)
c)
d)
 | Bhavya Joshi answered |
(a) Describes radial wave function as a function of r for 1s
(b) Describes radial probability function as a function of r for 2s
(c) Describes radial wave function as a function r for 2s
(d) Describes radial probability function as a function of r for 2p
(b) Describes radial probability function as a function of r for 2s
(c) Describes radial wave function as a function r for 2s
(d) Describes radial probability function as a function of r for 2p
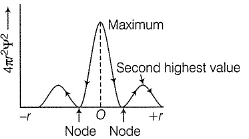
Can you explain the answer of this question below:C onsider the following figures A and B indicating distribution of charge density (electron probability 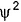 ) with distance r
) with distance r
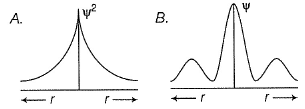
Select the correct statement(s).
- A:
Both A and B are for 1s
- B:
Both A and B are for 2s
- C:
A is for 2s and B is for 1s
- D:
A is for 1s and B is for 2s
The answer is d.
C onsider the following figures A and B indicating distribution of charge density (electron probability ) with distance r
Select the correct statement(s).
Both A and B are for 1s
Both A and B are for 2s
A is for 2s and B is for 1s
A is for 1s and B is for 2s
 | Nandini Nair answered |
in case of 1s, 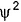 falls as r increases thus, (A) is for 1s.
falls as r increases thus, (A) is for 1s.
In case of 2s,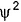 is maximum in the vicinity o f the nucleus, falls to zero giving spherical nodes and then rises to second highest value. Thus, (6) is for2s
is maximum in the vicinity o f the nucleus, falls to zero giving spherical nodes and then rises to second highest value. Thus, (6) is for2s
In case of 2s,
According to quantum mechanics ψ2(r) the wave function squared gives- a)probability density of finding a proton
- b)probability of finding an electron
- c)probability of finding a neutron
- d)probability density of finding an electron
Correct answer is option 'D'. Can you explain this answer?
According to quantum mechanics ψ2(r) the wave function squared gives
a)
probability density of finding a proton
b)
probability of finding an electron
c)
probability of finding a neutron
d)
probability density of finding an electron
 | Isha Chopra answered |
ψ has no physical significance while ψ2 represnts the probability density of finding an electron.
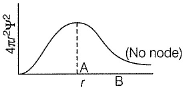
Radial probability density in the occupied orbital of a hydrogen atom in the ground state (1s) is given below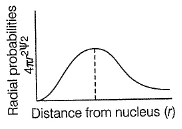
- a)Radial probability is maximum when (r = a0)
- b)Radial probabilit is maximum when
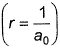
- c)Radial probability almost falls to zero when
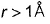
- d)Radial probability is independent of r
Correct answer is option 'A,C'. Can you explain this answer?
Radial probability density in the occupied orbital of a hydrogen atom in the ground state (1s) is given below
a)
Radial probability is maximum when (r = a0)
b)
Radial probabilit is maximum when 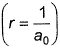
c)
Radial probability almost falls to zero when 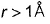
d)
Radial probability is independent of r
 | Aryan Sen answered |
Radial probability increases as r increase reaches a maximum value when r = a0 (Bohr’s radius) and then falls. When 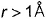 radial probability is very small.
radial probability is very small.
Thus, (a) and (c) are true.
Thus, (a) and (c) are true.
Direction (Q. Nos. 13-15) This section contains 3 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.Q. Which of the following properties can be described by wave function 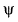 ?
?- a)Type of orbital occupied by electro
- b)Energy of the electron in the orbita
- c)Shape of the orbita
- d)Probability of finding the electron in any particular region within the orbit
Correct answer is option 'A,B,C,D'. Can you explain this answer?
Direction (Q. Nos. 13-15) This section contains 3 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
Q. Which of the following properties can be described by wave function 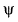 ?
?
a)
Type of orbital occupied by electro
b)
Energy of the electron in the orbita
c)
Shape of the orbita
d)
Probability of finding the electron in any particular region within the orbit
 | Rounak Desai answered |
The wave function describ es properties of the orbital and the electron that occupies the orbital
(a) Type of orbital is described : true
(b) Energy of electron : true
(c) Shape of the orbital: true
(d) Probability: true
(a) Type of orbital is described : true
(b) Energy of electron : true
(c) Shape of the orbital: true
(d) Probability: true
Chapter doubts & questions for Structure of Atom - 30-Day Revision Course for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Structure of Atom - 30-Day Revision Course for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup






