All Exams > NEET > 30-Day Revision Course for NEET > All Questions
All questions of Organic Chemistry: Basic Principles & Techniques for NEET Exam
The observed order of the stability of the cabocation is:- a)(CH3)2CH+ < (CH3)3C+ 3+ < CH3CH2+
- b)CH3+ < CH3CH2+ <(CH3)2CH+ < (CH3)3C+
- c)CH3CH2+ <(CH3)2CH+ < (CH3)3C+ < CH3+
- d)CH3+ < CH3CH2+ < (CH3)3C+ <(CH3)2CH+
Correct answer is option 'B'. Can you explain this answer?
The observed order of the stability of the cabocation is:
a)
(CH3)2CH+ < (CH3)3C+ 3+ < CH3CH2+
b)
CH3+ < CH3CH2+ <(CH3)2CH+ < (CH3)3C+
c)
CH3CH2+ <(CH3)2CH+ < (CH3)3C+ < CH3+
d)
CH3+ < CH3CH2+ < (CH3)3C+ <(CH3)2CH+
| | Rajesh Gupta answered |
Alkyl groups directly attached to the +vely charged carbon stabilize the carbocations due to inductive and hyperconjugation effects.
Inductive effect:
→ Stability of carbocation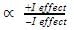
→ More number of +I group more stable carbocation.
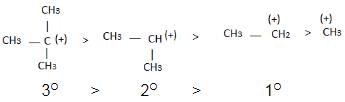
Hyperconjugation:
Inductive effect:
→ Stability of carbocation
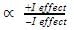
→ More number of +I group more stable carbocation.
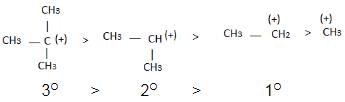
Hyperconjugation:
Stability
∝Number of canonical structures
∝Number of H (alpha hydrogen)
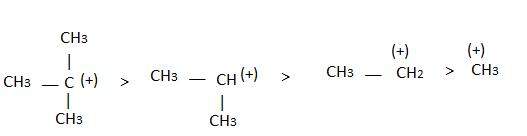
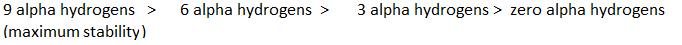
∝Number of canonical structures
∝Number of H (alpha hydrogen)
Heterolytic cleavage is a way to cleave the:- a)Non-ionic bonds
- b)Ionic bonds
- c)Covalent bonds
- d)Polar bonds
Correct answer is option 'C'. Can you explain this answer?
Heterolytic cleavage is a way to cleave the:
a)
Non-ionic bonds
b)
Ionic bonds
c)
Covalent bonds
d)
Polar bonds
| | Rahul Bansal answered |
In heterolytic cleavage, a covalent bond breaks in such a way that one fragment gets both of the shared electrons. In homolytic cleavage, a covalent bond breaks in such a way that each fragment gets one of the shared electrons. The word heterolytic comes from the Greek heteros, "different", and lysis, "loosening".
The organic reaction which proceeds through heterolytic bond cleavage are known as:- a)Covalent reactions
- b)Ionic reactions
- c)Free radical reaction
- d)Polar reactions
Correct answer is option 'B'. Can you explain this answer?
The organic reaction which proceeds through heterolytic bond cleavage are known as:
a)
Covalent reactions
b)
Ionic reactions
c)
Free radical reaction
d)
Polar reactions
| | Hansa Sharma answered |
In heterolytic bond cleavage the bond breaks unevenly and the shared pair of electrons is accommodated by one of the products, which produces one or more ions.As heterolytic bond cleavage gives ions. So the reaction which proceeds through heterolytic bond cleavage is an ionic reaction.
Hence B is the correct answer.
A sequential account of each step, describing details of electron movement, energetics during bond cleavage and bond formation, and the rates of transformation of reactants into products (kinetics) is referred to as:- a)Reaction mechanism
- b)Reaction kinetics
- c)Thermodynamics
- d)Equation
Correct answer is option 'A'. Can you explain this answer?
A sequential account of each step, describing details of electron movement, energetics during bond cleavage and bond formation, and the rates of transformation of reactants into products (kinetics) is referred to as:
a)
Reaction mechanism
b)
Reaction kinetics
c)
Thermodynamics
d)
Equation
 | Rounak Desai answered |
Reaction Mechanism
The correct answer is option 'A', reaction mechanism. A reaction mechanism provides a detailed account of each step involved in a chemical reaction, including the movement of electrons, the energetics during bond cleavage and bond formation, and the rates of transformation of reactants into products. It explains how reactant molecules rearrange and bond with each other to form products.
Electron Movement
In a chemical reaction, electrons play a crucial role in the formation and breaking of chemical bonds. The movement of electrons determines the reactivity and stability of molecules. The reaction mechanism describes how electrons are transferred or shared between atoms during a reaction. It explains the movement of electrons from high energy orbitals to low energy orbitals, leading to the formation of new bonds and the breaking of existing bonds.
Energetics during Bond Cleavage and Bond Formation
During a chemical reaction, bonds between atoms are broken and new bonds are formed. The reaction mechanism explains the energy changes associated with these processes. Bond cleavage requires an input of energy, known as bond dissociation energy, as bonds are broken. This energy is absorbed from the surroundings. On the other hand, bond formation releases energy, known as bond formation energy, as new bonds are created. The reaction mechanism describes the energy changes during these processes and how they influence the overall energetics of the reaction.
Rates of Transformation (Kinetics)
The reaction mechanism also provides information about the rates of transformation of reactants into products. It describes the individual steps involved in the reaction and the order in which they occur. Each step in the mechanism has a specific rate constant associated with it, which determines how fast that step proceeds. The overall rate of the reaction is determined by the slowest step, known as the rate-determining step. The reaction mechanism helps to identify this step and understand the factors that control the reaction rate.
Conclusion
In summary, a reaction mechanism is a detailed account of the steps involved in a chemical reaction. It describes the movement of electrons, the energetics during bond cleavage and bond formation, and the rates of transformation of reactants into products. Understanding the reaction mechanism is crucial for predicting and controlling chemical reactions.
The correct answer is option 'A', reaction mechanism. A reaction mechanism provides a detailed account of each step involved in a chemical reaction, including the movement of electrons, the energetics during bond cleavage and bond formation, and the rates of transformation of reactants into products. It explains how reactant molecules rearrange and bond with each other to form products.
Electron Movement
In a chemical reaction, electrons play a crucial role in the formation and breaking of chemical bonds. The movement of electrons determines the reactivity and stability of molecules. The reaction mechanism describes how electrons are transferred or shared between atoms during a reaction. It explains the movement of electrons from high energy orbitals to low energy orbitals, leading to the formation of new bonds and the breaking of existing bonds.
Energetics during Bond Cleavage and Bond Formation
During a chemical reaction, bonds between atoms are broken and new bonds are formed. The reaction mechanism explains the energy changes associated with these processes. Bond cleavage requires an input of energy, known as bond dissociation energy, as bonds are broken. This energy is absorbed from the surroundings. On the other hand, bond formation releases energy, known as bond formation energy, as new bonds are created. The reaction mechanism describes the energy changes during these processes and how they influence the overall energetics of the reaction.
Rates of Transformation (Kinetics)
The reaction mechanism also provides information about the rates of transformation of reactants into products. It describes the individual steps involved in the reaction and the order in which they occur. Each step in the mechanism has a specific rate constant associated with it, which determines how fast that step proceeds. The overall rate of the reaction is determined by the slowest step, known as the rate-determining step. The reaction mechanism helps to identify this step and understand the factors that control the reaction rate.
Conclusion
In summary, a reaction mechanism is a detailed account of the steps involved in a chemical reaction. It describes the movement of electrons, the energetics during bond cleavage and bond formation, and the rates of transformation of reactants into products. Understanding the reaction mechanism is crucial for predicting and controlling chemical reactions.
Which among the following is a very unstable and reactive species:- a)Carbaanion
- b)Polar ions
- c)Carbocation
- d)Free radical
Correct answer is option 'C'. Can you explain this answer?
Which among the following is a very unstable and reactive species:
a)
Carbaanion
b)
Polar ions
c)
Carbocation
d)
Free radical
 | Geethika Reddy answered |
Actually carbanions are filled with octet, so they are stable and less reactive, in case of polar ions already they are stable, and free radicle is heptet i.e, near to octet whereas carbocation is sextet in nature, so in order to gain to octet it is more reactive and less stable
In a free radical reaction, free radicals are formed at- a)initiation step
- b)propagation step
- c)termination step
- d)both A and B
Correct answer is option 'D'. Can you explain this answer?
In a free radical reaction, free radicals are formed at
a)
initiation step
b)
propagation step
c)
termination step
d)
both A and B
 | Nitin Nair answered |
Once a reactive free radical is generated, it can react with stable molecules to form new free radicals. These new free radicals go on to generate yet more free radicals, and so on. Propagation steps often involve hydrogen abstraction or addition of the radical to double bonds.
A carbon species carrying a negative charge on carbon atom is known as:- a)Polar ions
- b)Carbanion
- c)Free radical
- d)Carbocation
Correct answer is option 'B'. Can you explain this answer?
A carbon species carrying a negative charge on carbon atom is known as:
a)
Polar ions
b)
Carbanion
c)
Free radical
d)
Carbocation
 | Nitin Nair answered |
A carbanion has a negatively charged, trivalent carbon atom that has eight electrons in its valence shell. Thus, a carbanion is not electron deficient. As a result, carbanions have pyramidal geometry. Carbocations, radicals, and carbanions can be stabilized by resonance.
Chapter doubts & questions for Organic Chemistry: Basic Principles & Techniques - 30-Day Revision Course for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Organic Chemistry: Basic Principles & Techniques - 30-Day Revision Course for NEET in English & Hindi are available as part of NEET exam. Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup




